Abstract
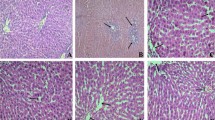
Paeoniflorin (PF), a monoterpene glucoside, is a primary bioactive component of paeony, the root extract of Paeonia lactiflora. We tested the antioxidant effects of PF and its ability to prevent lipopolysaccharide (LPS)-induced oxidative stress. We intraperitoneally administered PF (2.5, 5, or 10 mg/kg) to rats for 20 days. On day 21, we injected the rats with LPS 4 h before sacrifice and measured serum levels of glutamate oxaloacetate transaminase, glutamate pyruvate transaminase, lactate dehydrogenase as well as the levels of malondialdehyde, superoxide dismutase, catalase, and glutathione peroxidase in liver whole-cell homogenates and mitochondrial fractions. LPS treatment increased levels of glutamate oxaloacetate transaminase, glutamate pyruvate transaminase, lactate dehydrogenase, and malondialdehyde, but PF treatment blocked these increases. LPS treatment also decreased antioxidant levels of superoxide dismutase, catalase, and glutathione peroxidase, but PF blocked these decreases. PF also protected liver tissue, as shown by histopathology. These results suggest that PF can protect against LPS-induced liver inflammation.
Similar content being viewed by others
References
Aderem, A. and Ulevitch, R. J., Toll-like receptors in the induction of the innate immune response. Nature, 406, 782–787 (2000).
Aebi, H., A Catalase in vitro. Meth. Enzymol., 105, 121–126 (1984).
Aniya, Y., Koyama, T., Miyagi, C., Miyahira, M., Inomata, C., Kinoshita, S., and Ichiba, T., Free radical scavenging and hepatoprotective actions of the medicinal herb, Crassocephalum crepidioides from the Okinawa Islands. Biol. Pharm. Bull., 28, 19–23 (2005).
Bautista, A. P., Meszaros, K., Bojta, J., and Spitzer, J. J., Superoxide anion generation in the liver during the early stage of endotoxemia in rats. J. Leukoc. Biol., 48, 123–128 (1990).
Beauchamp, C. and Fridovich, I., Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal. Biochem., 44, 276–287 (1971).
Beutler, B. and Rietschel, E. T., Innate immune sensing and its roots: the story of endotoxin. Immunology, 3, 169–176 (2003).
Butterfield, D. A. and Kanski, J., Brain protein oxidation in agerelated neurodegenerative disorders that are associated with aggregated proteins. Mech. Ageing Dev., 122, 945–962 (2001).
Dezaki, K., Kimura, I., Miyahara, K., and Kimura, M., Complementary effects of paeoniflorin and glycyrrhizin on intracellular Ca2+ mobilization in the nerve-stimulated skeletal muscle of mice. Jpn. J. Pharmacol., 69, 281–284 (1996).
Finkel, T., Oxygen radicals and signaling. Curr. Opin. Cell Biol., 10, 248–253 (1998).
Fridovich, I., Superoxide radical and superoxide dismutase. Acc. Chem. Res., 5, 321–323 (1972).
Frings, C. S. and Dunn, R. T., A colorimetric method for determination of total serum lipid based on the sulfuricphospho-vanillin reaction. Am. J. Clin. Pathol., 53, 89–92 (1970).
Ha, B. J., Lee, S. H., Kim, H. J., and Lee, J. Y., The role of salicornia herbacea in ovariectomy-induced oxidative stress. Biol. Pharm. Bull., 29, 1305–1309 (2006).
Hall, E. D. and Braughler, J. R., Role of lipid peroxidation in post-traumatic spinal cord degeneration: a review. Cent. Nerv. Syst. Trauma., 3, 281–294 (1986).
Hassid, A., Arabshahi, H., Bourcier, T., Dhaunsi, G.S. and Matthews, C., Nitric oxide selectively amplifies FGF-2-induced mitogenesis in primary rat aortic smooth muscle cells. Am. J. Physiol., 267, 1040–1048 (1994).
Hoffmann, J. A., Kafatos, F. C., Janeway, C. A., and Ezekowitz, R. A., Phylogenetic perspectives in innate immunity. Science, 284, 1313–1317 (1999).
Hsu, F. L., Lai, C. W., and Cheng, J. T., Antihyperglycemic effects of paeoniflorin and 8-debenzoylpaeoniflorin, glucosides from the root of Paeonia lactiflora. Planta Med., 63, 323–325 (1997).
Jones, P. and Suggett, A., The catalase-hydrogen peroxide system. A theoretical appraisal of the mechanism of catalase action. Biochem. J., 110, 621–629 (1968).
