Abstract
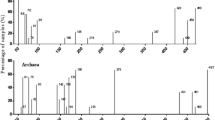
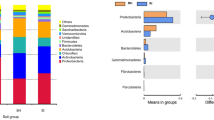
Microbial communities in hot pepper (Capsicum annuum L.) cultivation fields under different cultivation methods were investigated by terminal restriction fragment length polymorphism (T-RFLP) analysis. Rhizosphere soil and leaf samples were collected from control, conventional and nature-friendly cultivation fields between May and July, 2009. Two Bacillus subtilis strains were applied to nature-friendly cultivation fields as biocontrol agents during the sampling period. Relative abundances of bacteria and plant pathogenic fungi related T-RFs were also measured to monitor the effect of biocontrol agents on potential plant pathogenic fungi. In the principal component analysis (PCA) based on T-RFLP profiles, the microbial communities from rhizosphere soil samples in July, including bacteria and fungi, showed distinct difference between nature-friendly cultivation fields and other cultivation fields. However, there was no correlation between cultivation methods and leaf microbial communities at any sampling period. Changes in the abundance of bacteria related T-RF in the rhizosphere of nature-friendly cultivation fields were observed clearly two months after application of biocontrol agent, while the abundance of plant pathogenic fungi related T-RFs significantly decreased.
Similar content being viewed by others
References
Bankhead, S.B., B.B. Landa, E. Lutton, D.M. Weller, and B. McSpadden Gardener. 2004. Minimal changes in rhizobacterial population structure following root colonization by wild type and transgenic biocontrol strains. FEMS Microbiol. Ecol. 49, 307–318.
Benítez, M. and B. McSpadden Gardener. 2009. Linking sequence to function in soil bacteria: sequence-directed isolation of novel bacteria contributing to soilborne plant disease suppression. Appl. Environ. Microbiol. 75, 915–924.
Benítez, M., F.B. Tustasa, D. Rotenberga, M.D. Kleinhenzb, J. Cardinab, D. Stinnerc, S.A. Millera, and B. McSpadden Gardener. 2007. Multiple statistical approaches of community fingerprint data reveal bacterial populations associated with general disease suppression arising from the application of different organic field management strategies. Soil Biol. Biochem. 39, 2289–2301.
Berg, G., A. Krechel, M. Ditz, R.A. Sikora, A. Ulrich, and J. Hallmann. 2005. Endophytic and ectophytic potato-associated bacterial communities differ in structure and antagonistic function against plant pathogenic fungi. FEMS Microbiol. Ecol. 51, 215–229.
Blackwood, C.B., T. Marsh, S.H. Kim, and E.A. Paul. 2003. Terminal restriction fragment length polymorphism data analysis for quantitative comparison of microbial communities. Appl. Environ. Microbiol. 69, 926–932.
Borneman, J. and J.O. Becker. 2007. Identifying microorganisms involved in specific pathogen suppression in soil. Annu. Rev. Phytopathol. 45, 153–172.
Castro, H., S. Newman, K.R. Reddy, and A. Ogram. 2005. Distribution and stability of sulfate-reducing prokaryotic and hydrogenotrophic methanogenic assemblages in nutrient-impacted regions of the Florida Everglades. Appl. Environ. Microbiol. 71, 2695–2704.
Chelius, M.K. and E.W. Triplett. 2001. The diversity of archaea and bacteria in association with the roots of Zea mays L. Microb. Ecol. 41, 252–263.
Conn, V.M. and C.M.M. Franco. 2004. Effect of microbial inoculants on the indigenous actinobacterial endophytic population in the roots of wheat as determined by terminal restriction fragment length polymorphism. Appl. Environ. Microbiol. 70, 6407–6413.
Culman, S.W., H.G. Gauch, C.B. Blackwood, and J.E. Thies. 2008. Analysis of T-RFLP data using analysis of variance and ordination methods: a comparative study. J. Microbiol. Methods 75, 55–63.
Duncan, D.B. 1955. Multiple range and multiple F test. Biometrics 11, 1–42.
Edel-Hermmann, V., C. Dreumont, A. Pérez-Piqueres, and C. Steinberg. 2004. Terminal restriction fragment length polymorphism analysis of ribosomal RNA genes to assess changes in fungal community structure in soils. FEMS Microbiol. Ecol. 47, 397–404.
