Abstract
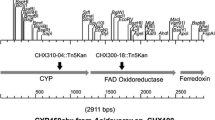
Cyclohexanone monooxygenase (CHMO), a type of Baeyer-Villiger oxidation, catalyzes the oxidation of cyclohexanone into ɛ-caprolactone, which has been utilized as a building block in organic synthesis. A bacterium that is capable of growth on cyclohexanone as a sole carbon source was recently isolated and was identified as Arthrobacter sp. L661. The strain is believed to harbor a CHMO gene (chnB), considering the high degradablity of cyclohexanone. In order to characterize the CHMO, a chnB gene was cloned from Arthrobacter sp. L661. The deduced amino acids of the chnB gene evidenced the highest degree of homology (90% identity) with the CHMO of Arthrobacter sp. BP2 (accession no. AY123972). The CHMO of L661 was shown to be functionally expressed in Escherichia coli cells, purified via affinity chromatography, and characterized. The specific activity of the purified enzyme was 24.75 μmol/min/mg protein. The optimum pH was 7.0 and the enzyme maintained over 70% of its activity for up to 24 h in a pH range of 6.0 to 8.0 at 4°C. The CHMO of L661 readily oxidized cyclobutanone and cyclopentanone whereas less activity was detected with those of Arthrobacter sp. BP2, Rhodococcus sp. Phi1, and Rhodococcus sp. Phi2, thereby suggesting that the CHMO of L661 evidenced the different substrate specificities compared with other CHMOs. These results can provide us with useful information for the development of biocatalysts applicable to commercial organic syntheses, especially because only a few CHMO genes have been identified thus far.
Similar content being viewed by others
References
Renz, M. and B. Meunier (1999) 100 years of Baeyer-Villiger oxidations. Eur. J. Org. Chem. 1999: 737–750.
Pirkle, W. H. and P. E. Adams (1978) Synthesis of the carpenter bee pheromone. Chiral 2-methyl-5-hydroxyhexanoic acid lactones. J. Org. Chem. 43: 378–379.
Renz, M., T. Blasco, A. Corma, V. Fornes, R. Jensen, and L. Nemeth (2002) Selective and shape-selective Baeyer-Villiger oxidations of aromatic aldehydes and cyclic ketones with Sn-beta zeolites and H2O2. Chemisty8: 4708–4717.
Jung, Y. J., J. S. Yoo, Y. S. Lee, I. H. Park, S. H. Kim, S. C. Lee, M. Yasuda, S. Y. Chung, and Y. L. Choi (2007) Purification and characterization of thermostable β-1,3-1,4 glucanase from Bacillus sp. A8-8. Biotechnol. Bioprocess Eng. 12: 265–270.
Lien, T. S., S. T. Yu, S. T. Wu, and J. R. Too (2007) Induction and purification of a thermophilic chitinase produced by Aeromonas sp. DYU-Too7 using glucosamin. Biotechnol. Bioprocess Eng. 12: 610–617.
Mohorčič, M., J. Friedrich, I. Renimel, P. André, D. Mandin, and J. P. Chaumont (2007) Production of melanin bleaching enzyme of fungal origin and its application in cosmetics. Biotechnol. Bioprocess Eng. 12: 200–206.
Zambianchi, F., P. Pasta, G. Carrea, S. Colonna, N. Gaggero, and J. M. Woodley (2002) Use of isolated cyclohexanone monooxygenase from recombinant Escherichia coli as a biocatalyst for Baeyer-Villiger and sulfide oxidations. Biotechnol. Bioeng. 78: 489–496.
Choi, J. H., T. K. Kim, Y. M. Kim, W. C. Kim, K. Park, and I. K. Rhee (2006) Cloning and characterization of a gene cluster for cyclohexanone oxidation in Rhodococcus sp. TK6. J. Microbiol. Biotechnol. 16: 511–518.
Choi, J. H., T. K. Kim, Y. M. Kim, W. C. Kim, G. J. Joo, K. Y. Lee, and I. K. Rhee (2005) Cloning and characterization of cyclohexanol dehydrogenase gene from Rhodococcus sp. TK6. J. Microbiol. Biotechnol. 15: 1186–1196.
