Abstract
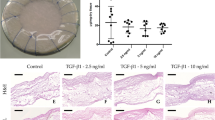
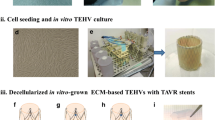
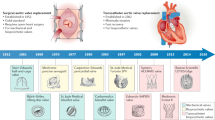
Tissue-engineered heart valves with self-repair and regeneration properties may overcome the problem of long-term degeneration of currently used artificial prostheses. The aim of this study was the development and in vivo proof-of-concept of next-generation off-the-shelf tissue-engineered sinus valve (TESV) for transcatheter pulmonary valve replacement (TPVR). Transcatheter implantation of off-the-shelf TESVs was performed in a translational sheep model for up to 16 weeks. Transapical delivery of TESVs was successful and showed good acute and short-term performance (up to 8 weeks), which then worsened over time most likely due to a non-optimized in vitro valve design. Post-mortem analyses confirmed the remodelling potential of the TESVs, with host cell infiltration, polymer degradation, and collagen and elastin deposition. TESVs proved to be suitable as TPVR in a preclinical model, with encouraging short-term performance and remodelling potential. Future studies will enhance the clinical translation of such approach by improving the valve design to ensure long-term functionality.






Similar content being viewed by others
Abbreviations
- TPVR:
-
Transcatheter Pulmonary Valve Replacement
- TE:
-
Tissue Engineering
- TEHV(s):
-
Tissue-engineered heart valve(s)
- TESV(s):
-
Tissue-Engineered Sinus Valve(s)
- GLP:
-
Good laboratory practice
- ECM:
-
Extracellular matrix
- GAGs:
-
Glycosaminoglycans
- HYP:
-
Hydroxyproline
- H&E:
-
Hematoxilin and Eosin
- α-SMA:
-
Alpha-smooth muscle actin
- VG-EL:
-
Van Gieson-Elastin
- Vim:
-
Vimentin
References
Ghawi, H., Kenny, D., & Hijazi, Z. M. (2012). Transcatheter pulmonary valve replacement. Cardiology and Therapy, 1, 5.
Kenny, D. P., & Hijazi, Z. M. (2017). Current status and future potential of transcatheter interventions in congenital heart disease. Circulation Research, 120, 1015–1026.
Emmert, M. Y., & Hoerstrup, S. P. (2016). Tissue engineered heart valves: Moving towards clinical translation. Expert Review of Medical Devices, 13, 417–419.
Driessen-Mol, A., Emmert, M. Y., Dijkman, P. E., Frese, L., Sanders, B., Weber, B., Cesarovic, N., Sidler, M., Leenders, J., Jenni, R., Grunenfelder, J., Falk, V., Baaijens, F. P., & Hoerstrup, S. P. (2014). Transcatheter implantation of homologous "off-the-shelf" tissue-engineered heart valves with self-repair capacity: Long-term functionality and rapid in vivo remodeling in sheep. Journal of the American College of Cardiology, 63, 1320–1329.
Schmitt, B., Spriestersbach, H., O.H.I, D., Radtke, T., Bartosch, M., Peters, H., Sigler, M., Frese, L., Dijkman, P. E., Baaijens, F. P., Hoerstrup, S. P., & Berger, F. (2016). Percutaneous pulmonary valve replacement using completely tissue-engineered off-the-shelf heart valves: Six-month in vivo functionality and matrix remodelling in sheep. EuroIntervention, 12, 62–70.
Kluin, J., Talacua, H., Smits, A. I., Emmert, M. Y., Brugmans, M. C., Fioretta, E. S., Dijkman, P. E., Sontjens, S. H., Duijvelshoff, R., Dekker, S., Janssen-van den Broek, M. W., Lintas, V., Vink, A., Hoerstrup, S. P., Janssen, H. M., Dankers, P. Y., Baaijens, F. P., & Bouten, C. V. (2017). In situ heart valve tissue engineering using a bioresorbable elastomeric implant - from material design to 12 months follow-up in sheep. Biomaterials, 125, 101–117.
Emmert, M. Y., Weber, B., Behr, L., Frauenfelder, T., Brokopp, C. E., Grunenfelder, J., Falk, V., & Hoerstrup, S. P. (2011). Transapical aortic implantation of autologous marrow stromal cell-based tissue-engineered heart valves: First experiences in the systemic circulation. JACC. Cardiovascular Interventions, 4, 822–823.
