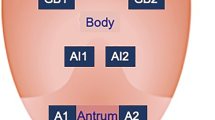
Abstract
Gastric cancer (GC) is the first cancer-related cause of death in Chile; however, no plan for GC early detection has been implemented in this country. The OLGA system characterizes gastritis from stages 0 to IV according to the risk of developing GC based on H. pylori infection, atrophy, metaplasia and GC. In this study, the performance of the OLGA system was evaluated in 485 Chilean patients receiving routine endoscopy to improve the detection of early GC or preneoplastic lesions. The results showed that OLGA scores, atrophy, metaplasia and GC increased significantly with age (p < 0.001). Conversely, H. pylori infection was higher in younger groups (p < 0.05). All gastric lesions were more frequent in men than women. The majority of patients with atrophy also had metaplasia (99%, p < 0.0001). Patients with H. pylori infection had more gastric atrophy and metaplasia than those without infection (p < 0.05). Of the 485 patients, 21 (4.3%) had GC, being 2.3 times more frequent among men than women and about 2/3 (14) were in OLGA stage ≥2. In addition, 19 (90%) GC patients had atrophy and 18 (85%) had metaplasia (p < 0.001). In conclusion, the OLGA system facilitated the evaluation of GC precursor lesions particularly in patients with an OLGA score > 2 between 45 and 56 years old, because this group showed atrophy and intestinal metaplasia more frequently. Therefore, biennial endoscopic surveillance of patients with an OLGA >2 can be an important health policy in Chile for diagnosing GC in its early stages and reducing mortality over the next two decades.

Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A (2013) Cancer statistics, 2013. CA Cancer J Clin 63:11–30. https://doi.org/10.3322/caac.21166
Brenner H, Rothenbacher D, Arndt V (2009) Epidemiology of stomach cancer. Methods Mol Biol 472:467–477. https://doi.org/10.1007/978-1-60327-492-0_23
Heise K, Bertran E, Andia ME, Ferreccio C (2009) Incidence and survival of stomach cancer in a high-risk population of Chile. World J Gastroenterol 15:1854–1862. https://doi.org/10.3748/wjg.15.1854
Ferreccio C, Rollán A, Harris PR et al (2007) Gastric cancer is related to early helicobacter pylori infection in a high-prevalence country. Cancer Epidemiol Biomark Prev 16:662–667. https://doi.org/10.1158/1055-9965.EPI-06-0514
Department of Statistics and Health Information. Ministry of Health. Chile http://www.deis.cl/defunciones-y-mortalidad-por-causas/
Mickevicius A, Ignatavicius P, Markelis R, Parseliunas A, Butkute D, Kiudelis M, Endzinas Z, Maleckas A, Dambrauskas Z (2014) Trends and results in treatment of gastric cancer over last two decades at single east European Centre: a cohort study. BMC Surg 14:98. https://doi.org/10.1186/1471-2482-14-98
Riquelme I, Letelier P, Riffo-Campos A, Brebi P, Roa J (2016) Emerging role of miRNAs in the drug resistance of gastric Cancer. Int J Mol Sci 17:424. https://doi.org/10.3390/ijms17030424
IARC (2014) Helicobacter pylori Eradication as a Strategy for Preventing Gastric Cancer (IARC Working Group Reports Volume 8)
Lim SM, Lim JY, Cho JY (2014) Targeted therapy in gastric cancer: personalizing cancer treatment based on patient genome. World J Gastroenterol 20:2042–2050. https://doi.org/10.3748/wjg.v20.i8.2042
Riquelme I, Tapia O, Leal P et al (2015) miR-101-2, miR-125b-2 and miR-451a act as potential tumor suppressors in gastric cancer through regulation of the PI3K/AKT/mTOR pathway. Cell Oncol (Dordr) 39(1):23–33. https://doi.org/10.1007/s13402-015-0247-3
Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ (2001) Helicobacter pylori infection and the development of gastric cancer. N Engl J Med 345:784–789. https://doi.org/10.1056/NEJMoa001999
Correa P, Piazuelo MB (2011) Helicobacter pylori infection and gastric adenocarcinoma. US Gastroenterol Hepatol Rev 7:59–64
Correa P (1992) Human gastric carcinogenesis: a multistep and multifactorial process--first American Cancer Society award lecture on Cancer epidemiology and prevention. Cancer Res 52:6735–6740
Pasechnikov V, Chukov S, Fedorov E, Kikuste I, Leja M (2014) Gastric cancer: prevention, screening and early diagnosis. World J Gastroenterol 20:13842–13862. https://doi.org/10.3748/wjg.v20.i38.13842
Pasechnikov VD, Chukov SZ, Kotelevets SM, Mostovov AN, Mernova VP, Polyakova MB (2005) Invasive and non-invasive diagnosis of helicobacter pylori-associated atrophic gastritis: a comparative study. Scand J Gastroenterol 40:297–301
Rugge M (2007) Secondary prevention of gastric cancer. Gut 56:1646–1647. https://doi.org/10.1136/gut.2007.133926
Zhou Y, Li H-Y, Zhang J-J, Chen XY, Ge ZZ, Li XB (2016) Operative link on gastritis assessment stage is an appropriate predictor of early gastric cancer. World J Gastroenterol 22:3670–3678. https://doi.org/10.3748/wjg.v22.i13.3670
