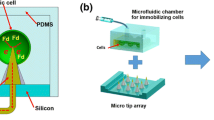
Abstract
Immobilizing cells while maintaining their long-term viability is important to utilize cells in biosensors and energy devices. In this study, we fabricated a hydrogel film of 10 μm thickness immobilizing photosynthetic cells, using a polydimethylsiloxane microfluidic device, and we monitored the viability of the cells for 30 days. Cell viability was measured by chronoamperometry using two electrodes located in the microfluidic device and was compared between hydrogel-immobilized and non-immobilized cells. The non-immobilized cells showed variation in viability. In contrast, the hydrogel-immobilized cells remained viable for 30 days. A simulation of the oxygen distribution changes by photosynthesis of the cells and mass transfer of cell culture nutrients (NaNO3) suggested that a proper environment for cell survival was effectively established inside the hydrogel. We successfully fabricated a photosynthetic cell-laden hydrogel with potential use in next-generation photosynthesis-based solar cells and sensors.
Similar content being viewed by others
References
Buenger, D., F. Topuz, and J. Groll (2012) Hydrogels in sensing applications. Prog. Polym. Sci. 37: 1678–1719.
Kim, J., K. Yang, H. J. Park, S. W. Cho, S. Han, Y. Shin, S. Chung, and J. H. Lee (2014) Implantable microfluidic device for the formation of three-dimensional vasculature by human endothelial progenitor cells. Biotechnol. Bioproc. Eng. 19: 379–385.
Heller, A. (2006) Electron-conducting redox hydrogels: Design, characteristics and synthesis. Curr. Opin. Chem. Biol. 10: 664–672.
Jen, A. C., M. C. Wake, and A. G. Mikos (1996) Review: Hydrogels for cell immobilization. Biotechnol. Bioeng. 50: 357–364.
Liu, X. W., Y. X. Huang, X. F. Sun, G. P. Sheng, F. Zhao, S. G. Wang, and H. Q. Yu (2014) Conductive carbon nanotube hydrogel as a bioanode for enhanced microbial electrocatalysis. Acs Appl. Mater. Inter. 6: 8158–8164.
Liu, X. W., Y. P. Wang, Y. X. Huang, X. F. Sun, G. P. Sheng, R. J. Zeng, F. Li, F. Dong, S. G. Wang, Z. H. Tong, and H. Q. Yu (2011) Integration of a microbial fuel cell with activated sludge process for energy-saving wastewater treatment: Taking a sequencing batch reactor as an example. Biotechnol. Bioeng. 108: 1260–1267.
Qiao, X. L., Z. Liu, Z. W. Liu, Y. L. Zeng, and Z. J. Zhang (2010) Immobilization of activated sludge in poly(ethylene glycol) by UV technology and its application in micro-polluted wastewater. Biochem. Eng. J. 50: 71–76.
Chen, Y. P., Y. Zhao, K. Q. Qiu, J. Chu, R. Lu, M. Sun, X. W. Liu, G. P. Sheng, H. Q. Yu, J. Chen, W. J. Li, G. Liu, Y. C. Tian, and Y. Xiong (2011) An innovative miniature microbial fuel cell fabricated using photolithography. Biosens. Bioelectron. 26: 2841–2846.
Lee, N. Y., Y. K. Jung, and H. G. Park (2006) On-chip colorimetric biosensor based on polydiacetylene (PDA) embedded in photopolymerized poly(ethylene glycol) diacrylate (PEG-DA) hydrogel. Biochem. Eng. J. 29: 103–108.
Nichol, J. W., S. T. Koshy, H. Bae, C. M. Hwang, S. Yamanlar, and A. Khademhosseini (2010) Cell-laden microengineered gelatin methacrylate hydrogels. Biomat. 31: 5536–5544.
Zorlutuna, P., N. Annabi, G. Camci-Unal, M. Nikkhah, J. M. Cha, J. W. Nichol, A. Manbachi, H. J. Bae, S. C. Chen, and A. Khademhosseini (2012) Microfabricated biomaterials for engineering 3D tissues. Adv. Mater. 24: 1782–1804.
