Abstract
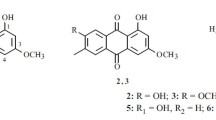
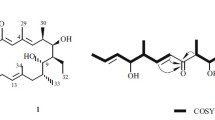
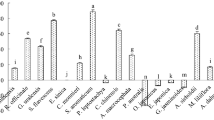
A new carbazole alkaloid named clauraila E (1) together with 8 known compounds were isolated from the methanol extract of the roots of Clausena harmandiana. All compounds were evaluated for antifungal activity against Pythium insidiosum using disc diffusion assay. Pythium insidiosum is a fungus-like microorganism, for which antifungals available now are not effective. It was found that compounds 3, 6, 7 and 9 could inhibit the mycelia growth of P. insidiosum. The results show convincingly that they may be lead to compounds for the development of probiotic or novel antifungal drugs.



Similar content being viewed by others
References
Alexopoulos, C.J., C.W. Mims, and M. Blackwell. 1996. Introductory Mycology, 4th (edn). John Wiley New York, pp. 61–85, 683–737.
Chakraborty, A., C. Saha, G. Podder, B.K. Chowdhury, and P. Bhattacharyya. 1995. Carbazole alkaloid with antimicrobial activity from Clausena heptaphylla. Phytochemistry 38: 787–789.
De Cock, A.W., L. Mendoza, A.A. Padhye, L. Ajello, and L. Kaufman. 1987. Pythium insidiosum sp. nov., the etiologic agent of pythiosis. Journal of Clinical Microbiology 25: 344–349.
He, H.P., J.X. Zhang, Y.M. Shen, Y.N. He, C. Chen, and X.J. Hao. 2002. Tetranortriterpenoids from Clausena excavata. Helvetica Chimica Acta 85: 671–677.
Ito, C., S. Katsuno, H. Ohta, M. Omura, I. Kajura, and H. Furukawa. 1997. Constituents of Clausena excavata. Isolation and structural elucidation of new carbazole alkaloids. Chemical & Pharmaceutical Bulletin 45: 48–52.
Ito, C., M. Itoigawa, S. Katsuno, M. Omura, H. Tokuda, H. Nishino, and H. Furukawa. 2000. Chemical constituents of Clausena excavata: Isolation and structure elucidation of novel furanone-coumarins with inhibitory effects for tumor-promotion. Journal of Natural Products 63: 1218–1224.
Ito, C., M. Itoigawa, K. Aizawa, K. Yoshida, N. Ruangrungsi, and H. Furukawa. 2009. γ-Lactone carbazoles from Clausena anisata. Journal of Natural Products 72: 1202–1204.
Kataeva, O., M.P. Krahl, and H.J. Knoelker. 2005. First total synthesis of the biologically active 2,7-dioxygenated tricyclic carbazole alkaloids 7-methoxy-O-methylmukonal, clausine H (clauszoline-C), clausine K (clauszoline-J) and clausine O. Organic & Biomolecular Chemistry 3: 3099–3101.
Kongkathip, B., N. Kongkathip, A. Sunthitikawinsakul, C. Napaswat, and C. Yoosook. 2005. Anti-HIV-1 constituents from Clausena excavata: Part II. Carbazoles and a pyrano-coumarin. Phytotherapy Research 19: 728–731.
Krajaejun, T., B. Sathapatayavongs, R. Pracharktam, P. Nitiyanant, P. Leelachaikul, and W. Wanachiwanawin. 2006. Clinical and epidemiological analyses of human pythiosis in Thailand. Clinical Infectious Diseases 43: 569–576.
Luo, X.M., S.H. Qi, H. Yin, C.H. Gao, and S. Zhang. 2009. Alkaloids from the stem bark of Micromelum falcatum. Chemical & Pharmaceutical Bulletin 57: 600–602.
Maneerat, W., T. Ritthiwigrom, S. Cheenpracha, T. Promgool, K. Yossathera, S. Deachathai, W. Phakhodee, and S. Laphookhieo. 2012. Bioactive carbazole alkaloids from Clausena wallichii Roots. Journal of Natural Products 75: 741–746.