Kao, E. S., Wang, C. J., Lin, W. L., Yin, Y. F., Wang, C. P., and Tseng, T. H., Anti-inflammatory potential of flavonoid contents from dried fruit of Crataegus pinnatifida in vitro and in vivo. J. Agric. Food Chem., 53, 430–436 (2005).
Kimura, M., Kimura, I., Muroi, M., Nakamura, T., and Shibata, S., Depolarizing effects of glycyrrhizin-derivatives relating to the blend effects with paeoniflorin in mouse diaphragm muscle. Jpn. J. Pharmacol., 41, 263–265 (1986).
Ketterer, B., Detoxification reactions of glutathione and glutathione reductase. Xenobiotica, 16, 957–975 (1986).
Kono, H., Asakawa, M., Fujii, H., Maki, A., Amemiya, H., Yamamoto, M., Matsuda, M., and Matsumoto, Y., Edaravone, A novel free radical scavenger, prevents liver injury and mortality in rats administered endotoxin. J. Pharmacol. Exp. Ther., 307, 74–82 (2003).
Lakics, V. and Vogel, S. N., Lipopolysaccharide and ceramide use divergent signaling pathways to induce cell death in murine macrophages. J. Immunol., 161, 2490–2500 (1998).
Lawrence, R. A. and Burk, R. F., Glutathione peroxidase activity in selenium-deficient rat liver. Biochem. Biophys. Res. Commun., 71, 952–958 (1976).
Levy, E. and Ruebner, B. H., Hepatic changes produced by a single dose of endotoxin in the mouse. Light microscopy and histochemistry. Am. J. Pathol., 51, 269–285 (1967).
Liang, J., Zhou, A., Chen, M., and Xu, S., Negatively regulatory effects of paeoniflorin on immune cells. Eur. J. Pharmacol., 183, 901–902 (1990).
Lim, H. K., Kim, H. S., Choi, H. S., Oh, S., Jang, C. G., Choi, J., Kim, S. H., and Chang, M. J., Effects of acetylbergenin against d-galactosamine induced hepatotoxicity in rats. Pharmacol. Res., 5, 471–474 (2000).
Lima, C. F., Carvalho, F., Fernandes, E., Bastos, M. L., Santos-Gomes, P. C., Fermandes-Ferreira, M., and Pereira-Wilson, C., Evaluation of toxic/protective effects of the essential oil of Salvia officinalis on freshly isolated rat hepatocytes. Toxicol. In Vitro, 18, 457–465 (2004).
Lowry, O. H. and Rosenbrough, N. J., Protein measurement with folin reagent. J. Biol. Chem., 193, 265–275 (1951).
Machlin, L. J. and Bendich, A., Free radical tissue damage: protective role of antioxidant nutrients. FASEB J., 1, 441–445 (1987).
Matsuda, H., Ishikado, A., Nishida, N., Ninomiya, K., Fujiwara, H., Kobayashi, Y., and Yoshikawa, M., Hepatoprotective, superoxide scavenging, and antioxidative activities of aromatic constituents from the bark of Betula platyphylla var. japonica. Bioorg. Med. Chem. Lett., 8, 2939–2944 (1998).
Mitra, S. K., Venkataranganna, M. V., Sundaram, R., and Gopumadhavan, S., Protective effect of HD-03, a herbal formulation, against various hepatotoxic agents in rats. J. Ethnopharmacol., 63, 181–186 (1998).
Moncada, S., Palmer, R. M., and Higgs, E. A., Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol. Rev., 43, 109–142 (1991).
Ohta, H., Ni, J. W., Matsumoto, K., Watanabe, H., and Shimizu, M., Peony and its major constituent, paeoniflorin, improve radial maze performance impaired by scopolamine in rats. Pharmacol. Biochem. Behav., 45, 719–723 (1993).
Pari, L. and Ashod, K. N., Hepatoprotective activity of moringuoleifera on antitubercular drug induced liver damage in rats. J. Med. Food, 5, 171–177 (2002).
Pari, L. and Marugan, P., Protective role of tetrahydrocur cumin against erythromycin estolate-induced hepatotoxicity. Pharmacol. Res., 49, 481–486 (2004).
Park, J. Y., Park, C. M., Kim, J. J., Noh, K. H., Cho, C. W., and Song, Y. S., The protective effect of chlorophyll a aginst oxidative stress and inflammatory processes in LPS-stimulated macrophages. Food Sci. Biotechnol., 16,205–211 (2007).
