Abstract
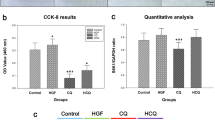
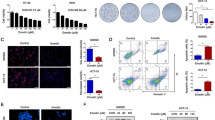
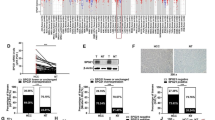
Hepatocellular carcinoma (HCC) is one of the most common tumor types globally. Despite the progress made in surgical procedures and therapeutic options, HCC remains a considerable cause of cancer-related mortality. In this study, we investigated the antitumor effects of sanguinarine (Sang) on HCC and its potential mechanisms. Our findings showed that Sang impairs the acidic environment of lysosomes by inhibiting cathepsin D maturation. In addition, Sang inhibited the formation of autolysosomes in RFP-GFP-LC3 transfected cells, subsequently suppressing late mitophagy. Sang also induced reactive oxygen species (ROS)-dependent autophagy and apoptosis in HCC cells, which was significantly attenuated following treatment with a ROS scavenger. Further investigation using autophagy inhibitors revealed that sanguinarine-induced mitochondrial dysfunction and mitophagy led to mitochondrial apoptosis in HCC cells. Immunohistochemical staining of sanguinarine-treated xenograft samples revealed that it initiated and blocked autophagy. In summary, our findings suggest that in HCC cells, Sang impairs lysosomal function and induces ROS-dependent mitophagy and apoptosis.





Similar content being viewed by others
References
Adams JM, Cory S (1998) The Bcl-2 protein family: arbiters of cell survival. Science (New York, NY) 281(5381):1322–1326. https://doi.org/10.1126/science.281.5381.1322
Allard B, Aspeslagh S, Garaud S, Dupont FA, Solinas C, Kok M, Routy B, Sotiriou C, Stagg J, Buisseret L (2018) Immuno-oncology-101: overview of major concepts and translational perspectives. Semin Cancer Biol 52(Pt 2):1–11. https://doi.org/10.1016/j.semcancer.2018.02.005
Anwanwan D, Singh SK, Singh S, Saikam V, Singh R (2020) Challenges in liver cancer and possible treatment approaches. Biochim Biophys Acta Rev Cancer 1873(1):188314–188331. https://doi.org/10.1016/j.bbcan.2019.188314
Chen Y, Chen HN, Wang K, Zhang L, Huang Z, Liu J, Zhang Z, Luo M, Lei Y, Peng Y, Zhou ZG, Wei Y, Huang C (2019) Ketoconazole exacerbates mitophagy to induce apoptosis by downregulating cyclooxygenase-2 in hepatocellular carcinoma. J Hepatol 70(1):66–77. https://doi.org/10.1016/j.jhep.2018.09.022
Fivenson EM, Lautrup S, Sun N, Scheibye-Knudsen M, Stevnsner T, Nilsen H, Bohr VA, Fang EF (2017) Mitophagy in neurodegeneration and aging. Neurochem Int 109:202–209. https://doi.org/10.1016/j.neuint.2017.02.007
Glasauer A, Chandel NS (2014) Targeting antioxidants for cancer therapy. Biochem Pharmacol 92(1):90–101. https://doi.org/10.1016/j.bcp.2014.07.017
Kalpage HA, Bazylianska V, Recanati MA, Fite A, Liu J, Wan J, Mantena N, Malek MH, Podgorski I, Heath EI, Vaishnav A, Edwards BF, Grossman LI, Sanderson TH, Lee I, Huttemann M (2019) Tissue-specific regulation of cytochrome c by post-translational modifications: respiration, the mitochondrial membrane potential, ROS, and apoptosis. FASEB J 33(2):1540–1553. https://doi.org/10.1096/fj.201801417R
Kimura S, Noda T, Yoshimori T (2007) Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3(5):452–460. https://doi.org/10.4161/auto.4451
Lemasters JJ (2014) Variants of mitochondrial autophagy: types 1 and 2 mitophagy and micromitophagy (type 3). Redox Biol 2:749–754. https://doi.org/10.1016/j.redox.2014.06.004
Li L, Tan J, Miao Y, Lei P, Zhang Q (2015) ROS and autophagy: interactions and molecular regulatory mechanisms. Cell Mol Neurobiol 35(5):615–621. https://doi.org/10.1007/s10571-015-0166-x
Li W, Li Y, Siraj S, Jin H, Fan Y, Yang X, Huang X, Wang X, Wang J, Liu L, Du L, Chen Q (2019) FUN14 domain-containing 1-mediated mitophagy suppresses hepatocarcinogenesis by inhibition of inflammasome activation in mice. Hepatology 69(2):604–621. https://doi.org/10.1002/hep.30191
Liou GY, Storz P (2010) Reactive oxygen species in cancer. Free Radic Res 44(5):479–496. https://doi.org/10.3109/10715761003667554
Lopez J, Tait SW (2015) Mitochondrial apoptosis: killing cancer using the enemy within. Br J Cancer 112(6):957–962. https://doi.org/10.1038/bjc.2015.85
