Abstract
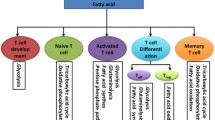
While proteins are critical for immunity, T-cells constitute a critical component of adaptive immunity by clearing cancerous cells among other abnormal cells. However, cancer cells exhibit a potential to escape T-cell control by employing mechanisms not completely delineated. Interesting work has investigated how certain amino acids affect the proliferation rate of T-cells as well as their effectiveness in clearing tumors. The role of amino acids cysteine, glutamine, phenylalanine, tryptophan and arginine in immunomodulation and particularly regarding T-cell proliferation and activation is discussed. The redox balance is reported to affect T-cell proliferation via modulation of cysteine availability. In addition antigen presenting cells (APCs), similar to myeloid cells determine the availability of amino acids in the extracellular microenvironment affecting T-cell proliferation and activation. A better mechanistic understanding of T-cell function modulation via amino acid signaling or metabolic properties may be helpful towards optimization of adaptive immunity with implications for cancer prognosis and treatment.

Similar content being viewed by others
References
Steinke JW, Lawrence MG (2014) T-cell biology in immunotherapy. Ann Allergy Asthma Immunol 112:195–199
Smith KA (2014) Revisiting the first long-term culture of antigen-specific cytotoxic T cells. Front Immunol 5:194
Viret C, Janeway CA Jr (1999) MHC and T cell development. Rev Immunogenet 1:91–104
von Essen MR, Kongsbak M, Geisler C (2012) Mechanisms behind functional avidity maturation in T cells. Clin Dev Immunol 2012:163453
Robinson AT, Miller N, Alexander DR (1993) CD3 antigen-mediated calcium signals and protein kinase C activation are higher in CD45RO+ than in CD45RA+ human T lymphocyte subsets. Eur J Immunol 23(1):61–68
von Essen MR, Kongsbak M, Schjerling P, Olgaard K, Ødum N, Geisler C (2010) Vitamin D controls T cell antigen receptor signaling and activation of human T cells. Nat Immunol 11(4):344–349
Ericsson PO, Orchansky PL, Carlow DA, Teh HS (1996) Differential activation of phospholipase C-γ1 and mitogen-activated protein kinase in naïve and antigen-primed CD4 T cells by the peptide/MHC ligand. J Immunol 156(6):2045–2053
Viganò S, Utzschneider DT, Perreau M, Pantaleo G, Zehn D, Harari A (2012) Functional avidity: a measure to predict the efficacy of effector T cells? Clin Dev Immunol 153863
Ebert PJ, Jiang S, Xie J, Li QJ, Davis MM (2009) An endogenous positively selecting peptide enhances mature T cell responses and becomes an autoantigen in the absence of microRNA miR-181a. Nat Immunol 10(11):1162–1169
Garcia Z, Pradelli E, Celli S, Beuneu H, Simon A, Bousso P (2007) Competition for antigen determines the stability of T cell-dentritic cell interactions during clonal expansion. Proc Natl Acad Sci U S A 104(11):4553–4558
McMahan RH, Slansky JE (2007) mobilizing the low-avidity T cell repertoire to kill tumors. Semin Cancer Biol 17(4):317–329
Choi EML, Chen JL, Wooldridge et al (2003) High avidity antigen-specific CTL identified by CD8-idependent tetramer staining. J Immunol 171(10):5116–5123
Xiao Z, Mescher MF, Jameson SC (2007) Detuning CD8 T cells: down-regulation of CD8 expression, tetramer binding, and response during CTL activation. J Exp Med 294(11):2667–2677
Slifka MK, Whitton JL (2001) Functional avidity maturation of CD8+ T cells without selection of higher affinity TCR. Nat Immunol 2(8):711–717
Rechtsteiner G, Warger T, Hofmann M, Rammensee HG, Schild HJ, Radsak MP (2006) Precursor frequency can compensate for lower TCR expression in T cell competition during priming in vivo. Eur J Immunol 36(10):2613–2623
