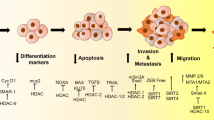
Abstract
Arrest defective 1 is an acetyltransferase that acetylates N-terminal amino acid or internal lysine residues of its target proteins. By acetylating its target proteins, ARD1 plays roles in many cellular activities, including proliferation, differentiation, autophagy, and apoptosis. In recent years, a number of investigations have emerged reporting the dysregulated expression of ARD1 in different types of cancer, including lung, liver, pancreas, breast, prostate, and colon cancer. Furthermore, the expression level of ARD1 in cancer tissues has been correlated with the progression and metastasis of the cancer and the survival of cancer patients. Consequently, mechanistic studies have revealed that ARD1-mediated protein acetylation plays an important role in modulating several cellular events that are important for cancer development, such as cell cycle progression, cell death, and migration. On the basis of this evidence, targeting of ARD1 has been proposed as a promising avenue for the development of novel cancer therapeutics. This review summarizes the biological functions of ARD1 in different types of cancer and provides a deep insight into the biochemical activities of ARD1 during tumor progression.


Similar content being viewed by others
References
Aksnes H, Ree R, Arnesen T (2019) Co-translational, post-translational, and non-catalytic roles of N-terminal acetyltransferases. Mol Cell 73:1097–1114. https://doi.org/10.1016/j.molcel.2019.02.007
Arnesen T, Gromyko D, Pendino F, Ryningen A, Varhaug JE, Lillehaug JR (2006) Induction of apoptosis in human cells by RNAi-mediated knockdown of hARD1 and NATH, components of the protein N-alpha-acetyltransferase complex. Oncogene 25:4350–4360. https://doi.org/10.1038/sj.onc.1209469
Arnesen T, Thompson PR, Varhaug JE, Lillehaug JR (2008) The protein acetyltransferase ARD1: a novel cancer drug target? Curr Cancer Drug Targets 8:545–553
Balogh J, Victor D 3rd, Asham EH, Burroughs SG, Boktour M, Saharia A, Li X, Ghobrial RM, Monsour HP Jr (2016) Hepatocellular carcinoma: a review. J Hepatocell Carcinoma 3:41–53. https://doi.org/10.2147/jhc.S61146
Banreti A, Sass M, Graba Y (2013) The emerging role of acetylation in the regulation of autophagy. Autophagy 9:819–829. https://doi.org/10.4161/auto.23908
Chien MH, Lee WJ, Yang YC, Tan P, Pan KF, Liu YC, Tsai HC, Hsu CH, Wen YC, Hsiao M, Hua KT (2018) N-alpha-acetyltransferase 10 protein promotes metastasis by stabilizing matrix metalloproteinase-2 protein in human osteosarcomas. Cancer Lett 433:86–98. https://doi.org/10.1016/j.canlet.2018.06.033
DePaolo JS, Wang Z, Guo J, Zhang G, Qian C, Zhang H, Zabaleta J, Liu W (2016) Acetylation of androgen receptor by ARD1 promotes dissociation from HSP90 complex and prostate tumorigenesis. Oncotarget 7:71417–71428. https://doi.org/10.18632/oncotarget.12163
Dörfel MJ, Lyon GJ (2015) The biological functions of Naa10—from amino-terminal acetylation to human disease. Gene 567:103–131. https://doi.org/10.1016/j.gene.2015.04.085