Syedain, Z., Reimer, J., Schmidt, J., Lahti, M., Berry, J., Bianco, R., & Tranquillo, R. T. (2015). 6-month aortic valve implantation of an off-the-shelf tissue-engineered valve in sheep. Biomaterials, 73, 175–184.
Weber, B., Dijkman, P. E., Scherman, J., Sanders, B., Emmert, M. Y., Grunenfelder, J., Verbeek, R., Bracher, M., Black, M., Franz, T., Kortsmit, J., Modregger, P., Peter, S., Stampanoni, M., Robert, J., Kehl, D., van Doeselaar, M., Schweiger, M., Brokopp, C. E., Walchli, T., Falk, V., Zilla, P., Driessen-Mol, A., Baaijens, F. P., & Hoerstrup, S. P. (2013). Off-the-shelf human decellularized tissue-engineered heart valves in a non-human primate model. Biomaterials, 34, 7269–7280.
Katayama, S., Umetani, N., Sugiura, S., & Hisada, T. (2008). The sinus of Valsalva relieves abnormal stress on aortic valve leaflets by facilitating smooth closure. The Journal of Thoracic and Cardiovascular Surgery, 136, 1528–35e1.
Salica, A., Pisani, G., Morbiducci, U., Scaffa, R., Massai, D., Audenino, A., Weltert, L., Guerrieri Wolf, L., & De Paulis, R. (2016). The combined role of sinuses of Valsalva and flow pulsatility improves energy loss of the aortic valve. European Journal of Cardio-Thoracic Surgery, 49, 1222–1227.
Pisani, G., Scaffa, R., Ieropoli, O., Dell'Amico, E. M., Maselli, D., Morbiducci, U., & De Paulis, R. (2013). Role of the sinuses of Valsalva on the opening of the aortic valve. The Journal of Thoracic and Cardiovascular Surgery, 145, 999–1003.
Loerakker, S., Ristori, T., & Baaijens, F. P. (2016). A computational analysis of cell-mediated compaction and collagen remodeling in tissue-engineered heart valves. Journal of the Mechanical Behavior of Biomedical Materials, 58, 173–187.
Sanders, B., Loerakker, S., Fioretta, E. S., Bax, D. J. P., Driessen-Mol, A., Hoerstrup, S. P., & Baaijens, F. P. T. (2016). Improved geometry of decellularized tissue engineered heart valves to prevent leaflet retraction. Annals of Biomedical Engineering, 44, 1061–1071.
Dijkman, P. E., Driessen-Mol, A., Frese, L., Hoerstrup, S. P., & Baaijens, F. P. (2012). Decellularized homologous tissue-engineered heart valves as off-the-shelf alternatives to xeno- and homografts. Biomaterials, 33, 4545–4554.
Mol, A., Driessen, N. J., Rutten, M. C., Hoerstrup, S. P., Bouten, C. V., & Baaijens, F. P. (2005). Tissue engineering of human heart valve leaflets: A novel bioreactor for a strain-based conditioning approach. Annals of Biomedical Engineering, 33, 1778–1788.
Mol, A., van Lieshout, M. I., Dam-de Veen, C. G., Neuenschwander, S., Hoerstrup, S. P., Baaijens, F. P., & Bouten, C. V. (2005). Fibrin as a cell carrier in cardiovascular tissue engineering applications. Biomaterials, 26, 3113–3121.
Benes, P., Maceckova, V., Zdrahal, Z., Konecna, H., Zahradnickova, E., Muzik, J., & Smarda, J. (2006). Role of vimentin in regulation of monocyte/macrophage differentiation. Differentiation, 74, 265–276.
Mor-Vaknin, N., Punturieri, A., Sitwala, K., & Markovitz, D. M. (2003). Vimentin is secreted by activated macrophages. Nature Cell Biology, 5, 59–63.
Langer, R., & Vacanti, J. P. (1993). Tissue engineering. Science, 260, 920–926.
Loerakker, S., Argento, G., Oomens, C. W., & Baaijens, F. P. (2013). Effects of valve geometry and tissue anisotropy on the radial stretch and coaptation area of tissue-engineered heart valves. Journal of Biomechanics, 46, 1792–1800.