Rugge M, Correa P, Di Mario F et al (2008) OLGA staging for gastritis: a tutorial. Dig Liver Dis 40:650–658. https://doi.org/10.1016/j.dld.2008.02.030
Rugge M, Meggio A, Pennelli G, Piscioli F, Giacomelli L, de Pretis G, Graham DY (2007) Gastritis staging in clinical practice: the OLGA staging system. Gut 56:631–636. https://doi.org/10.1136/gut.2006.106666
Dixon MF, Genta RM, Yardley JH, Correa P (1996) Classification and grading of gastritis. The updated Sydney system. International workshop on the histopathology of gastritis, Houston 1994. Am J Surg Pathol 20:1161–1181
Rugge M, Genta RM (2005) Staging and grading of chronic gastritis. Hum Pathol 36:228–233. https://doi.org/10.1016/j.humpath.2004.12.008
Ohata H, Kitauchi S, Yoshimura N, Mugitani K, Iwane M, Nakamura H, Yoshikawa A, Yanaoka K, Arii K, Tamai H, Shimizu Y, Takeshita T, Mohara O, Ichinose M (2004) Progression of chronic atrophic gastritis associated withHelicobacter pylori infection increases risk of gastric cancer. Int J Cancer 109:138–143. https://doi.org/10.1002/ijc.11680
Cho S-J, Choi IJ, Kook M-C, Nam BH, Kim CG, Lee JY, Ryu KW, Kim YW (2013) Staging of intestinal- and diffuse-type gastric cancers with the OLGA and OLGIM staging systems. Aliment Pharmacol Ther 38:1292–1302. https://doi.org/10.1111/apt.12515
Nam JH, Choi IJ, Kook M-C, Lee JY, Cho SJ, Nam SY, Kim CG (2014) OLGA and OLGIM stage distribution according to age and Helicobacter pylori status in the Korean population. Helicobacter 19:81–89. https://doi.org/10.1111/hel.12112
Satoh K, Osawa H, Yoshizawa M, Nakano H, Hirasawa T, Kihira K, Sugano K (2008) Assessment of atrophic gastritis using the OLGA system. Helicobacter 13:225–229. https://doi.org/10.1111/j.1523-5378.2008.00599.x
Capelle LG, de Vries AC, Haringsma J, ter Borg F, de Vries RA, Bruno MJ, van Dekken H, Meijer J, van Grieken NCT, Kuipers EJ (2010) The staging of gastritis with the OLGA system by using intestinal metaplasia as an accurate alternative for atrophic gastritis. Gastrointest Endosc 71:1150–1158. https://doi.org/10.1016/j.gie.2009.12.029
Ricci C, Holton J, Vaira D et al (2007) Diagnosis of helicobacter pylori: invasive and non-invasive tests. Best Pract Res Clin Gastroenterol 21:299–313. https://doi.org/10.1016/j.bpg.2006.11.002
Rollan A, Ferreccio C, Gederlini A, Serrano C, Torres J, Harris P (2006) Non-invasive diagnosis of gastric mucosal atrophy in an asymptomatic population with high prevalence of gastric cancer. World J Gastroenterol 12:7172–7178. https://doi.org/10.3748/WJG.V12.I44.7172
Namekata T, Miki K, Kimmey M et al (2000) Chronic atrophic gastritis and helicobacter pylori infection among Japanese Americans in Seattle. Am J Epidemiol 151(8):820–830
Correa P, Haenszel W, Cuello C et al (1975) A model for gastric cancer epidemiology. Lancet 306:58–60. https://doi.org/10.1016/S0140-6736(75)90498-5
Tatsuta M, Iishi H, Nakaizumi A, Okuda S, Taniguchi H, Hiyama T, Tsukuma A, Oshima A (1993) Fundal atrophic gastritis as a risk factor for gastric cancer. Int J Cancer 53:70–74. https://doi.org/10.1002/ijc.2910530114
Sipponen P, Graham DY (2007) Importance of atrophic gastritis in diagnostics and prevention of gastric cancer: application of plasma biomarkers. Scand J Gastroenterol 42:2–10. https://doi.org/10.1080/00365520600863720
Graham DY, Nurgalieva ZZ, El-Zimaity HMT et al (2006) Noninvasive versus histologic detection of gastric atrophy in a Hispanic population in North America. Clin Gastroenterol Hepatol 4:306–314. https://doi.org/10.1016/j.cgh.2005.11.003
Naylor GM, Gotoda T, Dixon M, Shimoda T, Gatta L, Owen R, Tompkins D, Axon A (2006) Why does Japan have a high incidence of gastric cancer? Comparison of gastritis between UK and Japanese patients. Gut 55:1545–1552. https://doi.org/10.1136/gut.2005.080358
Acknowledgements
This work was supported by the DIUFRO Grant (N° DI17-0132 to MV) and The National Fund for Scientific and Technological Development (FONDECYT) Grant (N° 3170826 to IR and N° 11150802 to PB).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Informed consent was obtained from all individual participants included in the study according to ethical requirements of WMA Declaration of Helsinki - Principles for medical research involving human subjects. We declare that this manuscript is original, has not been published before and is not currently being considered for publication elsewhere. The funders (see Acknowledgements) had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Additional information
Enrique Bellolio and Ismael Riquelme are both first authors.
Rights and permissions
About this article
Cite this article
Bellolio, E., Riquelme, I., Riffo-Campos, A.L. et al. Assessment of Gastritis and Gastric Cancer Risk in the Chilean Population Using the OLGA System. Pathol. Oncol. Res. 25, 1135–1142 (2019). https://doi.org/10.1007/s12253-018-0532-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-018-0532-3