Drazic A, Myklebust LM, Ree R, Arnesen T (2016) The world of protein acetylation. Biochim Biophys Acta 1864:1372–1401. https://doi.org/10.1016/j.bbapap.2016.06.007
Fu M, Rao M, Wang C, Sakamaki T, Wang J, Di Vizio D, Zhang X, Albanese C, Balk S, Chang C, Fan S, Rosen E, Palvimo JJ, Janne OA, Muratoglu S, Avantaggiati ML, Pestell RG (2003) Acetylation of androgen receptor enhances coactivator binding and promotes prostate cancer cell growth. Mol Cell Biol 23:8563–8575. https://doi.org/10.1128/mcb.23.23.8563-8575.2003
Hua KT, Tan CT, Johansson G, Lee JM, Yang PW, Lu HY, Chen CK, Su JL, Chen PB, Wu YL, Chi CC, Kao HJ, Shih HJ, Chen MW, Chien MH, Chen PS, Lee WJ, Cheng TY, Rosenberger G, Chai CY, Yang CJ, Huang MS, Lai TC, Chou TY, Hsiao M, Kuo ML (2011) N-alpha-acetyltransferase 10 protein suppresses cancer cell metastasis by binding PIX proteins and inhibiting Cdc42/Rac1 activity. Cancer Cell 19:218–231. https://doi.org/10.1016/j.ccr.2010.11.010
Ji X, Tang C, Zhao Q, Wang W, Xiong Y (2014) Structural basis of cellular dNTP regulation by SAMHD1. Proc Natl Acad Sci 111:E4305–E4314. https://doi.org/10.1073/pnas.1412289111
Jiang B, Ren T, Dong B, Qu L, Jin G, Li J, Qu H, Meng L, Liu C, Wu J, Shou C (2010) Peptide mimic isolated by autoantibody reveals human arrest defective 1 overexpression is associated with poor prognosis for colon cancer patients. Am J Pathol 177:1095–1103. https://doi.org/10.2353/ajpath.2010.091178
Kalvik TV, Arnesen T (2013) Protein N-terminal acetyltransferases in cancer. Oncogene 32:269–276. https://doi.org/10.1038/onc.2012.82
Kuhns KJ, Zhang G, Wang Z, Liu W (2018) ARD1/NAA10 acetylation in prostate cancer. Exp Mol Med 50:89. https://doi.org/10.1038/s12276-018-0107-0
Kuo HP, Hung MC (2010) Arrest-defective-1 protein (ARD1): tumor suppressor or oncoprotein? Am J Transl Res 2:56–64
Kuo HP, Lee DF, Xia W, Lai CC, Li LY, Hung MC (2009) Phosphorylation of ARD1 by IKKbeta contributes to its destabilization and degradation. Biochem Biophys Res Commun 389:156–161. https://doi.org/10.1016/j.bbrc.2009.08.127
Kuo HP, Lee DF, Chen CT, Liu M, Chou CK, Lee HJ, Du Y, Xie X, Wei Y, Xia W, Weihua Z, Yang JY, Yen CJ, Huang TH, Tan M, Xing G, Zhao Y, Lin CH, Tsai SF, Fidler IJ, Hung MC (2010) ARD1 stabilization of TSC2 suppresses tumorigenesis through the mTOR signaling pathway. Sci Signal 3:ra9. https://doi.org/10.1126/scisignal.2000590
Lavery DN, Bevan CL (2011) Androgen receptor signalling in prostate cancer: the functional consequences of acetylation. J Biomed Biotechnol 2011:862125. https://doi.org/10.1155/2011/862125
Lee CF, Ou DS, Lee SB, Chang LH, Lin RK, Li YS, Upadhyay AK, Cheng X, Wang YC, Hsu HS, Hsiao M, Wu CW, Juan LJ (2010) hNaa10p contributes to tumorigenesis by facilitating DNMT1-mediated tumor suppressor gene silencing. J Clin Invest 120:2920–2930. https://doi.org/10.1172/JCI42275
Lee EJ, Seo JH, Park JH, Vo TTL, An S, Bae SJ, Le H, Lee HS, Wee HJ, Lee D, Chung YH, Kim JA, Jang MK, Ryu SH, Yu E, Jang SH, Park ZY, Kim KW (2017) SAMHD1 acetylation enhances its deoxynucleotide triphosphohydrolase activity and promotes cancer cell proliferation. Oncotarget 8:68517–68529. https://doi.org/10.18632/oncotarget.19704