Toninato, R., Salmon, J., Susin, F. M., Ducci, A., & Burriesci, G. (2016). Physiological vortices in the sinuses of Valsalva: An in vitro approach for bio-prosthetic valves. Journal of Biomechanics, 49, 2635–2643.
Bellhouse, B. J., & Bellhouse, F. H. (1968). Mechanism of closure of the aortic valve. Nature, 217, 86–87.
Markl, M., Kilner, P. J., & Ebbers, T. (2011). Comprehensive 4D velocity mapping of the heart and great vessels by cardiovascular magnetic resonance. Journal of Cardiovascular Magnetic Resonance, 13, 7.
Kvitting, J. P., Ebbers, T., Wigstrom, L., Engvall, J., Olin, C. L., & Bolger, A. F. (2004). Flow patterns in the aortic root and the aorta studied with time-resolved, 3-dimensional, phase-contrast magnetic resonance imaging: Implications for aortic valve-sparing surgery. The Journal of Thoracic and Cardiovascular Surgery, 127, 1602–1607.
Ranga, A., Bouchot, O., Mongrain, R., Ugolini, P., & Cartier, R. (2006). Computational simulations of the aortic valve validated by imaging data: Evaluation of valve-sparing techniques. Interactive Cardiovascular and Thoracic Surgery, 5, 373–378.
Leo, H. L., Simon, H., Carberry, J., Lee, S. C., & Yoganathan, A. P. (2005). A comparison of flow field structures of two tri-leaflet polymeric heart valves. Annals of Biomedical Engineering, 33, 429–443.
Saikrishnan, N., Yap, C. H., Milligan, N. C., Vasilyev, N. V., & Yoganathan, A. P. (2012). In vitro characterization of bicuspid aortic valve hemodynamics using particle image velocimetry. Annals of Biomedical Engineering, 40, 1760–1775.
Ducci, A., Tzamtzis, S., Mullen, M. J., & Burriesci, G. (2013). Hemodynamics in the Valsalva sinuses after transcatheter aortic valve implantation (TAVI). The Journal of Heart Valve Disease, 22, 688–696.
Suleiman, T., Kavinsky, C. J., Skerritt, C., Kenny, D., Ilbawi, M. N., & Caputo, M. (2015). Recent development in pulmonary valve replacement after tetralogy of Fallot repair: The emergence of hybrid approaches. Frontiers in Surgery, 2, 22.
Butera, G., Milanesi, O., Spadoni, I., Piazza, L., Donti, A., Ricci, C., Agnoletti, G., Pangrazi, A., Chessa, M., & Carminati, M. (2013). Melody transcatheter pulmonary valve implantation. Results from the registry of the Italian Society of Pediatric Cardiology. Catheterization and Cardiovascular Interventions, 81, 310–316.
Cheatham, J. P., Hellenbrand, W. E., Zahn, E. M., Jones, T. K., Berman, D. P., Vincent, J. A., & McElhinney, D. B. (2015). Clinical and hemodynamic outcomes up to 7 years after transcatheter pulmonary valve replacement in the US melody valve investigational device exemption trial. Circulation, 131, 1960–1970.
McElhinney, D. B., Hellenbrand, W. E., Zahn, E. M., Jones, T. K., Cheatham, J. P., Lock, J. E., & Vincent, J. A. (2010). Short- and medium-term outcomes after transcatheter pulmonary valve placement in the expanded multicenter US melody valve trial. Circulation, 122, 507–516.
Eicken, A., Ewert, P., Hager, A., Peters, B., Fratz, S., Kuehne, T., Busch, R., Hess, J., & Berger, F. (2011). Percutaneous pulmonary valve implantation: Two-centre experience with more than 100 patients. European Heart Journal, 32, 1260–1265.
Van Dijck, I., Budts, W., Cools, B., Eyskens, B., Boshoff, D. E., Heying, R., Frerich, S., Vanagt, W. Y., Troost, E., & Gewillig, M. (2015). Infective endocarditis of a transcatheter pulmonary valve in comparison with surgical implants. Heart, 101, 788–793.
Lee, C., Park, C. S., Lee, C. H., Kwak, J. G., Kim, S. J., Shim, W. S., Song, J. Y., Choi, E. Y., & Lee, S. Y. (2011). Durability of bioprosthetic valves in the pulmonary position: Long-term follow-up of 181 implants in patients with congenital heart disease. The Journal of Thoracic and Cardiovascular Surgery, 142, 351–358.
