Abstract
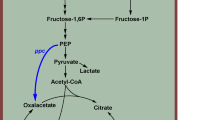
5-aminolevulinic acid (ALA) has extensive use in photodynamic cancer therapy, tumor diagnosis, and agriculture. In the microbial production of ALA, most efforts have focused on engineering enzymes and the metabolic pathways involved in ALA biosynthesis. The aim of this study was to enhance ALA production using recombinant Streptomyces coelicolor expressing the ALA synthase gene (hem A) of Rhodobacter sphaeroides with a novel two-stage pH control strategy. Batch cultures were performed in production medium at different pH values. Although cells grew well at neutral pH (6.8–7.2), the highest amount of ALA was produced with a long culture time (140 h) at a weakly acidic pH (5.5–6.0). In response, a two-stage pH control strategy was developed in which pH was maintained at 6.8–7.2 for cell growth and then shifted to 5.5–6.0 to promote ALA synthesis, resulting in a significant enhancement in ALA production compared to a one-stage pH control strategy. The titer of ALA was further improved up to 482 mg/L in the two-stage pH culture by supplying more glucose in the medium and shifting the pH during the early phase of cultivation.
Similar content being viewed by others
References
Liu, S., G. Zhang, X. Li, and J. Zhang (2014) Microbial production and applications of 5-aminolevulinic acid. Appl. Microbiol. Biotechnol. 98: 7349–7357.
Sasaki, K., T. Tanaka, Y. Nishizawa, and M. Hayashi (1990) Production of a herbicide, 5-aminolevulinic acid, by Rhodobacter sphaeroides using the effluent of swine waste from an anaerobic digestor. Appl. Microbiol. Biotechnol. 32: 727–731.
Nishikawa, S., K. Watanabe, T. Tanaka, N. Miyachi, Y. Hotta, and Y. Murooka (1999) Rhodobacter sphaeroides mutants which accumulate 5-aminolevulinic acid under aerobic and dark conditions. J. Biosci. Bioeng. 87: 798–804.
Fu, W., J. Lin, and P. Cen (2007) 5-Aminolevulinate production with recombinant Escherichia coli using a rare codon optimizer host strain. Appl. Microbiol. Biotechnol. 75: 777–782.
Lin, J., W. Fu, and P. Cen (2009) Characterization of 5-aminolevulinate synthase from Agrobacterium radiobacter, screening new inhibitors for 5-aminolevulinate dehydratase from Escherichia coli and their potential use for high 5-aminolevulinate production. Bioresour. Technol. 100: 2293–2297.
Zhang, J., Z. Kang, J. Chen, and G. Du (2015) Optimization of the heme biosynthesis pathway for the production of 5-aminolevulinic acid in Escherichia coli. Sci. Rep. 5: 8584.
Li, F., Y. Wang, K. Gong, Q. Wang, Q. Liang, and Q. Qi (2014) Constitutive expression of RyhB regulates the heme biosynthesis pathway and increases the 5-aminolevulinic acid accumulation in Escherichia coli. FEMS Microbiol. Lett. 350: 209–215.
Zhang, J., H. Weng, Z. Zhou, G. Du, and Z. Kang (2019) Engineering of multiple modular pathways for high-yield production of 5-aminolevulinic acid in Escherichia coli. Bioresour. Technol. 274: 353–360.
Cui, Z., Z. Jiang, J. Zhang, H. Zheng, X. Jiang, K. Gong, Q. Liang, Q. Wang, and Q. Qi (2019) Stable and efficient biosynthesis of 5-aminolevulinic acid using plasmid-free Escherichia coli. J. Agric. Food Chem. 67: 1478–1483.
Chen, J., Y. Wang, X. Guo, D. Rao, W. Zhou, P. Zheng, J. Sun, and Y. Ma (2020) Efficient bioproduction of 5-aminolevulinic acid, a promising biostimulant and nutrient, from renewable bioresources by engineered Corynebacterium glutamicum. Biotechnol. Biofuels. 13: 41.
Ko, Y. J., S. K. You, M. Kim, E. Lee, S. K. Shin, H. M. Park, Y. Oh, and S. O. Han (2019) Enhanced production of 5-aminolevulinic acid via flux redistribution of TCA cycle toward L-glutamate in Corynebacterium glutamicum. Biotechnol. Bioprocess Eng. 24: 915–923.
Yang, P., W. Liu, X. Cheng, J. Wang, Q. Wang, and Q. Qi (2016) A new strategy for production of 5-aminolevulinic acid in recombinant Corynebacterium glutamicum with high yield. Appl. Environ. Microbiol. 82: 2709–2717.
Feng, L., Y. Zhang, J. Fu, Y. Mao, T. Chen, X. Zhao, and Z. Wang (2016) Metabolic engineering of Corynebacterium glutamicum for efficient production of 5-aminolevulinic acid. Biotechnol. Bioeng. 113: 1284–1293.
Hara, K. Y., M. Saito, H. Kato, K. Morikawa, H. Kikukawa, H. Namura, T. Fujimoto, Y. Hirono-Hara, S. Watanabe, K. Kanamaru, and A. Kondo (2019) 5-Aminolevulinic acid fermentation using engineered Saccharomyces cerevisiae. Microb. Cell Fact. 18: 194.
Zhu, C., J. Chen, Y. Wang, L. Wang, X. Guo, N. Chen, P. Zheng, J. Sun, and Y. Ma (2019) Enhancing 5-aminolevulinic acid tolerance and production by engineering the antioxidant defense system of Escherichia coli. Biotechnol. Bioeng. 116: 2018–2028.