Lee D, Jang MK, Seo JH, Ryu SH, Kim JA, Chung YH (2018a) ARD1/NAA10 in hepatocellular carcinoma: pathways and clinical implications. Exp Mol Med 50:88. https://doi.org/10.1038/s12276-018-0106-1
Lee EJ, Seo JH, Kim KW (2018b) Special issue on protein acetylation: from molecular modification to human disease. Exp Mol Med 50:84. https://doi.org/10.1038/s12276-018-0103-4
Lee MN, Kweon HY, Oh GT (2018c) N-α-acetyltransferase 10 (NAA10) in development: the role of NAA10. Exp Mol Med 50:87. https://doi.org/10.1038/s12276-018-0105-2
Lim JH, Park JW, Chun YS (2006) Human arrest defective 1 acetylates and activates beta-catenin, promoting lung cancer cell proliferation. Cancer Res 66:10677–10682. https://doi.org/10.1158/0008-5472.CAN-06-3171
Lonergan PE, Tindall DJ (2011) Androgen receptor signaling in prostate cancer development and progression. J Carcinog 10:20. https://doi.org/10.4103/1477-3163.83937
Lozada EM, Andrysik Z, Yin M, Redilla N, Rice K, Stambrook PJ (2016) Acetylation and deacetylation of Cdc25A constitutes a novel mechanism for modulating Cdc25A functions with implications for cancer. Oncotarget 7:20425–20439. https://doi.org/10.18632/oncotarget.7966
Luczak MW, Jagodzinski PP (2006) The role of DNA methylation in cancer development. Folia Histochem Cytobiol 44:143–154
Markopoulos AK (2012) Current aspects on oral squamous cell carcinoma. Open Dent J 6:126–130. https://doi.org/10.2174/1874210601206010126
Marley AR, Nan H (2016) Epidemiology of colorectal cancer. Int J Mol Epidemiol Genet 7:105–114
Merriel SWD, Funston G, Hamilton W (2018) Prostate Cancer Prim Care. Adv Ther 35:1285–1294. https://doi.org/10.1007/s12325-018-0766-1
Moskovitz J, Bar-Noy S, Williams WM, Requena J, Berlett BS, Stadtman ER (2001) Methionine sulfoxide reductase (MsrA) is a regulator of antioxidant defense and lifespan in mammals. Proc Natl Acad Sci USA 98:12920–12925. https://doi.org/10.1073/pnas.231472998
Nguyen KT, Mun SH, Lee CS, Hwang CS (2018) Control of protein degradation by N-terminal acetylation and the N-end rule pathway. Exp Mol Med 50:91. https://doi.org/10.1038/s12276-018-0097-y
Park EC, Szostak JW (1992) ARD1 and NAT1 proteins form a complex that has N-terminal acetyltransferase activity. EMBO J 11:2087–2093. https://doi.org/10.1002/j.1460-2075.1992.tb05267.x
Park YH, Seo JH, Park JH, Lee HS, Kim KW (2017) Hsp70 acetylation prevents caspase-dependent/independent apoptosis and autophagic cell death in cancer cells. Int J Oncol 51:573–578. https://doi.org/10.3892/ijo.2017.4039
Qian X, Li X, Lu Z (2017) Protein kinase activity of the glycolytic enzyme PGK1 regulates autophagy to promote tumorigenesis. Autophagy 13:1246–1247. https://doi.org/10.1080/15548627.2017.1313945
Ree R, Varland S, Arnesen T (2018) Spotlight on protein N-terminal acetylation. Exp Mol Med 50:90. https://doi.org/10.1038/s12276-018-0116-z
Ren T, Jiang B, Jin G, Li J, Dong B, Zhang J, Meng L, Wu J, Shou C (2008) Generation of novel monoclonal antibodies and their application for detecting ARD1 expression in colorectal cancer. Cancer Lett 264:83–92. https://doi.org/10.1016/j.canlet.2008.01.028