Sikalidis AK, Lee J-I, Stipanuk MH (2011) Cellular responses to indispensable amino acid deficiency are mediated through the integrated stress response pathway. Amino Acids 41(1):159–171
Sikalidis AK, Stipanuk MH (2010) Growing rats respond to a sulfur amino acid-deficient diet by phosphorylation of eIF2α and induction of adaptive components of the integrated stress. J Nutr 140(6):1080–1085
Bella DL, Hirschberger LL, Hosokawa Y, Stipanuk MH (1999) The mechanisms involved in the regulation of key enzymes of cysteine metabolism in rat liver in vivo. Am J Physiol 276 (Endocrin. Metab. 39): E326–E335
Bella DL, Hahn C, Stipanuk MH (1999) The effects of non-sulfur amino acids and of sulfur amino acids on the regulation of hepatic enzymes of cysteine metabolism. Am J Phsyiol 277(Endocrin. Metab. 40): E144-E153
Cresenzi CL, Lee JI, Stipanuk MH (2003) Cysteine is the metabolic signal responsible for dietary regulation of hepatic cysteine dioxygenase and glutamate cysteine ligase in intact rats. J Nutr 133(9):2697–2702
Yang BH, Wang X, Ren X (2012) Amino acid metabolism related to immune tolerance by MDSCs. Int Rev Immunol 31(3):177–183
Rodríguez PC, Ochoa AC (2006) T cell dysfunction in cancer: role of myeloid cells and tumor cells regulating amino acid availability and oxidative stress. Semin Cancer Biol 16(1):66–72
Levring TB, Hansen AK, Nielsen BL, Kongsbak M, von Essen MR, Woetmann A, Odum N, Bonefeld CM, Geisler C (2012) Activated human CD4+ T cells express transporters for both cysteine and cystine. Sci Rep 2:266
Srivastava MK, Sinha P, Clements VK, Rodriguez P, Ostrand-Rosenberg S (2010) Myeloid-derived suppressor cells inhibit T-cell activation by depleting cystine and cysteine. Cancer Res 70(1):68–77
Yan Z, Banerjee R (2010) Redox remodeling as an immunoregulatory strategy. Biochemistry 49(6):1059–1066
Sido B, Lasitschka F, Giese T, Gassler N, Funke B, Schröder-Braunstein J, Brunnemer U, Meuer SC, Autschbach F (2008) A prominent role for mucosal cystine/cysteine metabolism in intestinal immunoregulation. Gastroenterology 134(1):179–191
Yan Z, Garg SK, Banerjee R (2010) Regulatory T cells interfere with glutathione metabolism in dendritic cells and T cells. J Biol Chem 285(53):41525–41532
Edinger AL, Thompson CB (2002) Antigen-presenting cells control T cell proliferation by regulating amino acid availability. Proc Natl Acad Sci U S A 99(3):1107–1109
Dröge W, Eck HP, Gmünder H, Mihm S (1991) Dysregulation of plasma amino acid levels in HIV-infection and cancer and its relevance for the immune system. Amino Acids 1(2):193–198
Mihm S, Galter D, Droge W (1995) Modulation of transcription factor NF kappa B activity by intracellular glutathione levels and by variations of the extracellular cysteine supply. FASEB J 9(2):246–252
Dröge W, Eck HP, Gmunder H, Mihm S (1991) Modulation of lymphocyte functions and immune response by cysteine and cysteine derivatives. Am J Med 91(3C):140S–144S
Grimble RF (2006) The effects of sulfur amino acid intake on immune function in humans. J Nutr 136(6 Suppl):1660S–1665S
Murphy G, Fan JH, Mark SD, Dawsey SM, Selhub J, Wang J, Taylor PR, Qiao YL, Abnet CC (2011) Prospective study of serum cysteine levels and oesophageal and gastric cancers in China. Gut 60(5):618–623
Gostner JM, Becker K, Fuchs D, Sucher R (2013) Redox regulation of the immune response. Redox Rep 18(3):88–94
Yan Z, Garg SK, Kipnis J, Banerjee R (2009) Extracellular redox modulation by regulatory T cells. Nat Chem Biol 5(10):721–723
Matsue H, Edelbaum D, Shalhevet D, Mizumoto N, Yang C, Mummert ME et al (2003) Generation and function of reactive oxygen species in dendritic cells during antigen presentation. J Immunol 171(6):3010–3018