Peter, O., Marjolijn, T. M., Marina, H. J., Frans, D., Barbro, N. M., Han, M., Maarten, J. H. S., Dirk, K. F. M., Klaas, P., and Geny, M. M. G., Rat liver slices as a tool to study LPS-induced inflammatory response in the liver. J. Hepatol., 35, 187–194 (2001).
Rupprecht, H. D., Akagi, Y., Keil, A., and Hofer, G., Nitric oxide inhibits growth of glomerular mesangial cells: role of the transcription factor EGR-1. Kidney Int., 57, 70–82 (2000).
Sallie, R., Tredger, J. M., and William, R., Drugs and the liver. Biopharm. Drug Dispos., 12, 251–259 (1997).
Sandhir, R., Julka, D., and Gill, K. D., Lipoperoxidative damage on lead exposure in rat brain and its implicationa on membrane bound enzymes. Pharmacol. Toxicol., 74, 66–71 (1994).
Schmidt, H. H. and Murad, F., Purification and characterization of a human NO synthase. Biochem. Biophys. Res. Commun., 181, 1372–1377 (1991).
Shoji, Y., Uedono, Y., Ishikura, H., Takeyama, N., and Tanaka, T., DNA damage induced by tumour necrosis factor-alpha in L929 cells is mediated by mitochondrial oxygen radical formation. Immunology, 84, 543–548 (1995).
Stuehr, D. J. and Nathan, C. F., Nitric oxide. A macrophage product responsible for cytostasis and respiratory inhibition in tumor target cell. J. Exp. Med., 169, 1543–1555 (1989).
Sugino, K., Dohi, K., Yamada, K., and Kawasaki, T., The role of lipid peroxidation in endotoxin-induced hepatic damage and the protective effect of antioxidants. Surgery, 101, 746–752 (1987).
Sugishita, E., Amagaya, S., and Ogihara, Y., Studies on the combination of Glycyrrhizae Radix in Shakuyakukanzo-To. J. Pharmacobio-dyn., 7, 427–435 (1984).
Takagi, K. and Harada, M., Pharmacological studies on herb paeony root. Central effects of paeoniflorin, and combined effects with licorice component FM100. Yakugaku Zasshi, 89, 879–886 (1969).
Tamaya, T., Sato, S., and Okada, H. H., Possible mechanism of steroid action of the plant herb extracts glycyrrhizin, glycyrrhetinic acid, and paeoniflorin: Inhibition by plant herb extracts of steroid protein binding in the rabbit. Am. J. Obstet. Gynecol., 155, 1134–1139 (1986).
Tsuboi, H., Hossain, K., Akhand, A. A., Takeda, K., and Du, J, R. M., Paeoniflorin induces apoptosis of lymphocytes through a redoxlinked mechanism. J. Cell. Biochem., 93, 162–172 (2004).
Vilwanathan, R. and Kanchi, S. S., Thiruvengadam, Devaki., Hepatoprotective activity of Tridax procumbens against d-galactosamine/lipopolysaccharide induced hepatitis in rats. J. Ethnopharmacol., 101, 55–60 (2005).
Wills, P. J. and Asha, V. V., Protective effect of Lygodium flexuosum (L.) Sw. extract against carbon tetrachlorideinduced acute liver injury in rats. J. Ethnopharmacol., 108, 320–326 (2006).
Yamahara, J., Yamada, T., Kimura, H., Sawada, T., and Fujimura, H., Biologically active principles of crude drugs: Antiallergic principles of “shoseiryu-to”: Effect on delayedtype allergy reaction. Yakugaku Zasshi, 102, 881–886 (1982).
Yan, D., Saito, K., Ohmi, Y., Fujie, N., and Ohtsuka, K., Paeoniflorin, a novel heat shock protein-inducing compound. Cell Stress Chaperones, 9, 378–389 (2004).
Yoon, H. J., Moon, M. E., Park, H. S., Im, S. Y., and Kim, Y. H., Chitosan oligosaccharide (COS) inhibits LPS-induced inflammatory effects in RAW 264.7 macrophage cells. Biochem. Biophys. Res. Commun., 358, 954–959 (2007).
Yousef, M. I., Aluminium-induced changes in hematobiochemical parameters, lipid peroxidation and enzyme activities of male rabbits: protective role of ascorbic acid. Toxicology, 199, 47–57 (2004).
Yousef, M. I., Awad, T. I., and Mohamed, E. H., Deltamethrin-induced oxidative damage and biochemical alterations in rats and its attenuation by vitamin E. Toxicology, 227, 240–247 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, I.D., Ha, B.J. The effects of paeoniflorin on LPS-induced liver inflammatory reactions. Arch. Pharm. Res. 33, 959–966 (2010). https://doi.org/10.1007/s12272-010-0620-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-0620-8