Abstract
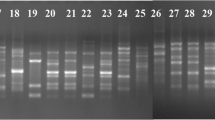
Twenty one isolates of Fusarium oxysporum f. sp. psidii (Fop), causing a vascular wilt in guava (Psidium guajava L.), were collected from different agro-ecological regions of India. The pathogenicity test was performed in guava seedlings, where the Fop isolates were found to be highly pathogenic. All 21 isolates were confirmed as F. oxysporum f. sp. psidii by a newly developed, species-specific primer against the conserved regions of 28S rDNA and the intergenic spacer region. RAPD and PCR-RFLP were used for genotyping the isolates to determine their genetic relationships. Fifteen RAPD primers were tested, of which five primers produced prominent, polymorphic, and reproducible bands. RAPD yielded an average of 6.5 polymorphic bands per primer, with the amplified DNA fragments ranging from 200–2,000 bp in size. A dendrogram constructed from these data indicated a 22–74% level of homology. In RFLP analysis, two major bands (350 and 220 bp) were commonly present in all isolates of F. oxysporum. These findings provide new insight for rapid, specific, and sensitive disease diagnosis. However, genotyping could be useful in strain-level discrimination of isolates from different agro-ecological regions of India.
Similar content being viewed by others
References
Abd-Elsalam, K.A., Schnieder, F., Khalil, M.S., Aly, A.A., and Verreet, J.A. 2003. Genetic variation at the intra- and interspecific level in Fusarium spp. associated from Egyptian cottons. J. Plant Dis. Prot. 110, 46–53.
Achenback, L.A., Patrick, J.A., and Gray, L.E. 1997. Genetic homogeneity among isolates of Fusarium solani that cause soybean sudden death syndrome. Theor. Appl. Genet. 95, 474–478.
Annamalai, P., Ishii, H., Lalithakumari, D., and Revathi, R. 1995. Polymerase chain reaction and its applications in fungal disease diagnosis. J. Plant Dis. Protect. 102, 91–104.
Appel, D.J. and Gordon, T.R. 1996. Relationships among pathogenic and nonpathogenic isolates of Fusarium oxysporum based on the partial sequence of the intergenic spacer region of the ribosomal DNA. Mol. Plant Microbe Interact. 9, 125–138.
Assigbetse, K.B., Fernandez, D., Doubios, M.P., and Geiger, J.P. 1994. Differentiation of Fusarium oxysporum f. sp. vasinfectum races on cotton by random amplified polymorphic DNA (RAPD) analysis. Phytopathol. 84, 622–626.
Bruns, T.D., White, T.J., and Taylor, J.W. 1991. Fungal molecular systematics. Ann. Rev. Ecol. Sys. 22, 525–564.
Bryan, G.T., Daniels, M.J., and Osbourn, A.E. 1995. Comparison of fungi within the Gaeumannomyces-Phialophora complex by analysis of ribosomal DNA sequence. Appl. Environ. Microbiol. 61, 681–689.
Cenis, J.L. 1992. Rapid extraction of fungal DNA for PCR amplification. Nucleic Acids Res. 20, 2380.
Chattapadhyay, S.B. and Bhattachariya, S.K. 1968. Investigations on the wilt disease of guava (Psidium guajava L.) in West Bengal I. Ind. J. Agri. Sci. 38, 65–72.
Danaldson, G.C., Ball, L.A., Axelrood, P.E., and Glass, N.L. 1995. Primer sets developed to amplify conserved genes from filamentous ascomycetes are useful in differentiating Fusarium species associated with conifers. Appl. Environ. Microbiol. 61, 1331–1340.
Edel, V., Steinberg, C., Avelange, I., Lagurre, G., and Alabouvette, C. 1995. Comparison of three molecular methods for the characterization of Fusarium oxysporum strains. Phytopathol. 85, 579–585.
Guadet, J., Julien, J., Lafay, J.F., and Brygoo, Y. 1989. Phylogeny of some Fusarium species, as determined by large-subunit rRNA sequence comparison. Mol. Biol. Ecol. 6, 227–242.
Gupta, V.K. 2012. PCR-RAPD profiling of Fusarium spp. causing guava wilt disease in India. J. Environ. Sci. Health Part B. 47, 315–325.
Hyun, J.W. and Clark, C.A. 1998. Analysis of Fusarium lateritium using RAPD and rDNA RFLP techniques. Mycol. Res. 102, 1259–1264.
Ibrahim, G. and Nirenberg, H.I. 2000. Recent studies on Fusarium vascular wilt of cotton at the Fedral Biological Center for Agricultural and Forstery (BBA), Berlin. — Mitt. Biol. BundAnst Ld- u.Forstwirtsch H. 377, 87–88.
Jana, T.K., Sharma, T.R., Prasad, D., and Arora, D.K. 2003. Molecular characterization of Macrophomina phaseolina and Fusarium species by single primer RAPD technique. Microbiol. Res. 158, 264–274.
