Abstract
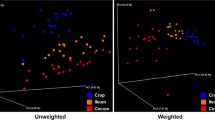
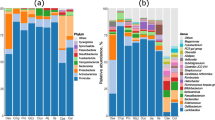
The divergence of gut bacterial community on broiler chickens has been reported as potentially possible keys to enhancing nutrient absorption, immune systems, and increasing poultry health and performance. Thus, we compared cecal bacterial communities and functional predictions by sex and body weight regarding the association between cecal microbiota and chicken growth performance. In this study, a total of 12 male and 12 female 1-day-old broiler chickens were raised for 35 days in 2 separate cages. Chickens were divided into 3 subgroups depending on body weight (low, medium, and high) by each sex. We compared chicken cecal microbiota compositions and its predictive functions by sex and body weight difference. We found that bacterial 16S rRNA genes were classified as 3 major phyla (Bacteroidetes, Firmicutes, and Proteobacteria), accounting for > 98% of the total bacterial community. The profiling of different bacterial taxa and predictive metagenome functions derived from 16S rRNA genes were performed over chicken sex and bodyweight. Male chickens were related to the enrichment of Bacteroides while female chickens were to the enrichment of Clostridium and Shigella. Male chickens with high body weight were associated with the enrichment of Faecalibacterium and Shuttleworthia. Carbohydrate and lipid metabolisms were suggested as candidate functions for weight gain in the males. This suggests that the variation of cecal bacterial communities and their functions by sex and body weight may be associated with the differences in the growth potentials of broiler chickens.
Similar content being viewed by others
References
Bokulich., N.A., Rideout, J.R., Kopylova, E., Bolyen, E., Patnode, J., Ellett, Z., McDonald, D., Wolfe, B., Maurice, C.F., Dutton, R.J., et al. 2015. A standardlized, extensible framework for optimizing classification improves marker-gene taxonomic assignments. PeerJ. Prepr. 230313, 1–17.
Burt, D.W. 2007. Emergence of the chicken as a model organism: implications for agriculture and biology. Poult. Sci. 86, 1460–1471.
Chang, Q., Luan, Y., and Sun, F. 2011. Variance adjusted weighted UniFrac: a powerful beta diversity measure for comparing communities based on phylogeny. BMC Bioinformatics 12, 118.
Cho, I., Yamanishi, S., Cox, L., Methé, B.A., Zavadil, J., Li, K., Gao, Z., Mahana, D., Raju, K., Teitler, I., et al. 2012. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 488, 621–626.
Choi, J.H., Kim, G.B., and Cha, C.J. 2014. Spatial heterogeneity and stability of bacterial community in the gastrointestinal tracts of broiler chickens. Poult. Sci. 93, 1942–1950.
Choi, K.Y., Lee, T.K., and Sul, W.J. 2015. Metagenomic analysis of chicken gut microbiota for improving metabolism and health of chickens - A review. Asian-Australasian J. Anim. Sci. 28, 1217–1225.
Clarke, S.F., Murphy, E.F., Nilaweera, K., Ross, P.R., Shanahan, F., O’Toole, P.W., and Cotter, P.D. 2012. The gut microbiota and its relationship to diet and obesity: new insights. Gut Microbes 3, 186–202.
Conterno, L., Fava, F., Viola, R., and Tuohy, K.M. 2011. Obesity and the gut microbiota: Does up-regulating colonic fermentation protect against obesity and metabolic disease? Genes Nutr. 6, 241–260.
De Filippo, C., Cavalieri, D., Di Paola, M., Ramazzotti, M., Poullet, J.B., Massart, S., Collini, S., Pieraccini, G., and Lionetti, P. 2010. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 107, 14691–14696.
Edgar, R.C. 2010. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461.
Eren, A.M., Vineis, J.H., Morrison, H.G., and Sogin, M.L. 2013. A filtering method to generate high quality short reads using illumina paired-end technology. PLoS One 8, e66643.
Faith, D.P. and Baker, A.M. 2006. Phylogenetic diversity (PD) and biodiversity conservation: some bioinformatics challenges. Evol. Bioinform. 2, 121–128.
Fei, N. and Zhao, L. 2013. An opportunistic pathogen isolated from the gut of an obese human causes obesity in germfree mice. ISME J. 7, 880–884.
Guo, X., Xia, X., Tang, R., Zhou, J., Zhao, H., and Wang, K. 2008. Development of a real-time PCR method for Firmicutes and Bacteroidetes in faeces and its application to quantify intestinal population of obese and lean pigs. Lett. Appl. Microbiol. 47, 367–373.
Kogut, M.H. 2013. The gut microbiota and host innate immunity: Regulators of host metabolism and metabolic diseases in poultry? J. Appl. Poult. Res. 22, 637–646.
Lan, Y., Verstegen, M., Tamminga, S., and Williams, B.A. 2005. The role of the commensal gut microbial community in broiler chickens. Worlds Poult. Sci. J. 61, 95–104.
Langille, M., Zaneveld, J., Caporaso, J.G., McDonald, D., Knights, D., Reyes, J., Clemente, J., Burkepile, D., Vega Thurber, R., Knight, R., et al. 2013. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat. Biotechnol. 31, 814–821.