Mendoza, L. 2005. Phytium insidiosum. In Topley and Wilson’s Microbiology and Microbial Infections. Medical Mycology, 10th ed, ed. W.G. Merz, and R.J. Hay, 617–630. London: Hodder Arnold.
Nakamura, T., N. Kodama, Y. Arai, T. Kumamoto, Y. Higuchi, C. Chaichantipyuth, T. Ishikawa, K. Ueno, and S. Yano. 2009. Inhibitory effect of oxycoumarins isolated from the Thai medicinal plant Clausena guillauminii on the inflammation mediators, iNOS, TNF-α, and COX-2 expression in mouse macrophage RAW 264.7. Journal of Natural Medicines 63: 21–27.
Noipha, K., T. Thongthoom, U. Songsiang, C. Boonyarat, and C. Yenjai. 2010. Carbazoles and coumarins from Clausena harmandiana stimulate glucose uptake in L6 myotubes. Diabetes Research and Clinical Practice 90: e67–e71.
Shi, X.J., G. Ye, W.J. Tang, and W.M. Zhao. 2010. A New coumarin and carbazole alkaloid from Clausena vestita D.D. Tao. Helvetica Chimica Acta 93: 985–990.
Songsiang, U., T. Thongthoom, C. Boonyarat, and C. Yenjai. 2011. Claurailas A–D, Cytotoxic carbazole alkaloids from the roots of Clausena harmandiana. Journal of Natural Products 74: 208–212.
Songsiang, U., T. Thongthoom, P. Zeekpudsa, V. Kukongviriyapan, C. Boonyarat, J. Wangboonskul, and C. Yenjai. 2012. Antioxidant activity and cytotoxicity against cholangiocarcinoma of carbazoles and coumarins from Clausena harmandiana. ScienceAsia 38: 75–81.
Thongthoom, T., U. Songsiang, C. Phaosiri, and C. Yenjai. 2010. Biological activity of the chemical constituents from Clausena harmandiana. Archives of Pharmacal Research 33: 675–680.
Thongthoom, T., P. Promsuwan, and C. Yenjai. 2011. Synthesis and cytotoxic activity of the heptaphylline and 7-methoxyheptaphylline series. European Journal of Medicinal Chemistry 46: 3755–3761.
Tohyama, S., T. Choshi, S. Azuma, H. Fujioka, and S. Hibino. 2009. A new synthetic route to the 1-oxygenated carbazole alkaloids, mukonine and clausine E (clauzoline I). Heterocycles 79: 955–965.
Wu, T.S., S.C. Huang, J.S. Lai, C.M. Teng, F.N. Ko, and C.S. Kuoh. 1993. Chemical and antiplatelet aggregative investigation of the leaves of Clausena excavata. Phytochemistry 32: 449–451.
Wu, T.S., S.C. Huang, P.L. Wu, and C.S. Kuoh. 1999. Alkaloidal and other constituents from the root bark of Clausena excavata. Phytochemistry 52: 523–527.
Xin, Z.Q., J.J. Lu, C.Q. Ke, C.X. Hu, L.P. Lin, and Y. Ye. 2008. Constituents from Clausena excavata. Chemical & Pharmaceutical Bulletin 56: 827–830.
Acknowledgments
We thank the Royal Golden Jubilee Scholarship (PHD/0008/2553) and the National Research University Project of Thailand through the Advanced Functional Materials Cluster of Khon Kaen University for financial support. The Center of Excellence for Innovation in Chemistry (PERCH-CIC), Office of the Higher Education Commission, Ministry of Education is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sriphana, U., Thongsri, Y., Prariyachatigul, C. et al. Clauraila E from the roots of Clausena harmandiana and antifungal activity against Pythium insidiosum . Arch. Pharm. Res. 36, 1078–1083 (2013). https://doi.org/10.1007/s12272-013-0115-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0115-5