Los M, Schenk H, Hexel K, Baeuerle PA, Dröge W, Schulze-Osthoff K (1995) IL-2 gene expression and NF-kappa B activation through CD28 requires reactive oxygen production by 5-lipoxygenase. EMBO J 14(15):3731–3740
Miller TW, Wang EA, Gould S, Stein EV, Kaur S, Lim L, Amarnath S, Fowler DH, Roberts DD (2012) Hydrogen sulfide is an endogenous potentiator of T cell activation. J Biol Chem 287(6):4211–4221
Kontny E, Szczepanska K, Kowalczewski J, Kurowska M, Janicka I, Marcinklewicz J, Maslinski W (2000) The mechanism of taurine chloramine inhibition of cytokine IL-6, IL-8 production by rheumatoid arthritis fibroblast-like synoviocytes. Arthritis Rheum 43(10):2169–2177
Grimble RF (1994) Sulphur amino acids and the metabolic response to cytokines. Adv Exp Med Biol 359:41–49
Huxtable RJ (1996) Taurine past, present and future. Adv Exp Med Biol 403:641–650
Grimble RF (1996) Theory and efficacy of antioxidant therapy. Curr Opin Crit Care 2:260–266
Chorazy M, Kontny E, Marcinklewicz J, Maslinski W (2002) Taurine chloramine modulates cytokine production by human peripheral blood mononuclear cells. Amino Acids 23(4):407–413
Winder B, Lebilhuber F, Frick B, Laich A, Artner-Dworzak E, Fuchs D (2002) Moderate hyperchromocysteinaemia ad immune activation in Parkinson’s disease. J Neural Transm 109(12):1445–1452
Gottfried E, Kreutz M, Mackensen A (2012) Tumor metabolism as modulator of immune response and tumor progression. Semin Cancer Biol 22:335–341
Newsholme P (2001) Why is L-glutamine metabolism important to cells of the immune system in health, postinjury, surgery or infection? J Nutr 131(9 Suppl):2512–2522
Wilmore DW, Shabert JK (1998) The role of glutamine in immunologic responses. Nutrition 14(4):618–626
Hammami I, Chen J, Bronte V, DeCrescenzo G, Jolicoeur M (2012) L-glutamine is a key parameter in the immunosuppression phenomenon. Biochem Biophys Res Commun 425(4):724–729
Raber P, Ochoa AC, Paulo C (2012) Metabolism of L-arginine by myeloid-derived suppressor cells in cancer: mechanisms of T cell suppression and therapeutic perspectives. Immunol Investig 41(6–7):614–634
Barbul A (1990) Arginine and immune function. Nutrition 6(6–7):53–58
Taheri F, Ochoa JB, Faghiri Z, Culotta K, Park HJ, Lan MS et al (2001) L-Arginine regulates the expression of the T-cell receptor zeta chain (CD3zeta) in Jurkat cells. Clin Cancer Res 7(3 Suppl):958s–965s
Rodrigez PC, Zen AH, Culotta KS, Zabaleta J, Ochoa JB, Ochoa AC (2002) Regulation of T cell receptor CD3 zeta chain expression by L-arginine. J Biol Chem 277(24):21123–21129
Albina JE, Caldwell MD, Henry WL Jr, Mills CD (1989) Regulation of macrophage functions by L-arginine. J Exp Med 169(3):1021–1029
Popovic PJ, Zeh HJ 3rd, Ochoa JB (2007) Arginine and immunity. J Nutr 137(6 Suppl 2):1681S–1686S
DeSanctis F, Sandri S, Ferrarini G, Pagliarello I, Sartoris S, Ugel S, Marigo I, Molon B, Bronte V (2014) The emerging immunological role of post-translational modifications by reactive nitrogen species in cancer microenvironment. Front Immunol 5:1–16
Terabe M, Swann J, Ambrosino E et al (2005) A nonclassical non-Valpha14Jalpha18 CD1d-restricted (type II) NKT cells is sufficient for down-regulation of tumor immunosurveillance. J Exp Med 202(12):1627–1633
Takahashi A, Hanson MGV, Norell HR et al (2005) Preferential cell death of CD8+ effector memory (CCR7−CD45RA−) T cells by hudrogen peroxide-induced oxidative stress. J Immunol (Baltimore) 174(10):6080–6087
Schroder AJ, Pavlidis P, Arimura A, Capece D, Rothman PB (2002) Cutting edge: STAT6 serves as a positive and negative regulator of gene expression in IL-4-stimulated B lymphocytes. J Immunol 168(3):996–1000
Godin-Ethier J, Hanafi LA, Piccirillo CA, Lapointe R (2011) Indoleamine 2,3-dioxygenase expression in human cancers: clinical and immunologic perspectives. Clin Cancer Res 17(22):6985–6991
