Abstract
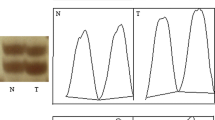
Cervical cancer is one of the most common malignant diseases affecting women worldwide. Studies on loss of heterozygosity have been made for PTEN gene specific microsatellite markers in malignancies like breast, ovary and lungs and the results have shown a significant association. However the role of this gene is not clearly understood in cervical cancer from Indian population. A total of 135 cervical carcinoma tissues samples were analyzed for loss of heterozygosity. DNA was isolated from the samples and their matched control specimens. Polymerase chain reaction was performed using primer specific for two intragenic markers (D10S198 & D10S192) and one marker (D10S541) in flanking region and further electrophoresed on 8% denaturing polyacrylamide gel. Overall, 31 out of 133(23%) informative cases showed loss of heterozygosity in at least one locus in the region examined. The percentage of loss of heterozygosity for these markers ranged from 8% (D10S192) to 13% (D10S198). Loss of heterozygosity was more frequently detected in intragenic region (D10S198 & D10S192) than in flanking region, D10S541 (21% versus 9%). These data argue that PTEN is a tumor suppressor gene whose inactivation may play an important role in the carcinoma of uterine cervix.


Similar content being viewed by others
References
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100:57–70
Lin JC, Scherer SW, Tougas L, Traverso G, Tsui LC, Andrulis IL, Jothy S, Park M (1996) Detailed deletion mapping with a refined physical map of 7q31 localizes a putative tumor suppressor gene for breast cancer in the region of MET. Oncogene 13:2001–2008
Knudson AG (1993) Antioncogenes and human cancer. Proc Natl Acad Sci USA 90:10914–10921
Marshall CJ (1991) Tumor suppressor genes. Cell 64:313–326
Goddard AD, Solomon E (1993) Genetic aspects of cancer. In: Harris CC, Hirschorn K (eds) Advances in human genetics. Plenum Press, New York 21: 321–376.
Siebert R, Gesk S, Harder S, Plötz S, Matthiesen P, Grote W, Schlegelberger B, Jandrig B, Grasmo-Wendler U, Scherneck S, Rosenwald A, Ott G (1998) Deletions in the long arm of chromosome 10 in lymphomas with t(14;18): A Pathogenetic Role of the Tumor Suppressor Genes PTEN/MMAC1 and MXI1? Blood 92(11):4487–4489
Steck PA, Lin H, Langford LA, Jasser SA, Koul D, Yung WK, Pershouse MA (1999) Functional and molecular analyses of 10q deletions in human gliomas. Genes Chromosomes Cancer 24(2):135–143
Matsuyama H, Pan Y, Yoshihiro S, Kudren D, Naito K, Bergerheim USR, Ekman P (2003) Clinical significance of chromosome 8p, 10q, and 16q deletions in prostate cancer. Prostate 54(2):103–111
Li YL, Tian Z, Wu DY, Fu BY, Xin Y (2005) Loss of heterozygosity on 10q23.3 and mutation of tumor suppressor gene PTEN in gastric cancer and precancerous lesions. World J Gastroenterology 11(2):285–288
Sankaranaryanan R, Buduk AM, Rajkumar R (2001) Effective Screening Programmes for Cervical Cancer in low- and middle-income developing countries. Bull World Health Organ 79:954–962
Ferlay J. GLOBOCAN 2002. Cancer incidence, mortality and prevalence worldwide. IARC 2004; CancerBase No.5, Version 2.0. IARC Press, Lyon.
NATIONAL CANCER REGISTRY PROGRAMME (2007) Consolidated report of hospital based cancer registries 2001–2003. ICMR, New Delhi
American Cancer Society. Facts & Figures 2009.
