Abstract
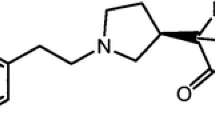
Capillary zone electrophoresis was successfully applied to the enantiomeric purity determination of dexlansoprazole using sulfobutyl ether-β-cyclodextrin and methyl-β-cyclodextrin as chiral selectors. Separations were carried out in a 50 μm, 64/56 cm fused-silica capillary. The optimized conditions included 90 mM phosphate buffer, pH 6.0, containing 30 mM sulfobutyl ether-β-cyclodextrin, 20 mM methyl-β-cyclodextrin as background electrolyte, an applied voltage of 25 kV and a temperature of 16 °C, detection was at 280 nm. The assay was validated for the S-(−)-lansoprazole in the range of 0.2–1.0%. The limit of detection was 0.07%, the limit of quantitation was 0.20%, relative to a total concentration of 4.0 mg mL−1. Intra-day precision varied between 1.72 and 2.07%. Relative standard deviations of inter-day precision ranged between 1.62 and 1.96% for peak area ratio. The assay was applied for the determination of the chiral purity of dexlansoprazole capsules. Recovery in capsules was ranged between 101.7 and 103.1%.
Graphical Abstract





Similar content being viewed by others
References
Barradell LB, Faulds D, McTavish D (1992) Lansoprazole: a review of its pharmacodynamic and pharmacokinetic properties and its therapeutic efficacy in acid-related disorders. Drugs 44(2):225–250
Borner K, Borner E, Lode H (1998) Separation of lansoprazole enantiomers in human serum by HPLC. Chromatographia 47:171–175
Chankvetadze B (1997) Capillary electrophoresis in chiral analysis. Wiley, Chinchester
Cirilli R, Ferretti R, Gallinella B, Turchetto L, Zanitti L, La Torre F (2009) Development and validation of an enantioselective and chemoselective HPLC method using a Chiralpak IA column to simultaneously quantify (R)-(+)- and (S)-(−)-lansoprazole enantiomers and related impurities. J Pharm Biomed Anal 50:9–14
Fillet M, Hubert P, Crommen J (1998) Method development strategies for the enantioseparation of drugs by capillary electrophoresis using cyclodextrines as chiral additives. Electrophoresis 19:2834–2840
Guttman A, Cooke Nelson (1994) Practical aspects of chiral separations of pharmaceuticals by capillary electrophoresis I: separation optimization. J Chromatogr A 680:157–162
International Conference on Harmonisation (ICH) (2005) Guideline Q2(R1), validation of analytical procedure: methodology. http://www.ich.org/products/guidelines/quality/article/quality-guidelines.html
Katsuki H, Hamada A, Nakamura C, Arimori K, Nakano M (2001) High-performance liquid chromatographic assay for the simultaneous determination of lansoprazole enantiomers and metabolites in human liver microsomes. J Chromatogr B 757:127–133
Lee KR, Nguyen NVT, Lee YJ, Choi S, Kang JS, Kim KH (2015) Determination of the R-enantiomer of valsartan in pharmaceutical formulation by capillary electrophoresis. Arch Pharm Res 38:826–833
Miura M, Tada H, Suzuki T (2004) Simultaneous determination of lansoprazole enantiomers and their metabolites in plasma by liquid chromatography with solid-phase extraction. J Chromatogr B 804:389–395
Nevado JJ, Peñalvo GC, Sánchez JC, Mochón MC, Dorado RM, Navarro MV (2009) Optimisation and validation of a new CE method for the determination of lansoprazole enantiomers in pharmaceuticals. Electrophoresis 30:2940–2946
Penn SG, Goodall DM, Loran JS (1993) Differential binding of tioconazole enantiomers to hydroxypropyl-beta-cyclodextrin studied by capillary electrophoresis. J Chromatogr 636:149–152
Shibukawa A, Lloyd DK, Wainer IW (1993) Simultaneous chiral separation of leucovorin and its major metabolite 5-methyl-tetrahydrofolate by capillary electrophoresis using cyclodextrins as chiral selectors: estimation of the formation constant and mobility of the solute-cyclodextrin complexes. Chromatographia 35:419–429
Stavrou IJ, Mavroudi MC, Kapnissi-Christodoulou CP (2015) Chiral selectors in CE: recent developments and applications (2012-mid 2014). Electrophoresis 36:101–123
Sun L, Cao Y, Jiao H, Fang Y, Yang Z, Bian M, Zhang H, Gong X, Wang Y (2015) Enantioselective determination of (R)- and (S)-lansoprazole in human plasma by chiral liquid chromatography with mass spectrometry and its application to a stereoselective pharmacokinetic study. J Sep Sci 38:3696–3703
Wang H, Sun Y, Meng X, Yang B, Wang J, Yang Y, Gu J (2015) Determination of lansoprazole enantiomers in dog plasma by column-switching liquid chromatography with tandem mass spectrometry and its application to a preclinical pharmacokinetic study. J Sep Sci 38:2960–2967
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The authors thank the Institute of New Drug Development Research and the Central Laboratory of Kangwon National University for the use of analytical instrument.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared no conflict of interest.
Additional information
Hyun Kyu Chung and Quoc-Ky Truong have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chung, H.K., Truong, QK., Mai, XL. et al. Determination of S-(−)-lansoprazole in dexlansoprazole preparation by capillary zone electrophoresis. Arch. Pharm. Res. 40, 962–971 (2017). https://doi.org/10.1007/s12272-017-0936-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-017-0936-8