Maiuri MC, Le Toumelin G, Criollo A, Rain JC, Gautier F, Juin P, Tasdemir E, Pierron G, Troulinaki K, Tavernarakis N, Hickman JA, Geneste O, Kroemer G (2007) Functional and physical interaction between Bcl-X(L) and a BH3-like domain in Beclin-1. EMBO J 26(10):2527–2539. https://doi.org/10.1038/sj.emboj.7601689
Marques ARA, Di Spiezio A, Thiessen N, Schmidt L, Grotzinger J, Lullmann-Rauch R, Damme M, Storck SE, Pietrzik CU, Fogh J, Bar J, Mikhaylova M, Glatzel M, Bassal M, Bartsch U, Saftig P (2020) Enzyme replacement therapy with recombinant pro-CTSD (cathepsin D) corrects defective proteolysis and autophagy in neuronal ceroid lipofuscinosis. Autophagy 16(5):811–825. https://doi.org/10.1080/15548627.2019.1637200
Matkar SS, Wrischnik LA, Hellmann-Blumberg U (2008) Sanguinarine causes DNA damage and p53-independent cell death in human colon cancer cell lines. Chem Biol Interact 172(1):63–71. https://doi.org/10.1016/j.cbi.2007.12.006
Pan QZ, Pan K, Wang QJ, Weng DS, Zhao JJ, Zheng HX, Zhang XF, Jiang SS, Lv L, Tang Y, Li YQ, He J, Liu Q, Chen CL, Zhang HX, Xia JC (2015) Annexin A3 as a potential target for immunotherapy of liver cancer stem-like cells. Stem Cells 33(2):354–366. https://doi.org/10.1002/stem.1850
Park H, Bergeron E, Senta H, Guillemette K, Beauvais S, Blouin R, Sirois J, Faucheux N (2010) Sanguinarine induces apoptosis of human osteosarcoma cells through the extrinsic and intrinsic pathways. Biochem Biophys Res Commun 399(3):446–451. https://doi.org/10.1016/j.bbrc.2010.07.114
Rivera E, Gomez H (2010) Chemotherapy resistance in metastatic breast cancer: the evolving role of ixabepilone. Breast Cancer Res. https://doi.org/10.1186/bcr2573
Sanmamed MF, Chen L (2018) A paradigm shift in cancer immunotherapy: from enhancement to normalization. Cell 175(2):313–326. https://doi.org/10.1016/j.cell.2018.09.035
Su Q, Fan M, Wang J, Ullah A, Ghauri MA, Dai B, Zhan Y, Zhang D, Zhang Y (2019) Sanguinarine inhibits epithelial–mesenchymal transition via targeting HIF-1alpha/TGF-beta feed-forward loop in hepatocellular carcinoma. Cell Death Dis 10(12):939–953. https://doi.org/10.1038/s41419-019-2173-1
Su Q, Wang J, Liu F, Zhang Y (2020) Blocking Parkin/PINK1-mediated mitophagy sensitizes hepatocellular carcinoma cells to sanguinarine-induced mitochondrial apoptosis. Toxicol In Vitro 66:104840–104846. https://doi.org/10.1016/j.tiv.2020.104840
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249. https://doi.org/10.3322/caac.21660
Tong L, Chuang CC, Wu S, Zuo L (2015) Reactive oxygen species in redox cancer therapy. Cancer Lett 367(1):18–25. https://doi.org/10.1016/j.canlet.2015.07.008
Vrba J, Dolezel P, Vicar J, Ulrichova J (2009) Cytotoxic activity of sanguinarine and dihydrosanguinarine in human promyelocytic leukemia HL-60 cells. Toxicol In Vitro 23(4):580–588. https://doi.org/10.1016/j.tiv.2009.01.016
Weerasinghe P, Hallock S, Liepins A (2001) Bax, Bcl-2, and NF-kappaB expression in sanguinarine induced bimodal cell death. Exp Mol Pathol 71(1):89–98. https://doi.org/10.1006/exmp.2001.2355
Xu HD, Qin ZH (2019) Beclin 1, Bcl-2 and autophagy. Adv Exp Med Biol 1206:109–126. https://doi.org/10.1007/978-981-15-0602-4_5
Yuan X, Wang B, Yang L, Zhang Y (2018) The role of ROS-induced autophagy in hepatocellular carcinoma. Clin Res Hepatol Gastroenterol 42(4):306–312. https://doi.org/10.1016/j.clinre.2018.01.005
Zhang B, Wang X, Deng J, Zheng H, Liu W, Chen S, Tian J, Wang F (2019) p53-dependent upregulation of miR-16-2 by sanguinarine induces cell cycle arrest and apoptosis in hepatocellular carcinoma. Cancer Lett 459:50–58. https://doi.org/10.1016/j.canlet.2019.05.042
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant 81773772 and 81903643) and the Fundamental Research Funds for the Central Universities (xjj2018167, xtr0118022 and xzy012020086).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Su, Q., Wu, Q. et al. Sanguinarine impairs lysosomal function and induces ROS-dependent mitophagy and apoptosis in human hepatocellular carcinoma cells. Arch. Pharm. Res. 44, 1025–1036 (2021). https://doi.org/10.1007/s12272-021-01356-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-021-01356-0