Mitra AB, Murty VVVS, Li RG, Pratap M, Luthra UK, Chaganti RSK (1994) Allelotype analysis of cervical carcinoma. Cancer Res 54:4481–4487
Jones MH, Koi S, Fujimoto I et al (1994) Allelotype of uterine cancer by analysis of RFLP and microsatellite polymorphisms: frequent loss of heterozygosity on chromosome arms 3p, 9q, 10q, and 17p. Genes Chromosomes Cancer 9:119–123
Miayani K, Furugen Y, Matsumoto T, Iwabuchi K, Hirose S, Kinoshita K, Fujii H (2004) Loss of heterozygosity analysis in uterine cervical adenocarcinomas. Gynecol Oncol 94:115–120
Di Cristofano A, Pesce B, Cordon-Cardo C, Pandolfi PP (1998) PTEN is essential for embryonic development and tumour suppression. Nat Genet 19:348–355
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI, Puc J, Miliaresis C, Rodgers L, McCombie R, Bigner SH, Giovanella BC, Ittmann M, Tycko B, Hibshoosh H, Wigler MH, Parsons R (1997) PTEN: a putative protein tyrosine phosphatase gene mutated in human brain, breast and prostate cancer. Science 275:1943–1947
Stambolic V, Suzuki A, de la Pompa JL, Brothers GM, Mirtsos C, Sasaki T, Ruland J (1998) Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell 95:29–39
Reid AHM, Attard G, Ambroisine L, Fisher G, Kovacs G, Brewer D, Clark J, Flohr P, Edwards S, Berney DM, Foster CS, Fletcher A, Gerald WL, Møller H, Reuter VE, Scardino PT, Cuzick J, de Bono JS, Cooper CS (2010) Molecular characterisation of ERG, ETV1 and PTEN gene loci identifies patients at low and high risk of death from prostate cancer. Br J Cancer 102:678–684
Izycka-Swieszewska E, Brzeskwiniewicz M, Wozniak A, Drozynska E, Grajkowska W, Perek D, Balcerska A, Klepacka T, Limon J (2010) EGFR, PIK3CA and PTEN gene status and their protein product expression in neuroblastic tumours. Folia Neuropathol 48(4):238–245
Aneta JJ, Bożena K, Cyprian G, Urszula N (2010) TP53 mutations in endometrial cancers: relation to PTEN gene defects. Int J Gynecol Cancer 20(2):196–202
Shabbir Alam M, Rizvi MMA (2011) Cancer of the uterine cervix and PTEN tumor suppressor gene. Lambert Academic Publishing, Germany
Kurose K, Zhou XP, Araki T, Eng C (2000) Biallelic inactivating mutations and an occult germline mutation of PTEN in primary cervical carcinomas. Genes Chromosomes Cancer 29:166–172
Benedet JL, Bender H, Jones H III, Ngan HYS, Pecorelli S (2000) FIGO staging classifications and clinical practice guidelines in the management of gynecologic cancers. Int J Gynecol Obstet 70:209–262
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Lab Press, Plainview
Yuping X, Lili M, Chengbo H, Jinyi L, Lei X (2007) MSI/LOH and extron expression of the FHIT gene in gastric carcinoma. Front Med China 1(1):99–103
Preacher KJ (2001) Calculation for the chi-square test: An interactive calculation tool for chi-square tests of goodness of fit and independence [Computer software]. Available from http://www.quantpsy.org.
Hansen MF, Cavenee WK (1987) Genetics of cancer predeposition. Cancer Res 47:5518–5527
Kim S, Domon-Dell C, Wang Q, Chung DH, Di Cristofano A, Pandolfi PP, Freund JN, Evers BM (2002) PTEN and TNF-alpha regulation of the intestinal-specific Cdx-2 homeobox gene through a PI3K, PKB/Akt, and NF-kappaB-dependent pathway. Gastroenterology 123:1163–1178
McConnachie G, Pass I, Walker SM, Downes CP (2003) Interfacial kinetic analysis of the tumour suppressor phosphatase, PTEN: evidence for activation by anionic phospholipids. Biochem J 371(3):947–955
Brown KS, Blair D, Reid SD, Nicholson EK, Harnett MM (2004) FcgammaRIIb-mediated negative regulation of BCR signaling is associated with the recruitment of the MAPKinasephosphatase, Pac-1, and the 3′-inositol phosphatase, PTEN. Cell Signal 16:71–80
Cho SH, Lee CH, Ahn Y, Kim H, Kim H, Ahn CY, Yang KS, Lee SR (2004) Redox regulation of PTEN and protein tyrosine phosphatases in H(2)O(2) mediated cell signaling. FEBS Lett 560:7–13
Wishart MJ, Dixon JE (2002) PTEN and myotubularin phosphatases: from 3-phosphoinositide dephosphorylation to disease. Trends Cell Biol 12:579–585
Mills GB, Kohn E, Lu Y, Eder A, Fang X, Wang H, Bast RC, Gray J, Jaffe R, Hortobagyi G (2003) Linking molecular diagnostics to molecular therapeutics: targeting the PI3K pathway in breast cancer. Semin Oncol 30(5 Suppl 16):93–104
Orchiston EA, Bennett D, Leslie NR, Clarke RG, Winward L, Downes CP, Safrany ST (2004) PTEN M-CBR3, a versatile and selective regulator of inositol 1,3,4,5,6-pentakisphosphate (Ins (1,3, 4,5,6) P5). Evidence for Ins (1,3,4,5,6)P5 as a proliferative signal. J Biol Chem 279:1116–1122
Zhao H, Dupont J, Yakar S, Karas M, LeRoith D (2004) PTEN inhibits cell proliferation and induces apoptosis by downregulating cell surface IGF-IR expression in prostate cancer cells. Oncogene 23:786–794
Konopka B, Janiec-Jankowska A, Paszko Z, Goluda M (2004) The coexistence of ERBB2, INT2, and CMYC oncogene amplifications and PTEN gene mutations in endometrial carcinoma. J Cancer Res Clin Oncol 130:114–121
Acknowledgement
The financial assistance provided by the University Grants Commision (UGC), New Delhi, is gratefully acknowledged. The assistance of M. Irshad is acknowledged with thanks.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rizvi, M.M.A., Alam, M.S., Mehdi, S.J. et al. Allelic Loss of 10q23.3, the PTEN Gene Locus in Cervical Carcinoma from Northern Indian Population. Pathol. Oncol. Res. 18, 309–313 (2012). https://doi.org/10.1007/s12253-011-9446-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-011-9446-z