Dunbar, J., L. O. Ticknor, and C. R. Kuske (2000) Assessment of microbial diversity in four Southwestern United States soils by 16S rRNA gene terminal restriction fragment analysis. Appl. Environ. Microbiol. 66: 2943–2950.
Hopwood, D. A., M. J. Bibb, K. F. Chater, T. Kieser, C. J. Bruton, H. M. Kieser, D. J. Lydiate, C. P. Smith, and H. Schrempf (1985) Genetic Manipulation of Streptomyces — A Laboratory Manual, pp. 25–38. The John Innes Foundation, Norwich, England.
Iwaki, H., Y. Hasegawa, M. Teraoka, T. Tokuyama, H. Bergeron, and P. C. K. Lau (1999) Identification of a transcriptional activator (ChnR) and a 6-oxohexanoate dehydrogenase (ChnE) in the cyclohexanol catabolic pathway in Acinetobacter sp. strain NCIMB 9871 and localization of the genes that encode them. Appl. Environ. Microbiol. 65: 5158–5162.
Brzostowicz, P. C, K. L. Gibson, S. M. Thomas, M. S. Blasko, and P. E. Rouvière (2000) Simultaneous identification of two cyclohexanone oxidation genes from an environmental Brevibacterium isolate using mRNA differential display. J. Bacteriol. 182: 4241–4248.
Brzostowicz, P. C, D. M. Walters, S. M. Thomas, V. Nagarajan, and P. E. Rouvière (2003) mRNA differential display in a microbial enrichment culture: simultaneous identification of three cyclohexanone monooxy-genases from three species. Appl. Environ. Microbiol. 69: 334–342.
Cheng, Q., S. M. Thomas, K. Kostichka, J. R. Valentine, and V. Nagarajan (2000) Genetic analysis of a gene cluster for cyclohexanol oxidation in Acinetobacter sp. strain SE19 by in vitro transposition. J. Bacteriol. 182: 4744.4751.
van Beilen, J. B., F. Mourlane, M. A. Seeger, J. Kovac, Z. Li, T. H. M. Smits, U. Fritsche, and B. Witholt (2003) Cloning of Baeyer-Villiger monooxygenases from Comamonas, Xanthobacter and Rhodococcus using polymerase chain reaction with highly degenerate primers. Environ. Microbiol. 5:174–182.
Sambrook, J., E. F. Fritsch, and T. Maniatis (1989) Molecular Cloning: A Laboratory Manual. 2nd ed., pp. 9.31–9.57. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, USA.
Altschul, S. F., T. L. Madden, A. A. Schaffer, J. Zhang, Z. Zhang, W. Miller, and D. J. Lipman (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 25: 3389–3402.
Bradford, M. M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Shin, J. H., Y. M. Kim, J. W. Park, J. E. Kim, and I. K. Rhee (2003) Resistance of Saccharomyces cerevisiae to fungicide chlorothalonil. J. Micobiol. 41: 219–223.
Branchaud, B. P. and C. T. Walsh (1985) Functional group diversity in enzymatic oxygenation reactions catalyzed by bacterial flavin-containing cyclohexanone monooxygenase. J. Am. Chem. Soc. 107: 2153–2161.
Willetts, A. (1997) Structural studies and synthetic applications of Baeyer-Villiger monooxygenases. Trends Biotechnol. 15: 55–62.
Norris, D. B. and P. W. Trudgill (1976) Multiple forms of cyclohexanone oxygenase from Nocardia globerula CL1. Eur. J. Biochem. 63: 193–198.
Trower, M. K., R. M. Buckland, R. Higgins, and M. Griffin (1985) Isolation and characterization of a cyclohexane-metabolizing Xanthobacter sp. Appl. Environ. Microbiol. 49: 1282–1289.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, YM., Jung, SH., Chung, YH. et al. Cloning and characterization of a cyclohexanone monooxygenase gene from Arthrobacter sp. L661. Biotechnol Bioproc E 13, 40–47 (2008). https://doi.org/10.1007/s12257-007-0162-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-007-0162-1