Abstract
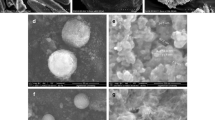
An amalgamation of solid dispersion and capillarity has been attempted in present study for enhancement of dissolution rate of poorly soluble drugs. Flash evaporation technique was utilized for enhancement of the dissolution rate of piroxicam. One of the major problems with this drug is its very low solubility in biological fluids, which results in poor bioavailability after oral administration. An attempt was made to enhance the dissolution rate of piroxicam by converting it into porous solid dispersion by flash evaporation method using polyvinylpyrrolidone (PVP) 40,000 as a water-soluble carrier. The resulting solid dispersions were characterized by DSC, FTIR, and X-ray diffraction. In vitro dissolution study revealed significant improvement of dissolution profile of piroxicam. The release of drug from porous solid dispersions containing PVP was superior to those of marketed product, conventional nonporous solid dispersion prepared by solvent evaporation method and drug alone. The steep increase in dissolution rate of porous form is attributable to combined effect of solid dispersion and capillarity.





Similar content being viewed by others
References
Myrdal PB, Yalkowsky SH. Solubilization of drugs in aqueous media. In: Swarbrick J, Boylan JC, editors. Encyclopedia of Pharmaceutical Technology. New York: Marcel Dekker; 2002. p. 2458–80.
Chiou WL, Riegalman S. Pharmaceutical applications of solid dispersion systems. J Pharm Sci. 1971;60(9):1281–302.
Serajuddin ATM. Solid dispersion of poorly water-soluble drugs: early promises, subsequent problems and recent breakthroughs. J Pharm Sci. 1999;88(10):1058–66.
Leuner C, Dressman J. Improving drug solubility for oral delivery using solid dispersions. Eur J Pharm Biopharm. 2000;50:47–60.
Borne RF. Non-steroidal anti-inflammatory drugs. In: Foye WO, Lembe TL, Williams DA, editors. Principles of medicinal chemistry. New Delhi: Waverly Private Limited; 1999. p. 567–9.
Martindale. Aspirin and similar analgesics and anti-inflammatory agents. In: Reynolds JEF, Prasad AB, editors. The Extra Pharmacopoeia. London: The Pharmaceutical Press Publication; 1989. pp. 31–32.
Amidon GL, Lennernas H, Shah VP, Crison JR. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res. 1995;12:413–20.
Clark DE. Rapid calculation of polar molecular surface area and its application to the prediction of transport phenomena.1.Prediction of intestinal absorption. J Pharm Sci. 1999;88:807–14.
Lebenberg R, Amidon GL. Modern bioavailability, bioequivalence and biopharmaceutics classification system. New approaches to international regulatory standards. Eur J Pharm Biopharm. 2000;50:3–12.
Roberts LJ, Morrow JD. Analgesic–antipyretic and anti-inflammatory agents and drugs employed in treatment of gout. In: Hardman JG, Limbird LE, Gilman AG, editors. Goodman and Gilman’s, The Pharmacological Basis of Therapeutics. New York: Mc Graw Hill; 1992. p. 713–4.
Patel DM, Shah RP, Jogani PD. Tablet formulation of piroxicam containing PVP K-30 and sodium lauryl sulphate. Indian J Pharm Sci. 2004;66(1):49–55.
United States Pharmacopoeia XXVI, National Formulary XXI. Asian Edn. Rockville, MD: US Pharmacopoeial Convention; 2003. pp. 2227–2229.
United States Pharmacopoeia XXV, National Formulary XX. Asian Edn. Rockville, MD: US Pharmacopoeial Convention; 2002. pp. 1388–1389.
Krile B, Caplar V, Kajfez F. Blazevic N. In: Florey K, editor. Analytical profiles of drug substances and excipients. New York: Academic; 1986. p. 509–31.
Tantishaiyakul V, Kaewnopparat N, Ingkatawornwong S. Properties of solid dispersions of piroxicam in polyvinylpyrrolidone. Int J Pharm. 1999;181:143–51.
Ingkatawornwong S, Kaewnopparat N, Tantishaiyakul V. Studies on aging piroxicam polyvinylpyrrolidone solid dispersions. Pharmazie. 2001;56(3):227–30.
Acknowledgments
Thanks are due to Bangia Pharmaceuticals, Karnal (Haryana), India, for providing gift sample of piroxicam. Thanks are also due to Jubilant Organosys Ltd., Noida, Uttar Pradesh, India, for providing facilities to carry out DSC, FTIR, and XRD studies.
Declaration of Interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dhall, M., Nanda, S. & Madan, A.K. Studies on Flash Evaporation for Preparation of Porous Solid Dispersions Using Piroxicam as a Model Drug. J Pharm Innov 6, 232–240 (2011). https://doi.org/10.1007/s12247-011-9116-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12247-011-9116-4