Albrecht, D. R., V. L. Tsang, R. L. Sah, and S. N. Bhatia (2005) Photo-and electropatterning of hydrogel-encapsulated living cell arrays. Lab Chip 5: 111–118.
Lee, N. Y., J. R. Lim, M. J. Lee, J. B. Kim, S. J. Jo, H. K. Baik, and Y. S. Kim (2006) Hydrophilic composite elastomeric mold for high-resolution soft lithography. Langmuir 22: 9018–9022.
Chan, V., J. H. Jeong, P. Bajaj, M. Collens, T. Saif, H. Kong, and R. Bashir (2012) Multi-material bio-fabrication of hydrogel cantilevers and actuators with stereolithography. Lab Chip 12: 88–98.
Lee, D. H., H. J. Oh, S. J. Bai, and Y. S. Song (2014) Photosynthetic solar cell using nanostructured proton exchange membrane for microbial biofilm prevention. Acs Nano 8: 6458–6465.
Morishima, K., Y. Tanaka, M. Ebara, T. Shimizu, A. Kikuchi, M. Yamato, T. Okano, and T. Kitamori (2006) Demonstration of a bio-microactuator powered by cultured cardiomyocytes coupled to hydrogel micropillars. Sensor Actuat. B-Chem. 119: 345–350.
Chen, S., J. J. Duan, M. Jaroniec, and S. Z. Qiao (2014) Nitrogen and oxygen dual-doped carbon hydrogel film as a substrate-free electrode for highly efficient oxygen evolution reaction. Adv. Mater. 26: 2925–2930.
Giraldo, J. P., M. P. Landry, S. M. Faltermeier, T. P. McNicholas, N. M. Iverson, A. A. Boghossian, N. F. Reuel, A. J. Hilmer, F. Sen, J. A. Brew, and M. S. Strano (2014) Plant nanobionics approach to augment photosynthesis and biochemical sensing. Nat. Mater. 13: 530–530.
Xu, H., J. Wu, C. C. Chu, and M. L. Shuler (2012) Development of disposable PDMS micro cell culture analog devices with photopolymerizable hydrogel encapsulating living cells. Biomed. Microdev. 14: 409–418.
Park, J. H., Y. S. Song, J. G. Ha, Y. K. Kim, S. K. Lee, and S. J. Bai (2013) Electrochemical sensing of high density photosynthetic cells using a microfluidic chip. Sensor Actuat. B-Chem 188: 1300–1305.
Ha, J. G., Y. S. Song, S. Jung, S. Jang, Y. K. Kim, S. J. Bai, J. H. Park, and S. K. Lee (2017) Novel microbial photobioelectrochemical cell using an invasive ultramicroelectrode array and a microfluidic chamber. Biotechnol. Lett. 39: 849–855.
Ryu, W., S. J. Bai, J. S. Park, Z. B. Huang, J. Moseley, T. Fabian, R. J. Fasching, A. R. Grossman, and F. B. Prinz (2010) Direct extraction of photosynthetic electrons from single algal cells by nanoprobing system. Nano Lett. 10: 1137–1143.
Bong, K. W., J. J. Kim, H. S. Cho, E. Lim, P. S. Doyle, and D. Irimia (2015) Synthesis of cell-adhesive anisotropic multifunctional particles by stop flow lithography and streptavidin-biotin interactions. Langmuir 31: 13165–13171.
Zanke, B. W., K. Boudreau, E. Rubie, E. Winnett, L. A. Tibbles, L. Zon, J. Kyriakis, F. F. Liu, and J. R. Woodgett (1996) The stress-activated protein kinase pathway mediates cell death following injury induced by cis-platinum, UV irradiation or heat. Curr. Biol.: CB 6: 606–613.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
You, SG., Bai, S.J. Long-term viability of photosynthetic cells stacked in a hydrogel film within a polydimethylsiloxane microfluidic device. Biotechnol Bioproc E 22, 474–480 (2017). https://doi.org/10.1007/s12257-017-0194-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-017-0194-0