Abstract
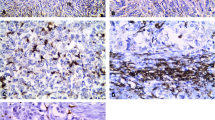
Tumor-associated macrophages (TAMs) have been correlated with increased angiogenesis and poor prognosis in breast cancer. However, the precise role of TAMs in tamoxifen resistance remains unclear. We used immunohistochemical method to examine the expression of epidermal growth factor receptor (EGFR) and CD163+ macrophages in 100 breast cancer tissues. The clinical and biological features of 100 patients were estrogen receptor (ER)-positive and human epidermal growth factor receptor 2(Her-2)-negative tumors. The tamoxifen resistant tissues (n = 48) were the surgical excision samples from patients who developed recurrence or metastasis at the time of adjuvant tamoxifen treatment. The tamoxifen resistant tissues were contrast to tamoxifen sensitive tissues (n = 52). Positive staining for EGFR and CD163+ macrophages were observed in 21 samples (43.8 %) and in 26 samples (54.2 %) respectively in tamoxifen resistance group, which were higher than that of tamoxifen sensitive group (P = 0.001 and P = 0.000279 respectively). Significant positive correlations were found between the expression of EGFR and CD163+ macrophages (r = 0.567, P < 0.01). CD163+ macrophages were positively correlated with tumor size, lymph node metastasis and obesity. Obesity was also related to tamoxifen resistance (P < 0.05). The patients with higher density of CD163+ macrophages infiltration suffered from shorter time to develop recurrence or metastasis (P < 0.05). TAMs may be associated with tamoxifen resistance. Further studies are needed to investigate the potential mechanism between TAMs and tamoxifen resistance.



Similar content being viewed by others
References
Key TJ, Verkasalo PK, Banks E (2001) Epidemiology of breast cancer. Lancet Oncol 2:133–140
Musgrove EA, Sutherland RL (2009) Biological determinants of endocrine resistance in breast cancer. Nat Rev Cancer 9:631–643
Gradishar WJ (2004) Tamoxifen-What next? Oncologist 9:378–384
Ring A, Dowsett M (2004) Mechanisms of tamoxifen resistance. Endocr Relat Cancer 11:643–658
Beelen K, Zwart W, Linn SC (2012) Can predictive biomarkers in breast cancer guide adjuvant endocrine therapy? Nat Rev Clin Oncol 9:529–541
Pollard JW (2008) Macrophages define the invasive microenvironment in breast cancer. J Leukoc Biol 84:623–630
Mantovani A (2005) Cancer: inflammation by remote control. Nature 435:752–753
Balkwill F, Mantovani A (2001) Inflammation and cancer: back to Virchow? Lancet 357:539–545
Sica A, Schioppa T, Mantovani A et al (2006) Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer 42:717–727
Mantovani A, Sica A, Sozzani S et al (2004) The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol 25:677–686
Lau SK, Chu PG, Weiss LM (2004) CD163 A specific marker of macrophages in paraffin-embedded tissue samples. Am J Clin Pathol 122:794–801
Leek RD, Harris AL (2002) Tumor-associated macrophages in breast cancer. J Mammary Gland Biol Neoplasia 7:177–189
Soeda S, Nakamura N, Ozeki T et al (2008) Tumor-associated macrophages correlate with vascular space invasion and myometrial invasion in endometrial carcinoma. Gynecol Oncol 109:122–128
Konecny GE, Santos L, Winterhoff B et al (2008) HER2 gene amplification and EGFR expression in a large cohort of surgically staged patients with nonendometrioid (type II) endometrial cancer. Br J Cancer 100:89–95
García-Becerra R, Santos N, Díaz L et al (2012) Mechanisms of resistance to endocrine therapy in breast cancer: focus on signaling pathways, miRNAs and genetically based resistance. Int J Mol Sci 14:108–145
McKenna NJ, Lanz RB, O’Malley BW (1999) Nuclear receptor coregulators: cellular and molecular biology. Endocr Rev 20:321–344
Frasor J, Danes JM, Komm B et al (2003) Profiling of estrogen up-and down-regulated gene expression in human breast cancer cells: insights into gene networks and pathways underlying estrogenic control of proliferation and cell phenotype. Endocrinol 144:4562–4574