Jurgenson, J.E., Zeller, K.A., and Le slie, J.F. 2002. Expanded genetic map of Gibberella moniliformis (Fusarium verticillioides). Appl. Environ. Microbiol. 68, 1972–1979.
Leong, S.K., Latiffah, Z., and Baharuddin, S. 2009. Molecular characterization of Fusarium oxysporum f. sp. cubense of banana. Ameri. J. Appl. Sci. 6, 1301–1307.
Manulis, S., Kogan, N., Reuven, M., and Benyephet, Y. 1994. Use of the RAPD technique for identification of Fusarium oxysporum f. sp. dianthi from carnation. Phytopathol. 84, 98–101.
McDonald, B.A. 1997. The population genetics of fungi: tools and techniques. Phytopathol. 87, 448–453.
Migheli, Q., Briatore, E., and Garibaldi, A. 1998. Use of random amplified polymorphic DNA (RAPD) to identify races 1, 2, 3 and 8 of Fusarium oxysporum f. sp. dianthi in Italy. Eur. J. Plant Pathol. 104, 49–57.
Misra, A.K. 2006 Wilt of guava-a disease of national importance. Ind. Phytopathol. 59, 269–280.
Misra, A.K. and Pandey, B.K. 1996. Wilt of guava and associated pathogens. Ind. J. Mycol. Plant Pathol. 22, 85–86.
Misra, A.K. and Pandey, B.K. 2000. Progressive natural wilting of guava plants during different months. Indian Phytopathol. 53, 423–427.
Mishra, R.K., Pandey, B.K., Muthukumar, M., Pathak, N., and Zeeshan, M. 2013. Detection of Fusarium wilt pathogens of Psidium guajava L. in soil using culture independent PCR (ciPCR). Saudi J. Biol. Sci. 20, 51–56.
NHB. 2012. Indian horticulture database. p. 4.
Nicholson, P., Simpson, D.R., Weston, G., Rezanoor, H.N., Lees, A.K., Parry, D.W., and Joyce, D. 1998. Detection and quantification of Fusarium culmorum and Fusarium graminearum in cereals using PCR assays. Physiol. Mol. Plant Pathol. 53, 17–37.
O’Donnell, K., Cigelnik, E., and Nirenberg, H.I. 1998a. Molecular systematic and phylogeography of the Gibberella fujikuroi species complex. Mycologia 90, 465–493.
O’Donnell, K., Kistler, H.C., Tacke, B.K., and Casper, H.H. 2000. Gene genealogies reveal global phylogeographic structure and reproductive isolation among lineages of Fusarium graminearum, the fungus causing wheat scab. Proc. Natl. Acad. Sci. USA 97, 7905–7910.
Prasad, N., Mehta, P.R., and Lal, S.B. 1952. Fusarium wilt of guava (Psidium guajava L.) in Uttar Pradesh, India. Nature 169, 753.
Rajan, S., Yadava, L.P., Kumar, R., and Saxena, S.K. 2007. GIS based diversity analysis of guava growing distribution in Uttar Pradesh. Acta Horti. 735, 109–113.
Rohlf, F.J. 1998. NT-SYS-pc: Numerical Taxonomy and Multivariate Analysis System. Version 2.11V, Exteer Software, Setauket, N.Y., USA.
Samuels, G.J. and Seifert, K.A. 1995. The impact of molecular characters on systematics of filamentous ascomycetes. Ann. Rev. Phytopathol. 33, 37–67.
Schroers, H.J., Geldenhuis, M.M., Wingfield, M.J., Schoeman, M.H., Yen, Y.F., Shen, W.C., and Wingfield, B.D. 2005. Classification of the guava wilt fungus Myxosporium psidii, the palm pathogen Gliocladium vermoesenii and the persimmon wilt fungus Acremonium diospyri in Nalanthamala. Mycologia 97, 375–395.
Voight, K., Schleier, S., and Bruckner, B. 1995. Genetic variability in Gibberella fujikuroi and some related species of the genus Fusarium based on random amplification of polymorphic DNA (RAPD). Curr. Genet. 27, 528–535.
White, T.J., Bruns, T., Lee, S., and Taylor, J.W. 1990. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics, pp. 315–322. PCR Protocols: A Guide to Methods and Applications. In Innis, M.A., Gelfand, D.H., Sninsky, J.J., and White, T.J. (eds.) Academic Press, Inc., New York, N.Y., USA.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Mishra, R.K., Pandey, B.K., Singh, V. et al. Molecular detection and genotyping of Fusarium oxysporum f. sp. psidii isolates from different agro-ecological regions of India. J Microbiol. 51, 405–412 (2013). https://doi.org/10.1007/s12275-013-2638-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-013-2638-3