Seo JH, Cha JH, Park JH, Jeong CH, Park ZY, Lee HS, Oh SH, Kang JH, Suh SW, Kim KH, Ha JY, Han SH, Kim SH, Lee JW, Park JA, Jeong JW, Lee KJ, Oh GT, Lee MN, Kwon SW, Lee SK, Chun KH, Lee SJ, Kim KW (2010) Arrest defective 1 autoacetylation is a critical step in its ability to stimulate cancer cell proliferation. Cancer Res 70:4422–4432. https://doi.org/10.1158/0008-5472.CAN-09-3258
Seo JH, Park JH, Lee EJ, Vo TT, Choi H, Kim JY, Jang JK, Wee HJ, Lee HS, Jang SH, Park ZY, Jeong J, Lee KJ, Seok SH, Park JY, Lee BJ, Lee MN, Oh GT, Kim KW (2016) ARD1-mediated Hsp70 acetylation balances stress-induced protein refolding and degradation. Nat Commun 7:12882. https://doi.org/10.1038/ncomms12882
Shang Y, Myers M, Brown M (2002) Formation of the androgen receptor transcription complex. Mol Cell 9:601–610
Shim JH, Chung YH, Kim JA, Lee D, Kim KM, Lim YS, Lee HC, Lee YS, Yu E, Lee YJ (2012) Clinical implications of arrest-defective protein 1 expression in hepatocellular carcinoma: a novel predictor of microvascular invasion. Dig Dis 30:603–608. https://doi.org/10.1159/000343090
Shin DH, Chun YS, Lee KH, Shin HW, Park JW (2009) Arrest defective-1 controls tumor cell behavior by acetylating myosin light chain kinase. PLoS ONE 4:e7451. https://doi.org/10.1371/journal.pone.0007451
Shin SH, Yoon H, Chun YS, Shin HW, Lee MN, Oh GT, Park JW (2014) Arrest defective 1 regulates the oxidative stress response in human cells and mice by acetylating methionine sulfoxide reductase A. Cell Death Dis 5:e1490. https://doi.org/10.1038/cddis.2014.456
Shtutman M, Zhurinsky J, Simcha I, Albanese C, D’Amico M, Pestell R, Ben-Ze’ev A (1999) The cyclin D1 gene is a target of the beta-catenin/LEF-1 pathway. Proc Natl Acad Sci USA 96:5522–5527. https://doi.org/10.1073/pnas.96.10.5522
Soppa J (2010) Protein acetylation in archaea, bacteria, and eukaryotes. Archaea. https://doi.org/10.1155/2010/820681
Stove SI, Blenski M, Stray-Pedersen A, Wierenga KJ, Jhangiani SN, Akdemir ZC, Crawford D, McTiernan N, Myklebust LM, Purcarin G, McNall-Knapp R, Wadley A, Belmont JW, Kim JJ, Lupski JR, Arnesen T (2018) A novel NAA10 variant with impaired acetyltransferase activity causes developmental delay, intellectual disability, and hypertrophic cardiomyopathy. Eur J Hum Genet 26:1294–1305. https://doi.org/10.1038/s41431-018-0136-0
Vo TTL, Park JH, Seo JH, Lee EJ, Choi H, Bae SJ, Le H, An S, Lee HS, Wee HJ, Kim KW (2017) ARD1-mediated aurora kinase A acetylation promotes cell proliferation and migration. Oncotarget 8:57216–57230. https://doi.org/10.18632/oncotarget.19332
Vo TTL, Jeong CH, Lee S, Kim KW, Ha E, Seo JH (2018) Versatility of ARD1/NAA10-mediated protein lysine acetylation. Exp Mol Med 50:86. https://doi.org/10.1038/s12276-018-0100-7
Wang ZH, Gong JL, Yu M, Yang H, Lai JH, Ma MX, Wu H, Li L, Tan DY (2011) Up-regulation of human arrest-defective 1 protein is correlated with metastatic phenotype and poor prognosis in breast cancer. Asian Pac J Cancer Prev 12:1973–1977
