Abstract
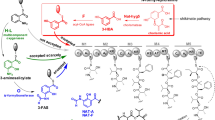
A novel cyclic dipeptide, named penicimutide (1), and four known cyclic dipeptides, cyclo(l-Val-l-Pro) (2), cyclo(l-Ile-l-Pro) (3), cyclo(l-Leu-l-Pro) (4) and cyclo(l-Phe-l-Pro) (5), were isolated from a neomycin-resistant mutant of the marine-derived fungus Penicillium purpurogenum G59. The structure of 1, including the absolute configuration, was determined by spectroscopic and chemical methods, especially NMR and Marfey’s analysis. An unusual amino acid in 1, 4,5-didehydro-l-leucine, was found for the first time occurring in nature. HPLC–ESI–MS analysis evidenced that 1–3 were produced only in the mutant strain, but 4 and 5 were produced in both the mutant and parental strains, indicating that the introduction of neomycin-resistance in the mutant activated pathways of 1–3 biosynthesis that were silent in the parental strain. Compound 1 selectively inhibited HeLa cells (among five tested human cancer cell lines) with an inhibition rate (IR %) of 39.4 % at 100 µg/mL, a similar inhibition intensity to that of the positive control 5-fluorouracil (IR % of 41.4 % at 100 µg/mL against HeLa cells). The present work exemplifies the effectiveness of our previous DMSO-mediated method for introducing drug-resistance in fungi to activate silent biosynthetic pathways to obtain new bioactive compounds.




Similar content being viewed by others
References
Aravind L, de Souza RF, Iyer LM (2010) Predicted class-I aminoacyl tRNA synthetase-like proteins in non-ribosomal peptide synthesis. Biol Direct 5:48
Arunrattiyakorn P, Nitoda T, Kanzaki H (2006) Enzymatic conversion-based method for screening cyclic dipeptide-producing microbes. Peptides 27:633–639
Bellezza I, Peirce MJ, Minelli A (2014) Cyclic dipeptides: from bugs to brain. Trends Mol Med 20:551–558
Bhushan R, Brückner H (2004) Marfey’s reagent for chiral amino acid analysis: a review. Amino Acids 27:231–247
Bode HB, Bethe B, Höfs R, Zeeck A (2002) Big effects from small changes: possible ways to explore nature’s chemical diversity. ChemBioChem 3:619–627
Chai YJ, Cui CB, Li CW, Hua W (2011) Antitumor metabolites newly produced by a gentamicin-resistant mutant of Penicillium purpurogenum G59. J Int Pharm Res 38:216–222
Chai YJ, Cui CB, Li CW, Wu CJ, Tian CK, Hua W (2012) Activation of the dormant secondary metabolite production by introducing gentamicin-resistance in a marine-derived Penicillium purpurogenum G59. Mar Drugs 10:559–582
Cichewicz RH, Henrikson JC, Wang X, Branscum KM (2010) Strategies for accessing microbial secondary metabolites from silent biosynthetic pathways. In: Baltz RH, Davies JE, Demain AL, Bull AT, Junker B, Katz L, Lynd LR, Masurekar PC, Reeves D, Zhao H (eds) Manual of industrial microbiology and biotechnology. ASM Press, Washington, pp 78–95
Cui CB (2010) A new approach for exploiting microbial new strain resources for drug screening. J Int Pharm Res 37:1–7
Dong Y, Cui CB, Li CW, Hua W, Wu CJ, Zhu TJ, Gu QQ (2014) Activation of dormant secondary metabolite production by introducing neomycin resistance into the deep-sea fungus, Aspergillus versicolor ZBY-3. Mar Drugs 12:4326–4352
Edagwa BJ, Taylor CM (2009) Peptides containing γ, δ-dihydroxy-l-leucine. J Org Chem 74:4132–4136
Fang SM, Cui CB, Li CW, Wu CJ, Zhang ZJ, Li L, Huang XJ, Ye WC (2012) Purpurogemutantin and purpurogemutantidin, new drimenyl cyclohexenone derivatives produced by a mutant obtained by diethyl sulfate mutagenesis of a marine-derived Penicillium purpurogenum G59. Mar Drugs 10:1266–1287
Fang SM, Wu CJ, Li CW, Cui CB (2014) A practical strategy to discover new antitumor compounds by activating silent metabolite production in fungi by diethyl sulphate mutagenesis. Mar Drugs 12:1788–1814
Fdhila F, Vázquez V, Sánchez JL, Riguera R (2003) dd-Diketopiperazines: antibiotics active against Vibrio anguillarum isolated from marine bacteria associated with cultures of Pecten maximus. J Nat Prod 66:1299–1301
Fu P, Jamison M, La S, MacMillan JB (2014) Inducamides A–C, chlorinated alkaloids from an RNA polymerase mutant strain of Streptomyces sp. Org Lett 16:5656–5659
Furukawa T, Akutagawa T, Funatani H, Uchida T, Hotta Y, Niwa M, Takaya Y (2012) Cyclic dipeptides exhibit potency for scavenging radicals. Bioorg Med Chem 20:2002–2009
Gondry M, Lautru S, Fusai G, Munier G, Ménez A, Genet R (2001) Cyclic dipeptide oxidase from Streptomyces noursei isolation, purification and partial characterization of a novel, amino acyl α, β-dehydrogenase. Eur J Biochem 268:1712–1721
Gondry M, Seuguet L, Belin P, Thai R, Amouroux R, Tellier C, Tuphile K, Jacquet M, Braud S, Courcon M, Masson C, Dubois S, Lautru S, Lecoq A, Hashimoto SI, Genet R, Pernodet JL (2009) Cyclodipeptide synthases are a family of tRNA-dependent peptide bond-forming enzymes. Nat Chem Biol 5:414–420
