Abstract
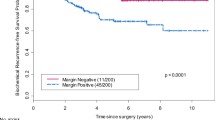
The clinical impact of lymph node dissection extent remains undetermined in the contemporary setting, as reflected in care pattern variations. Despite some series demonstrating a direct relationship between number of lymph nodes identified and detection of nodal involvement, the correlation between lymph node yield and disease control or survival outcomes remains unclear. Patients with clinically localized prostate cancer, pre-RP PSA <30, and pT2-3a/N0 disease at RP were retrospectively identified from two databases for inclusion. Those who received pre- or post-RP radiotherapy or hormone therapy were excluded. Kaplan-Meier method was employed for survival probability estimation. Cox regression models were used to assess bRFS differences between subsets. From 2002 to 2010, 667 eligible patients were identified. The median age was 61 yrs. (range, 43–76), with median PSA 5.6 ng/dL (0.9–28.0). At RP, most patients had pT2c (64%) disease with Gleason Score (GS) ≤6 (43%) or 7 (48%); 218 (33%) patients had positive margins (M+). At median clinical and PSA follow-up of 96 and 87 months, respectively, 146 patients (22%) experienced PSA failure with an estimated bRFS of 81%/76% at 5/8 years. For patients who underwent LND, univariable analysis identified PSA (at diagnosis), higher GS (≥7, at biopsy or RP), intermediate/high risk stratification, M+ as adversely associated with bRFS (all p < 0.01). A higher number of LNs excised was not associated with improved bRFS for the entire cohort (HR = 0.97, p = 0.27), nor for any clinical risk stratum, biopsy GS, or RP GS subgroup. This study did not demonstrate an association between LN yield and bRFS in patients with clinically localized pT2-3a/pN0 prostate cancer managed with RP alone, either in the entire population or with substratification by clinical risk stratum or GS.

Similar content being viewed by others
References
Ferlay JSI, Ervik M, Dikshit R, et al. GLOBOCAN 2012 v1.0, Cancer incidence and mortality worldwide: IARC CancerBase No 11 [Internet] Lyon, France: International Agency for Research on Cancer; 2013. Available from: http://globocan.iarc.fr, accessed on 01/11/2017
Lu-Yao GL, Greenberg ER (1994) Changes in prostate cancer incidence and treatment in USA. Lancet 343:251–254
Abdollah F, Suardi N, Gallina A et al (2013) Extended pelvic lymph node dissection in prostate cancer: a 20-year audit in a single center. Ann Oncol 24:1459–1466
Briganti A, Chun FK, Salonia A et al (2007) Critical assessment of ideal nodal yield at pelvic lymphadenectomy to accurately diagnose prostate cancer nodal metastasis in patients undergoing radical retropubic prostatectomy. Urology 69:147–151
Heidenreich A, Ohlmann CH, Polyakov S (2007) Anatomical extent of pelvic lymphadenectomy in patients undergoing radical prostatectomy. Eur Urol 52:29–37
Bhatta-Dhar N, Reuther AM, Zippe C, Klein EA (2004) No difference in six-year biochemical failure rates with or without pelvic lymph node dissection during radical prostatectomy in low-risk patients with localized prostate cancer. Urology 63:528–531
Allaf ME, Palapattu GS, Trock BJ, Carter HB, Walsh PC (2004) Anatomical extent of lymph node dissection: impact on men with clinically localized prostate cancer. J Urol 172:1840–1844
Kluth LA, Xylinas E, Rieken M et al (2014) Does increasing the nodal yield improve outcomes in contemporary patients without nodal metastasis undergoing radical prostatectomy? Urol Oncol 32:47.e1–47.e8
Epstein JI, Srigley J, Grignon D, Humphrey P, Association of Directors of A, Surgical P (2008) Recommendations for the reporting of prostate carcinoma: Association of Directors of Anatomic and Surgical Pathology. Am J Clin Pathol 129:24–30