Ley, R.E., Turnbaugh, P.J., Klein, S., and Gordon, J.I. 2006. Microbial ecology: human gut microbes associated with obesity. Nature 444, 1022–1023.
Lozupone, C. and Knight, R. 2005. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 71, 8228–8235.
Lumpkins, B.S., Batal, A.B., and Lee, M. 2008. The effect of gender on the bacterial community in the gastrointestinal tract of broilers. Poult. Sci. 87, 964–967.
McDonald, D., Price, M.N., Goodrich, J., Nawrocki, E.P., DeSantis, T.Z., Probst, A., Andersen, G.L., Knight, R., and Hugenholtz, P. 2012. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J. 6, 610–618.
Neufeld, J.D. and Mohn, W.W. 2005. Unexpectedly high bacterial diversity in arctic tundra relative to boreal forest soils, revealed by serial analysis of ribosomal sequence tags. Appl. Environ. Microbiol. 71, 5710–5718.
Nikiforova, V.J., Giesbertz, P., Wiemer, J., Bethan, B., Looser, R., Liebenberg, V., Ruiz Noppinger, P., Daniel, H., and Rein, D. 2014. Glyoxylate, a new marker metabolite of type 2 diabetes. J. Diabetes Res. 2014, 1–9.
Niu, Q., Li, P., Hao, S., Zhang, Y., Kim, S.W., Li, H., Ma, X., Gao, S., He, L., Wu, W., et al. 2015. Dynamic distribution of the gut microbiota and the relationship with apparent crude fiber digestibility and growth stages in pigs. Sci. Rep. 5, 9938.
Okeke, F., Roland, B.C., and Mullin, G.E. 2014. The role of the gut microbiome in the pathogenesis and treatment of obesity. Glob. Adv. Heal. Med. 3, 44–57.
Pourabedin, M. and Zhao, X. 2015. Prebiotics and gut microbiota in chickens. FEMS Microbiol. Lett. 362, 1–8.
R Developement Core Team. 2015. R: A language and environment for statistical computing. R Found. Stat. Comput. 1, 409.
Rinttilä, T. and Apajalahti, J. 2013. Intestinal microbiota and metabolites–Implications for broiler chicken health and performance. J. Appl. Poult Res. 22, 647–658.
Schumacher, M., Mattern, C., Ghoumari, A., Oudinet, J.P., Liere, P., Labombarda, F., Sitruk-Ware, R., De Nicola, A.F., and Guennoun, R. 2014. Revisiting the roles of progesterone and allopregnanolone in the nervous system: Resurgence of the progesterone receptors. Prog. Neurobiol. 113, 6–39.
Segata, N., Izard, J., Waldron, L., Gevers, D., Miropolsky, L., Garrett, W.S., and Huttenhower, C. 2011. Metagenomic biomarker discovery and explanation. Genome Biol. 12, R60.
Sharpton, T.J. 2014. An introduction to the analysis of shotgun metagenomic data. Front. Plant Sci. 5, 209.
Singh, P., Karimi, A., Devendra, K., Waldroup, P.W., Cho, K.K., and Kwon, Y.M. 2013. Influence of penicillin on microbial diversity of the cecal microbiota in broiler chickens. Poult. Sci. 92, 272–276.
Singla, P., Bardoloi, A., and Parkash, A.A. 2010. Metabolic effects of obesity: A review. World J. Diabetes 1, 76–88.
Tremaroli, V. and Bäckhed, F. 2012. Functional interactions between the gut microbiota and host metabolism. Nature 489, 242–249.
Torok, V.A., Dyson, C., McKay, A., and Ophel-Keller, K. 2013. Quantitative molecular assays for evaluating changes in broiler gut microbiota linked with diet and performance. Anim. Prod. Sci. 53, 1260–1268.
Torok, V.A., Hughes, R.J., Mikkelsen, L.L., Perez-Maldonado, R., Balding, K., MacAlpine, R., Percy, N.J., and Ophel-Keller, K. 2011. Identification and characterization of potential performancerelated gut microbiotas in broiler chickens across various feeding trials. Appl. Environ. Microbiol. 77, 5868–5878.
Turnbaugh, P.J., Ley, R.E., Mahowald, M.A., Magrini, V., Mardis, E.R., and Gordon, J.I. 2006. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031.
Voreades, N., Kozil, A., and Weir, T.L. 2014. Diet and the development of the human intestinal microbiome. Front. Microbiol. 5, 1–9.
Wei, S., Morrison, M., and Yu, Z. 2013. Bacterial census of poultry intestinal microbiome. Poult. Sci. 92, 671–683.
Wise, M.G. and Siragusa, G.R. 2007. Quantitative analysis of the intestinal bacterial community in one- to three-week-old commercially reared broiler chickens fed conventional or antibioticfree vegetable-based diets. J. Appl. Microbiol. 102, 1138–1149.
Zhao, L., Wang, G., Siegel, P., He, C., Wang, H., Zhao, W., Zhai, Z., Tian, F., Zhao, J., Zhang, H., et al. 2013. Quantitative genetic background of the host influences gut microbiomes in chickens. Sci. Rep. 3, 1163.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lee, KC., Kil, D.Y. & Sul, W.J. Cecal microbiome divergence of broiler chickens by sex and body weight. J Microbiol. 55, 939–945 (2017). https://doi.org/10.1007/s12275-017-7202-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-017-7202-0