Lee, D. H., W. J. Jun, D. H. Shin, H. Y. Cho, and B. S. Hong (2005) Effect of culture conditions on production of 5-aminolevulinic acid by recombinant Escherichia coli. Biosci. Biotechnol. Biochem. 69: 470–476.
Bunke, A., O. Zerbe, H. Schmid, G. Burmeister, H. P. Merkle, and B. Gander (2000) Degradation mechanism and stability of 5-aminolevulinic acid. J. Pharm. Sci. 89: 1335–1341.
Yen, H. W., H. P. Hsiao, and L. J. Chen (2013) The enhancement of rapamycin production using Streptomyces hygroscopicus through a simple pH-shifted control. J. Taiwan Inst. Chem. Eng. 44: 743–747.
Ren, F., L. Chen, S. Xiong, and Q. Tong (2017) Enhanced acarbose production by Streptomyces M37 using a two-stage fermentation strategy. PLoS One. 12: e0166985.
Chen, X. S., S. Li, L. J. Liao, X. D. Ren, F. Li, L. Tang, J. H. Zhang, and Z. G. Mao (2011) Production of ε-poly-L-lysine using a novel two-stage pH control strategy by Streptomyces sp. M-Z18 from glycerol. Bioprocess Biosyst. Eng. 34: 561–567.
Tran, N. T., D. N. Pham, and C. J. Kim (2019) Production of 5-aminolevulinic acid by recombinant Streptomyces coelicolor expressing hemA from Rhodobacter sphaeroides. Biotechnol. Bioprocess Eng. 24: 488–499.
Petricek, M., K. Petrickova, L. Havlicek, and J. Felsoberg (2006) Occurrence of two 5-aminolevulinate biosynthetic pathways in Streptomyces nodosus subsp. asukaensis is linked with the production of asukamycin. J. Bacteriol. 188: 5113–5123.
Kieser, T., M. J. Bibb, M. J. Buttner, K. F. Chater, and D. A. Hopwood (2000) Practical Streptomyces genetics. John Innes Centre, Norwich, UK.
Sohoni, S. V., P. M. Bapat, and A. E. Lantz (2012) Robust, small-scale cultivation platform for Streptomyces coelicolor. Microb. Cell Fact. 11: 9.
Kim, C. J., Y. K. Chang, and G. T. Chun (2000) Enhancement of kasugamycin production by pH shock in batch cultures of Streptomyces kasugaensis. Biotechnol. Prog. 16: 548–552.
Viollier, P. H., W. Minas, G. E. Dale, M. Folcher, and C. J. Thompson (2001) Role of acid metabolism in Streptomyces coelicolor morphological differentiation and antibiotic biosynthesis. J. Bacteriol. 183: 3184–3192.
Yuzbashev, T. V., E. Y. Yuzbasheva, I. A. Laptev, T. I. Sobolevskaya, T. V. Vybornaya, A. S. Larina, I. T. Gvilava, S. V. Antonova, and S. P. Sineoky (2011) Is it possible to produce succinic acid at a low pH? Bioeng. Bugs. 2: 115–119.
Guan, N. and L. Liu (2020) Microbial response to acid stress: mechanisms and applications. Appl. Microbiol. Biotechnol. 104: 51–65.
Pan, L., X. S. Chen, K. F. Wang, and Z. G. Mao (2020) Mechanisms of response to pH shock in microbial fermentation. Bioprocess Biosyst. Eng. 43: 361–372.
Stancik, L. M., D. M. Stancik, B. Schmidt, D. M. Barnhart, Y. N. Yoncheva, and J. L. Slonczewski (2002) pH-dependent expression of periplasmic proteins and amino acid catabolism in Escherichia coli. J. Bacteriol. 184: 4246–4258.
Doull, J. L. and L. C. Vining (1990) Nutritional control of actinorhodin production by Streptomyces coelicolor A3(2): suppressive effects of nitrogen and phosphate. Appl. Microbiol. Biotechnol. 32: 449–454.
Kontro, M., U. Lignell, M. R. Hirvonen, and A. Nevalainen (2005) pH effects on 10 Streptomyces spp. growth and sporulation depend on nutrients. Lett. Appl. Microbiol. 41: 32–38.
Wang, C., X. Ren, C. Yu, J. Wang, L. Wang, X. Zhuge, and X. Liu (2020) Physiological and transcriptional responses of Streptomyces albulus to acid stress in the biosynthesis of ε-poly-L-lysine. Front. Microbiol. 11: 1379.
Tretter, L., A. Patocs, and C. Chinopoulos (2016) Succinate, an intermediate in metabolism, signal transduction, ROS, hypoxia, and tumorigenesis. Biochim. Biophys. Acta. 1857: 1086–1101.
Zhang, X., T. Bao, Z. Rao, T. Yang, Z. Xu, S. Yang, and H. Li (2014) Two-stage pH control strategy based on the pH preference of acetoin reductase regulates acetoin and 2,3-butanediol distribution in Bacillus subtilis. PLoS One. 9: e91187.
Acknowledgements
This work was supported by grants (NRF-2012R1A1A2007214, 2017R1D1A1B03029032, and 2020R1F1A1054433) of the Basic Science Research Program through the National Research foundation (NRF) funded by the Ministry of Education, Science and Technology, Republic of Korea. The authors declare no conflicts of interest.
Neither ethical approval nor informed consent was required for this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Pham, D.N., Kim, CJ. A Novel Two-stage pH Control Strategy for the Production of 5-Aminolevulinic Acid Using Recombinant Streptomyces coelicolor. Biotechnol Bioproc E 26, 669–676 (2021). https://doi.org/10.1007/s12257-020-0376-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-020-0376-z