D’Amico AV, Whittington R, Malkowicz SB et al Pretreatment nomogram for prostate-specific antigen recurrence after radical prostatectomy or external-beam radiation therapy for clinically localized prostate cancer. J Clin Oncol 17:168–172
Masterson TA, Bianco FJ, Vickers AJ et al (2006) The association between total and positive lymph node counts, and disease progression in clinically localized prostate cancer. J Urol 175:1320–1324
Joslyn SA, Konety BR (2006) Impact of extent of lymphadenectomy on survival after radical prostatectomy for prostate cancer. Urology 68:121–125
Schreiber D, Rineer J, Sura S et al (2011) Radical prostatectomy for cT3-4 disease: an evaluation of the pathological outcomes and patterns of care for adjuvant radiation in a national cohort. BJU Int 108:360–365
Gandaglia G, Trinh QD, Hu JC et al (2014) The impact of robot-assisted radical prostatectomy on the use and extent of pelvic lymph node dissection in the "post-dissemination" period. Eur J Surg Oncol 40:1080–1086
Berglund RK, Sadetsky N, DuChane J, Carroll PR, Klein EA (2007) Limited pelvic lymph node dissection at the time of radical prostatectomy does not affect 5-year failure rates for low, intermediate and high risk prostate cancer: results from CaPSURE. J Urol 177:526–529
Murphy AM, Berkman DS, Desai M, McKiernan JM, Badani KK (2009) The number of negative pelvic lymph nodes removed does not affect the risk of biochemical failure after radical prostatectomy. BJU Int 105:176–179
Schiavina R, Manferrari F, Garofalo M et al (2010) The extent of pelvic lymph node dissection correlates with the biochemical recurrence rate in patients with intermediate- and high-risk prostate cancer. BJU Int 108:1262–1268
Abdollah F, Sun M, Thuret R et al (2012) Lymph node count threshold for optimal pelvic lymph node staging in prostate cancer. Int J Urol 19:645–651
Brawley OW (2012) Trends in prostate cancer in the United States. J Nat Cancer Inst Monogr 2012:152–156
Briganti A, Blute ML, Eastham JH et al (2009) Pelvic lymph node dissection in prostate cancer. Eur Urol 55:1251–1265
DiMarco DS, Zincke H, Sebo TJ et al (2005) The extent of lymphadenectomy for pTXN0 prostate cancer does not affect prostate cancer outcome in the prostate specific antigen era. J Urol 173:1121–1125
Joniau S, Van den Bergh L, Lerut E et al (2013) Mapping of pelvic lymph node metastases in prostate cancer. Eur Urol 63:450–458
Pagliarulo V, Hawes D, Brands FH et al (2006) Detection of occult lymph node metastases in locally advanced node-negative prostate cancer. J Clin Oncol 24:2735–2742
Maurer T, Gschwend JE, Rauscher I et al (2016) Diagnostic efficacy of (68)Gallium-PSMA positron emission tomography compared to conventional imaging for lymph node staging of 130 consecutive patients with intermediate to high risk prostate cancer. J Urol 195:1436–1443
Mattei A, Fuechsel FG, Dhar NB et al (2008) The template of the primary lymphatic landing sites of the prostate should be revisited: results of a multimodality mapping study. Eur Urol 53:118–125
Grivas N, Wit E, Pos F et al (2017) Sentinel lymph node dissection to select clinically node-negative prostate cancer pateitns for pelvic radiation therapy: effect on biochemical and systemic progression. Int J Radiat Oncol Biol Phys 97:347–354
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None of the authors declares that he or she has any potential or actual conflicts of interest with respect to the present investigation.
Rights and permissions
About this article
Cite this article
Seyedin, S.N., Mitchell, D.L., Mott, S.L. et al. Is More Always Better? An Assessment of the Impact of Lymph Node Yield on Outcome for Clinically Localized Prostate Cancer with Low/Intermediate Risk Pathology (pT2-3a/pN0) Managed with Prostatectomy Alone. Pathol. Oncol. Res. 25, 209–215 (2019). https://doi.org/10.1007/s12253-017-0349-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-017-0349-5