Freling, H. G., van Slooten, Y. J., van Melle, J. P., Ebels, T., Hoendermis, E. S., Berger, R. M., Hillege, H. L., Waterbolk, T. W., van Veldhuisen, D. J., Willems, T. P., & Pieper, P. G. (2015). Pulmonary valve replacement: Twenty-six years of experience with mechanical valvar prostheses. The Annals of Thoracic Surgery, 99, 905–910.
Miller, D. C. (2003). Valve-sparing aortic root replacement in patients with the Marfan syndrome. The Journal of Thoracic and Cardiovascular Surgery, 125, 773–778.
Al-Radi, O. O. (2016). Aortic ring autograft for reconstruction of the neo-pulmonary root in the arterial switch operation. The Journal of Thoracic and Cardiovascular Surgery, 151, e89–e91.
Settepani, F., Bergonzini, M., Barbone, A., Citterio, E., Basciu, A., Ornaghi, D., Gallotti, R., & Tarelli, G. (2009). Reimplantation valve-sparing aortic root replacement with the Valsalva graft: What have we learnt after 100 cases? Interactive Cardiovascular and Thoracic Surgery, 9, 113–116.
Dodge-Khatami, A., Hallhagen, S., Limacher, K., Soderberg, B., & Jenni, R. (2012). Minimally invasive insertion of an equine stented pulmonary valve with a built-in sinus portion in a sheep model. Catheterization and Cardiovascular Interventions, 79, 654–658.
Schmidt, D., Dijkman, P. E., Driessen-Mol, A., Stenger, R., Mariani, C., Puolakka, A., Rissanen, M., Deichmann, T., Odermatt, B., Weber, B., Emmert, M. Y., Zund, G., Baaijens, F. P., & Hoerstrup, S. P. (2010). Minimally-invasive implantation of living tissue engineered heart valves: A comprehensive approach from autologous vascular cells to stem cells. Journal of the American College of Cardiology, 56, 510–520.
Gottlieb, D., Kunal, T., Emani, S., Aikawa, E., Brown, D. W., Powell, A. J., Nedder, A., Engelmayr Jr., G. C., Melero-Martin, J. M., Sacks, M. S., & Mayer Jr., J. E. (2010). In vivo monitoring of function of autologous engineered pulmonary valve. The Journal of Thoracic and Cardiovascular Surgery, 139, 723–731.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All the authors have no conflicts of interest to declare. Author Sarah E. Motta declares that she has no conflict of interest. Author Emanuela S. Fioretta declares that she has no conflict of interest. Author Petra E. Dijkman declares that she has no conflict of interest. Author Valentina Lintas declares that she has no conflict of interest. Author Luc Behr declares that he has no conflict of interest. Author Simon P. Hoerstrup declares that he has no conflict of interest. Author Maximilian Y. Emmert declares that he has no conflict of interest.
Ethical Approval for Research Involving Animals
All institutional and national guidelines for the care and use of laboratory animals were followed and approved by the appropriate institutional committees. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Ethical Approval for Research Involving Humans
This article does not contain any studies with human participants performed by any of the authors.
Clinical Relevance of the Study
Valve geometry is considered a key factor to achieve a favourable remodelling and long-term functionality in TEHVs thereby building the basis for a safe clinical translation. To date, none of the currently used transcatheter TEHV solutions feature the sinus of Valsalva in their stent geometry. In this study, we demonstrated the technical feasibility of combining a sinus-shape stent with in vitro grown ECM-based TEHVs fully compatible with transcatheter implantation procedures.
Additional information
Associate Editor Adrian Chester oversaw the review of this article
Electronic Supplementary Material
ESM 1
(DOCX 8352 kb)
Rights and permissions
About this article
Cite this article
Motta, S.E., Fioretta, E.S., Dijkman, P.E. et al. Development of an Off-the-Shelf Tissue-Engineered Sinus Valve for Transcatheter Pulmonary Valve Replacement: a Proof-of-Concept Study. J. of Cardiovasc. Trans. Res. 11, 182–191 (2018). https://doi.org/10.1007/s12265-018-9800-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-018-9800-6