Fravel, D. 2005. Commercialization and implementation of biocontrol. Annu. Rev. Phytopathol. 43, 337–359.
Haas, D. and G. Defago. 2005. Biological control of soil-borne pathogens by fluorescent pseudomonads. Nature Rev. Microbiol. 3, 307–319.
Hjort, K., A. Lembke, A. Speksnijder, K. Smalla, and J.K. Jansson. 2007. Community structure of actively growing bacterial populations in plant pathogen suppressive soil. Microb. Ecol. 53, 399–413.
Jernberg, C., A. Sullivan, C. Edlund, and J.K. Jansson. 2005. Monitoring of antibiotic-induced alterations in the human intestinal microflora and detection of probiotic strains by use of terminal restriction fragment length polymorphism. Appl. Environ. Microbiol. 71, 501–506.
Joshi, R. and B. McSpadden Gardener. 2006. Identification and characterization of novel genetic markers associated with biological control activities of Bacillus subtilis. Phytopathology 96, 145–154.
Larena, I. and P. Melgarejo. 2009. Development of a new strategy for monitoring Epicoccum nigrum 282, a biological control agent used against brown rot caused by Monilinia spp. in peaches. Postharvest Biol. Technol. 54, 63–71.
Liu, W.T., T.L. Marsh, H. Cheng, and L.J. Forney. 1997. Characterization of microbial diversity by determining terminal restriction fragment length polymorphisms of genes encoding 16S rRNA. Appl. Environ. Microbiol. 63, 4516–4522.
Manter, D.K. and J.M. Vivanco. 2007. Use of the ITS primers, ITS1F and ITS4, to characterize fungal abundance and diversity in mixed-template samples by qPCR and length heterogeneity analysis. J. Microbiol. Methods 71, 7–14.
Mazzola, M. 2004. Assessment and management of soil microbial community structure for disease suppression. Annu. Rev. Phytopathol. 42, 35–59.
McSpadden Gardener, B. and D. Weller. 2001. Changes in populations of rhizosphere bacteria associated with take-all disease of wheat. Appl. Environ. Microbiol. 67, 4414–4425.
Paulitz, T. and R. Belanger. 2001. Biological control in greenhouse systems. Annu. Rev. Phytopathol. 39, 103–133.
Pujol, M., E. Badosa, C. Manceau, and E. Montesinos. 2006. Assessment of the environmental fate of the biological control agent of fire blight, Pseudomonas fluorescens EPS62e, on apple by culture and real-time PCR methods. Appl. Environ. Microbiol. 72, 2421–2427.
Sánchez, J.I., L. Rossetti, B. Martínez, A. Rodríguez, and G. Giraffa. 2006. Application of reverse transcriptase PCR-based T-RFLP to perform semi-quantitative analysis of metabolically active bacteria in dairy fermentations. J. Microbiol. Methods 65, 268–277.
Schena, L., M.F. Sialer, and D. Gallitelli. 2002. Molecular detection of strain L47 of Aureobasidium pullulans, a biocontrol agent of postharvest diseases. Plant Dis. 86, 54–60.
Wang, M., S. Ahrné, M. Antonsson, and G. Molin. 2004. T-RFLP combined with principle component analysis and 16S rRNA gene sequencing: an effective strategy for comparison of fecal microbiota in infants of different ages. J. Microbiol. Methods 59, 53–69.
Whipps, J.M. 2001. Microbial interactions and biocontrol in the rhizosphere. J. Exp. Bot. 52, 487–511.
Yin, B., L. Valinsky, X. Gao, J.O. Becker, and J. Borneman. 2003. Identification of fungal rDNA associated with soil suppressiveness against Heterodera schachtii using oligonucleotide fingerprinting. Phytopathol. 93, 1006–1013.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, Y.T., Cho, M., Jeong, J.Y. et al. Application of terminal restriction fragment length polymorphism (T-RFLP) analysis to monitor effect of biocontrol agents on rhizosphere microbial community of hot pepper (Capsicum annuum L.). J Microbiol. 48, 566–572 (2010). https://doi.org/10.1007/s12275-010-0126-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-010-0126-6