Deroo BJ, Korach KS (2006) Estrogen receptors and human disease. J Clin Invest 116:561–570
Norris JD, Fan D, Kerner SA et al (1997) Identification of a third autonomous activation domain within the human estrogen receptor. Mol Endocrinol 11:747–754
Nicholson RI, Hutcheson IR, Knowlden JM et al (2004) Nonendocrine pathways and endocrine resistance observations with antiestrogens and signal transduction inhibitors in combination. Clin Cancer Res 10:346s–354s
Arpino G, Green SJ, Allred DC et al (2004) HER-2 amplification, HER-1 expression, and tamoxifen response in estrogen receptor-positive metastatic breast cancer a southwest oncology group study. Clin Cancer Res 10:5670–5676
Tang X (2013) Tumor-associated macrophages as potential diagnostic and prognostic biomarkers in breast cancer. Cancer Lett
Volodko N, Reiner A, Rudas M et al (1998) Tumour-associated macrophages in breast cancer and their prognostic correlations. Breast 7:99–105
Mahmoud SMA, Lee AHS, Paish EC et al (2012) Tumour-infiltrating macrophages and clinical outcome in breast cancer. J Clin Pathol 65:159–163
Morris PG, Hudis CA, Giri D et al (2011) Inflammation and increased aromatase expression occur in the breast tissue of obese women with breast cancer. Cancer Prev Res (Phila) 4:1021–1029
Monteiro R, Azevedo I (2010) Chronic inflammation in obesity and the metabolic syndrome. Mediat Inflamm. doi:10.1155/2010/289645
Cinti S, Mitchell G, Barbatelli G et al (2005) Adipocyte death defınes macrophage localization and function in adipose tissue of obese mice and humans. J Lipid Res 46:2347–2355
Zhao Y, Agarwal VR, Mendelson CR et al (1996) Estrogen biosynthesis proximal to a breast tumor is stimulated by PGE2 via cyclic AMP, leading to activation of promoter II of the CYP19 (aromatase) gene. Endocrinology 137:5739–5742
Irahara N, Miyoshi Y, Taguchi T et al (2006) Quantitative analysis of aromatase mRNA expression derived from various promoters (I.4, I.3, PII and I.7) and its association with expression of TNF-alpha, IL-6 and COX-2 mRNAs in human breast cancer. Int J Cancer 118:1915–1921
Bachelot T, Ray-Coquard I, Menetrier-Caux C et al (2003) Prognostic value of serum levels of interleukin 6 and of serum and plasma levels of vascular endothelial growth factor in hormone-refractory metastatic breast cancer patients. Br J Cancer 88:1721–1726
Dandona P, Weinstock R, Thusu K et al (1998) Tumor necrosis factor-alpha in sera of obese patients: fall with weight loss. J Clin Endocrinol Metab 83:2907–2910
Leek RD, Hunt NC, Landers RJ et al (2000) Macrophage infiltration is associated with VEGF and EGFR expression in breast cancer. J Pathol 190:430–436
Yang J, Liao D, Chen C et al (2013) Tumor associated macrophages regulate murine breast cancer stem cells through a novel paracrine EGFR/Stat3/Sox-2 signaling pathway. Stem Cells 31:248–258
Vlaicu P, Mertins P, Thomas M et al (2013) Monocytes/macrophages support mammary tumor invasivity by co-secreting lineage-specific EGFR ligands and a STAT3 activator. BMC Cancer 13:197
Condeelis J, Segall JE (2003) Intravital imaging of cell movement in tumours. Nat Rev Cancer 3:921–930
Acknowledgments
This experiment was finished in the oncology key lab of Heilongjiang province institution of higher Education. This study was supported by the National Natural Science Foundation of China (Grant No. 81372839).
Conflict of Interest
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xuan, Qj., Wang, Jx., Nanding, A. et al. Tumor-Associated Macrophages are Correlated with Tamoxifen Resistance in the Postmenopausal Breast Cancer Patients. Pathol. Oncol. Res. 20, 619–624 (2014). https://doi.org/10.1007/s12253-013-9740-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-013-9740-z