Abstract
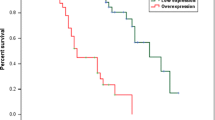
Phosphatase of regenerating liver-3 (PRL-3) is overexpressed in several human cancers and associated with tumor progression, invasion and metastasis. However, the correlation between PRL-3 expression and clinical outcome in ovarian cancer has not been studied. In the present study, we investigated the expression of PRL-3 in 119 ovarian cancers and 30 normal ovarian tissues by immunohistochemistry with an anti-PRL-3 mouse monoclonal antibody 3B6, and analyzed its relationship with clinicopathologic factors and survival. The results demonstrated that PRL-3 expression was significantly higher in ovarian cancers compared to normal ovarian tissues (P < 0.001). PRL-3 expression is not correlated with patient age, menstruation, tumor size, histological type, residual tumor, or other clinical findings. The patients with PRL-3-positive tumors had a significant poor prognosis than those with PRL-3-negative tumors. Univariate analysis identified PRL-3 expression as a poor outcome predictor (HR 1.925, 95% CI, 1.046–3.544, P = 0.035). Multivariate analysis indicated that PRL-3 expression was an independent prognostic factor of borderline significance (HR 1.695, 95% CI, 0.914–3.145, P = 0.094). Our results suggest that PRL-3 may serve as a valuable marker for diagnosis of ovarian cancer and as a potential independent prognostic factor for ovarian cancer.


Similar content being viewed by others
Abbreviations
- PRL-3:
-
phosphatase of regenerating liver-3
- OS:
-
overall survival
- HR:
-
hazard ratio
- TP:
-
TP based chemotherapy (combination of taxol and cisplatin)
- CAP:
-
CAP based chemotherapy (combination of cyclophosphamide, cisplatin and adriamycin)
References
Cannistra SA (2004) Cancer of the ovary. N Engl J Med 351:2519–2529
Zurawski VR Jr, Orjaseter H, Andersen A et al (1988) Elevated serum CA 125 levels prior to diagnosis of ovarian neoplasia: relevance for early detection of ovarian cancer. Int J Cancer 42:677–680
Mastropaolo W, Fernandez Z, Miller EL (1986) Pronounced increases in the concentration of an ovarian tumor marker, CA-125, in serum of a healthy subject during menstruation. Clin Chem 32:2110–2111
Takahashi K, Shibukawa T, Moriyama M (1986) Clinical usefulness and false-positive results of CA 125 as a tumor marker of ovarian cancer-a study on 674 patients. Jpn J Surg 16:305–310
Lyon M, Ducruet A, Wipf P, et al (2002) Dual-specificity phosphates as targets for antineoplastic agents. Nat Rev Drug Discov 1:961–976
Zeng Q, Hong W, Tan YH (1998) Mouse PRL-2 and PRL-3, two potentially prenylated protein tyrosine phosphatases homologous to PRL-1. Biochem Biophys Res Commun 244:421–427
Diamond RH, Cressman DE, Laz TM, et al (1994) PRL-1, a unique nuclear protein tyrosine phosphatase, affects cell growth. Mol Cell Biol 14:3752–3762
Werner SR, Lee PA, DeCamp MW, et al (2003) Enhanced cell cycle progression and down regulation of p21(Cip1/Waf1) by PRL tyrosine phosphatases. Cancer Lett 202:201–211
Polato F, Codegoni A, Fruscio R, et al (2005) PRL-3 phosphatase is implicated in ovarian cancer growth. Clin Cancer Res 11:6835–6839
Zeng Q, Dong JM, Guo K, et al (2003) PRL-3 and PRL-1 promote cell migration, invasion, and metastasis. Cancer Res 63:2716–2722
Wu X, Zeng H, Zhang X (2004) Phosphatase of regenerating liver-3 promotes motility and metastasis of mouse melanoma cells. Am J Pathol 23:23–27
Guo K, Li J, Tang JP, et al (2004) Catalytic domain of PRL-3 plays an essential role in tumor metastasis: formation of PRL-3 tumors inside the blood vessels. Cancer Biol Ther 3:945–951
Li J, Guo K, Koh VW, et al (2005) Generation of PRL-3- and PRL-1-specific monoclonal antibodies as potential diagnostic markers for cancer metastases. Clin Cancer Res 11:2195–2204
Guo K, Li J, Tang JP, et al (2008) Monoclonal antibodies target intracellular PRL phosphatases to inhibit cancer metastases in mice. Cancer Biol Ther 7
Peng L, Li Y, Meng L, et al (2004) Preparation and characterization of monoclonal antibody against protein tyrosine phosphatase PRL-3. Hybrid Hybridomics 23:23–27
Peng L, Ning J, Meng L, et al (2004b) The association of the expression level of protein tyrosine phosphatase PRL-3 protein with liver metastasis and prognosis of patients with colorectal cancer. J Cancer Res Clin Oncol 130:521–526
Wang L, Peng L, Dong B, et al (2006) Overexpression of phosphatase of regenerating liver-3 in breast cancer: association with a poor clinical outcome. Ann Oncol 17:1517–1522
Wang Y, Li ZF, He J, et al (2007) Expression of the human phosphatases of regenerating liver (PRLs) in colonic adenocarcinoma and its correlation with lymph node metastasis. Int J Colorectal Dis 22:1179–1184
Miskad UA, Semba S, Kato H, Yokozaki H (2004) Expression of PRL-3 phosphatase in human gastric carcinomas: close correlation with invasion and metastasis. Pathobiology 71:176–184
Miskad UA, Semba S, Kato H, et al (2007) High PRL-3 expression in human gastric cancer is a marker of metastasis and grades of malignancies: an in situ hybridization study. Virchows Arch 450:303–310
Stephens B, Han H, Gokhale V, et al (2005) PRL phosphatases as potential molecular targets in cancer. Mol Cancer Ther 4:1653–1661
Han H, Bearss DJ, Browne LW, et al (2002) Identification of differentially expressed genes in pancreatic cancer cells using cDNA microarray. Cancer Res 62:2890–2896
Stephens B, Han H, Hostetter G, et al (2008) Small interfering RNA-mediated knockdown of PRL phosphatases results in altered Akt phosphorylation and reduced clonogenicity of pancreatic cancer cells. Mol Cancer Ther 7:202–210
Kato H, Semba S, Miskad UA, et al (2004) High expression of PRL-3 promotes cancer cell motility and liver metastasis in human colorectal cancer: a predictive molecular marker of metachronous liver and lung metastases. Clin Cancer Res 10:7318–7328
Radke I, Gotte M, Kersting C, et al (2006) Expression and prognostic impact of the protein tyrosine phosphatases PRL-1, PRL-2, and PRL-3 in breast cancer. Br J Cancer 95:347–354
Acknowledgements
This work was supported by National 973 Program (2009CB521805), National Natural Sciences Foundation of China (30671024) and Natural Science Foundation of Beijing (5072015). We thank Dr. Like Qu for critical reading of the manuscript, Dr. Jiyou Li for histopathological diagnosis and Dr. Yunong Gao for providing the clinical data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tingting Ren, Beihai Jiang, Both authors contributed equally to this work
Rights and permissions
About this article
Cite this article
Ren, T., Jiang, B., Xing, X. et al. Prognostic Significance of Phosphatase of Regenerating Liver-3 Expression in Ovarian Cancer. Pathol. Oncol. Res. 15, 555–560 (2009). https://doi.org/10.1007/s12253-009-9153-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-009-9153-1