Wang Z, Wang Z, Guo J, Li Y, Bavarva JH, Qian C, Brahimi-Horn MC, Tan D, Liu W (2012) Inactivation of androgen-induced regulator ARD1 inhibits androgen receptor acetylation and prostate tumorigenesis. Proc Natl Acad Sci USA 109:3053–3058. https://doi.org/10.1073/pnas.1113356109
Wu Y, Lyon GJ (2018) NAA10-related syndrome. Exp Mol Med 50:85. https://doi.org/10.1038/s12276-018-0098-x
Xu H, Jiang B, Meng L, Ren T, Zeng Y, Wu J, Qu L, Shou C (2012) N-alpha-acetyltransferase 10 protein inhibits apoptosis through RelA/p65-regulated MCL1 expression. Carcinogenesis 33:1193–1202. https://doi.org/10.1093/carcin/bgs144
Xu W, Li Y, Liu C, Zhao S (2014) Protein lysine acetylation guards metabolic homeostasis to fight against cancer. Oncogene 33:2279–2285. https://doi.org/10.1038/onc.2013.163
Yamaguchi H, Wyckoff J, Condeelis J (2005) Cell migration in tumors. Curr Opin Cell Biol 17:559–564. https://doi.org/10.1016/j.ceb.2005.08.002
Yang H, Li Q, Niu J, Li B, Jiang D, Wan Z, Yang Q, Jiang F, Wei P, Bai S (2016) microRNA-342-5p and miR-608 inhibit colon cancer tumorigenesis by targeting NAA10. Oncotarget 7:2709–2720. https://doi.org/10.18632/oncotarget.6458
Yoon H, Kim HL, Chun YS, Shin DH, Lee KH, Shin CS, Lee DY, Kim HH, Lee ZH, Ryoo HM, Lee MN, Oh GT, Park JW (2014) NAA10 controls osteoblast differentiation and bone formation as a feedback regulator of Runx2. Nat Commun 5:5176. https://doi.org/10.1038/ncomms6176
Yu M, Ma M, Huang C, Yang H, Lai J, Yan S, Li L, Xiang M, Tan D (2009) Correlation of expression of human arrest-defective-1 (hARD1) protein with breast cancer. Cancer Invest 27:978–983. https://doi.org/10.3109/07357900902769723
Yu L, Chen Y, Tooze SA (2018) Autophagy pathway: cellular and molecular mechanisms. Autophagy 14:207–215. https://doi.org/10.1080/15548627.2017.1378838
Zeng Y, Min L, Han Y, Meng L, Liu C, Xie Y, Dong B, Wang L, Jiang B, Xu H, Zhuang Q, Zhao C, Qu L, Shou C (2014) Inhibition of STAT5a by Naa10p contributes to decreased breast cancer metastasis. Carcinogenesis 35:2244–2253. https://doi.org/10.1093/carcin/bgu132
Zeng Y, Zheng J, Zhao J, Jia PR, Yang Y, Yang GJ, Ma JF, Gu YQ, Xu J (2016) High expression of Naa10p associates with lymph node metastasis and predicts favorable prognosis of oral squamous cell carcinoma. Tumour Biol 37:6719–6728. https://doi.org/10.1007/s13277-015-4563-z
Zhang Y, Zhou H, Tao Y, Liu X, Yuan Z, Nie C (2018) ARD1 contributes to IKKbeta-mediated breast cancer tumorigenesis. Cell Death Dis 9:860. https://doi.org/10.1038/s41419-018-0921-2
Zhou Y, Bolton EC, Jones JO (2015) Androgens and androgen receptor signaling in prostate tumorigenesis. J Mol Endocrinol 54:R15–R29. https://doi.org/10.1530/jme-14-0203
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2016R1A6A1A03011325, NRF-2016R1D1A1B03935560) and the Korean Government (MSIT) (NRF-2019R1C1C1005855).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chaudhary, P., Ha, E., Vo, T.T.L. et al. Diverse roles of arrest defective 1 in cancer development. Arch. Pharm. Res. 42, 1040–1051 (2019). https://doi.org/10.1007/s12272-019-01195-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-019-01195-0