Henrikson JC, Hoover AR, Joyner PM, Cichewicz RH (2009) A chemical epigenetics approach for engineering the in situ biosynthesis of a cryptic natural product from Aspergillus niger. Org Biomol Chem 7:435–438
Hosaka T, Ohnishi-Kameyama M, Muramatsu H, Murakami K, Tsurumi Y, Kodani S, Yoshida M, Fujie A, Ochi K (2009) Antibacterial discovery in actinomycetes strains with mutations in RNA polymerase or ribosomal protein S12. Nat Biotechnol 27:462–464
Huang R, Zhou X, Xu T, Yang X, Liu Y (2010) Diketopiperazines from marine organisms. Chem Biodivers 7:2809–2829
Jayatilake GS, Thornton MP, Leonard AC, Grimwade JE, Baker BJ (1996) Metabolites from an Antarctic sponge-associated bacterium, Pseudomonas aeruginosa. J Nat Prod 59:293–296
Jia JM, Ma XC, Wu CF, Wu LJ, Hu GS (2005) Cordycedipeptide A, a new cyclodipeptide from the culture liquid of Cordyceps sinensis (Berk.) Sacc. Chem Pharm Bull 53:582–583
Kanzaki H, Imura D, Nitoda T, Kawazu K (2000) Effective production of dehydro cyclic dipeptide albonoursin exhibiting pronuclear fusion inhibitory activity II. Biosynthetic and bioconversion studies. J Antibiot 53:58–62
Lahoud G, Hou YM (2010) Biosynthesis: a new (old) way of hijacking tRNA. Nat Chem Biol 6:795–796
Marmann A, Aly AH, Lin W, Wang B, Proksch P (2014) Co-cultivation—a powerful emerging tool for enhancing the chemical diversity of microorganisms. Mar Drugs 12:1043–1065
Martins MB, Carvalho I (2007) Diketopiperazines: biological activity and synthesis. Tetrahedron 63:9923–9932
Ochi K (2007) From microbial differentiation to ribosome engineering. Biosci Biothenol Biochem 71:1373–1386
Ochi K, Hosaka T (2013) New strategies for drug discovery: activation of silent or weakly expressed microbial gene clusters. Appl Microbiol Biotechnol 97:87–98
Ochi K, Okamoto S, Tozawa Y, Inaoka T, Hosaka T, Xu J, Kurosawa K (2004) Ribosome engineering and secondary metabolite production. Adv Appl Microbiol 56:155–184
Pedras MSC, Yu Y, Liu J, Tandron-Moya YA (2005) Metabolites produced by the phytopathogenic fungus Rhizoctonia solani: isolation, chemical structure determination, syntheses and bioactivity. Z Naturforsch C 60:717–722
Takaya Y, Furukawa T, Miura S, Akutagawa T, Hotta Y, Ishikawa N, Niwa M (2007) Antioxidant constituents in distillation residue of Awamori spirits. J Agric Food Chem 55:75–79
Teixidó M, Zurita E, Malakoutikhah M, Tarrago T, Giralt E (2007) Diketopiperazines as a tool for the study of transport across the blood–brain barrier (BBB) and their potential use as BBB-shuttles. J Am Chem Soc 129:11802–11813
Tian CK, Cui CB, Han XX (2008) Isolation of fungal strains in unusual environment and screening for their antitumor activity. J Int Pharm Res 35:401–405
Wu CJ, Cui CB, Tian CK, Li CW (2010) Antitumor metabolites produced by two Penicillium purpurogenum G59 mutants. J Int Pharm Res 37:122–126
Wu CJ, Li CW, Cui CB (2014) Seven new and two known lipopeptides as well as five known polyketides: the activated production of silent metabolites in a marine-derived fungus by chemical mutagenesis strategy using diethyl sulphate. Mar Drugs 12:1815–1838
Wu CJ, Yi L, Cui CB, Li CW, Wang N, Han X (2015) Activation of the silent secondary metabolite production by introducing neomycin-resistance in a marine-derived Penicillium purpurogenum G59. Mar Drugs 13:2465–2487
Xia MW, Cui CB, Li CW, Wu CJ (2014) Three new and eleven known unusual C25 steroids: activated production of silent metabolites in a marine-derived fungus by chemical mutagenesis strategy using diethyl sulphate. Mar Drugs 12:1545–1568
Xia MW, Cui CB, Li CW, Wu CJ, Peng JX, Li DH (2015) Rare chromones from a fungal mutant of the marine-derived Penicillium purpurogenum G59. Mar Drugs 13:5219–5236
Acknowledgments
This work was supported by the Grants from the NSFC (81573300, 30973631), NHTRDP (2013AA092901, 2007AA09Z411), NSTMP (2009ZX09301-002, 2012ZX09301-003) and AMMS (2008), China, and the NSFC-Shandong Joint Fund for Marine Science Research Centers (U1406402), China. Penicillium purpurogenum G59 was identified by Prof. Liang-Dong Guo, the Institute of Microbiology, Chinese Academy of Sciences, China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, N., Cui, CB. & Li, CW. A new cyclic dipeptide penicimutide: the activated production of cyclic dipeptides by introduction of neomycin-resistance in the marine-derived fungus Penicillium purpurogenum G59. Arch. Pharm. Res. 39, 762–770 (2016). https://doi.org/10.1007/s12272-016-0751-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-016-0751-7