Mellor AL, Munn DH (2001) Tryptophan catabolism prevents maternal T cells from activating lethal anti-fetal immune responses. J Reprod Immunol 52(1–2):5–13
Munn DH, Shafizadeh E, Attwood JT, Bondarev I, Pashine A, Mellor AL (1999) Inhibition of T cell proliferation by macrophage tryptophan catabolism. J Exp Med 189(9):1363–1372
Lee GK, Park HJ, Macleod M, Chandler P, Munn DH, Mellor AL (2002) Tryptophan deprivation sensitizes activated T cells to apoptosis prior to cell division. Immunology 107(4):452–460
Weinlich G, Murr C, Richardsen L, Winkler C, Fuchs D (2007) Decreased serum tryptophan concentration predicts poor prognosis in malignant melanoma patients. Dermatology 214(1):8–14
Brandacher G, Perathoner A, Ladurner R, Schneeberger S, Obrist P, Winkler C, Werner ER, Werner-Felmayer G, Weiss HG, Göbel G, Margreiter R, Königsrainer A, Fuchs D, Amberger A (2006) Prognostic value of indoleamine 2,3-dioxygenase expression in colorectal cancer: effect on tumor-infiltrating T cells. Clin Cancer Res 12(4):1144–1151
Ramanathan A, Wang C, Schreiber SL (2005) Perturbational profiling of a cell-line model of tumorigenesis by using metabolic measurements. Proc Natl Acad Sci U S A 102(17):5992–5997
Elstrom RL, Bauer DE, Buzzai M, Karnauskas R, Harris MH, Plas DR et al (2004) Akt stimulates aerobic glycolysis in cancer cells. Cancer Res 64(11):3892–3899
Opitz CA, Litzenburger UM, Sahm F, Ott M, Tritschler I, Trump S, Schumacher T, Jestaedt L, Schrenk D, Weller M, Jugold M, Guillemin GJ, Miller CL, Lutz C, Radlwimmer B, Lehmann I, von Deimling A, Wick W, Platten M (2011) An endogenous tumour-promoting ligand of the human aryl hydrocarbon receptor. Nature 478(7368):197–203
Pilotte L, Larrieu P, Stroobant V, Colau D, Dolusic E, Frédérick R, De Plaen E, Uyttenhove C, Wouters J, Masereel B, Van den Eynde BJ (2012) Reversal of tumoral immune resistance by inhibition of tryptophan 2,3-dioxygenase. Proc Natl Acad Sci U S A 109(7):2497–2502
Platten M, Wick W, Van den Eynde BJ (2012) Tryptophan catabolism in cancer: beyond IDO and tryptophan depletion. Cancer Res 72(21):5435–5440
Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD (2002) Cancer immunoediting: from immunosurveillence to tumor escape. Nat Immunol 3(11):991–998
Mizoguchi H, O’Shea JJ, Longo DL, Loeffler CM, McVicar DW, Ochoa AC (1992) Alterations in signal transduction molecules in T lymphocytes from tumor-bearing mice. Science 258(5089):1795–1798
Lee J-I, Dominy JE Jr, Sikalidis AK, Hirschberger LL, Wang W, Stipanuk MH (2008) HepG2/C3A cells respond to cysteine-deprivation by induction of the amino acid deprivation/integrated stress response pathway. Physiol Genomics 33(2):218–229
Sikalidis AK, Mazor KM, Kang M, Liu H, Stipanuk MH (2013) Total 4EBP1 is elevated in liver of rats in response to low sulfur amino acid intake. J Amino Acids 2013:864757
Sikalidis AK (2013) Cellular and animal indispensable amino acid limitation responses and health promotion. Can the two be linked? A critical review. Int J Food Sci Nutr 64(3):300–311
Maciolek JA, Pasternak JA, Wilson HL (2014) Metabolism of activated T lymphocytes. Curr Opin Immunol 27:60–74
Baracos VE, Mackenzie ML (2006) Investigations of branched-chain amino acids and their metabolites in animal models of cancer. J Nutr 136(1 Suppl):237S–242S
Conflict of interest
The author declares no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sikalidis, A.K. Amino Acids and Immune Response: A Role for Cysteine, Glutamine, Phenylalanine, Tryptophan and Arginine in T-cell Function and Cancer?. Pathol. Oncol. Res. 21, 9–17 (2015). https://doi.org/10.1007/s12253-014-9860-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9860-0