Abstract
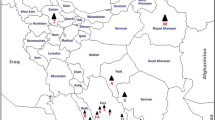
During 2010–14 surveys in the major sesame growing areas of Fars, Yazd and Isfahan provinces (Iran), genetic diversity and vector transmission of phytoplasmas associated with sesame phyllody were studied. Virtual RFLP, phylogenetic, and DNA homology analyses of partial 16S ribosomal sequences of phytoplasma strains associated with symptomatic plants revealed the presence of phytoplasmas referable to three ribosomal subgroups, 16SrII-D, 16SrVI-A, and 16SrIX-C. The same analyses using 16S rDNA sequences from sesame phyllody-associated phytoplasmas retrieved from GenBank database showed the presence of phytoplasmas clustering with strains in the same subgroups in other Iranian provinces including Bushehr and Khorasan Razavi. Circulifer haematoceps and Orosius albicinctus, known vectors of the disease in Iran, were tested for transmission of the strains identified in this study. C. haematoceps transmitted 16SrII-D, 16SrVI-A, and 16SrIX-C phytoplasmas, while O. albicinctus only transmitted 16SrII-D strains. Based on the results of the present study and considering the reported presence of phytoplasmas belonging to the same ribosomal subgroups in other crops, sesame fields probably play an important role in the epidemiology of other diseases associated with these phytoplasmas in Iran.





Similar content being viewed by others
References
Akhtar KP, Dickinson M, Sarwar G, Jamil FF, Haq MA (2008) First report on the association of a 16SrII phytoplasma with sesame phyllody in Pakistan. Plant Pathol 57:771
Akhtar KP, Sarwar G, Dickinson M, Ahmad M, Haq MA, Hameed S, Iqbal J (2009) Sesame phyllody disease: symptomatology, etiology and transmission in Pakistan. Turk J Agric For 5:477–486
Al-Sakeiti MA, Al-Subhi AM, Al-Saady NA, Deadman ML (2005) First report of witches’ broom disease of sesame (Sesamum indicum) in Oman. Plant Dis 89:530
Ashri A (2007) Sesame (Sesamum indicum L.). In: Singh RJ (ed) Genetics resources, chromosome engineering, and crop improvement, Vol.4, oilseed crops. CRC Press, Boca Raton
Bertaccini A, Duduk B (2009) Phytoplasma and phytoplasma diseases: a review of recent research. Phytopath Medit 48:355–378
Bertaccini A, Duduk B, Paltrinieri S, Contaldo N (2014) Phytoplasmas and phytoplasma diseases: a severe threat to agriculture. Am J Pl Sci 5:1763–1788
Catal M, Ikten C, Yol E, Ustun R, Uzun B (2013) First report of 16SrIX group phytoplasma associated with sesame phyllody in Turkey. Plant Dis 97:835
Choopanya D (1973) Mycoplasma like bodies associated with sesamum phyllody in Thailand. Phytopathology 63:1536–1537
Dehghan A, Salehi M, Khajazadeh Y, Taghizadeh M (2009) Studies on sesame phyllody and effect of sowing date and insecticide spray on disease control in Khuzestan. Appl Entomol Phytopath 77:23–36
Deng SJ, Hiruki C (1991) Amplification of 16S ribosomal- RNA genes from culturable and non culturable mollicutes. J Microbiol Meth 14:53–61
Desmits M, Laboucheix J (1974) Relationship between cotton phyllody and a similar disease of sesame. F.A.O. Plant Prot Bull 22:19–20
Doyle JJ, Doyle JI (1990) Isolation of DNA from fresh plant tissue. Focus 12:13–15
Esmailzadeh Hosseini SA, Khodakaramian G, Salehi M, Fani SR, Mirchenari SR, Elham S (2015a) Incidence, distribution and economic importance of alfalfa witches’ broom disease in Sistan-Baluchestan (Iran) and characterization of associated phytoplasma. Phytopath Moll 5:1–7
Esmailzadeh Hosseini SA, Salehi M, Khodakaramian G, Bolok Yazdi HR, Salehi M, Jafari Nodooshan A, Jadidi O, Bertaccini A (2015b) Status of sesame phyllody and its control methods in Yazd, Iran. Phytopath Moll 5(1-Suppl):S119–S120
Esmailzadeh Hosseini SA, Salehi M, Mirchenari SM, Tarizeh D, Gholampoor H (2015c) First report of a 16SrVI group related phytoplasma associated with cucumber phyllody in a greenhouse in Iran. New Dis Rep 32:5
Esmailzadeh Hosseini SA, Mirzaie A, Jafari-Nodooshan A, Rahimian H (2007) The first report of transmission of a phytoplasma associated with sesame phyllody by Orosius albicinctus in Iran. Austral Plant Dis Notes 2:33–34
Faostat (2013) Food and agriculture organization corporate statistical database. [online] URL: http://faostat3.fao.org
Gundersen DE, Lee I-M (1996) Ultrasensitive detection of phytoplasmas by nested-PCR assays using two universal primer pairs. Phytopath Medit 35:144–151
Hue, YH (1996) Bailey’s industrial oil and products. A Wiley Interscience Publication. 457 pp.
Ibrahimi, A, Minasian, V (1975) A list of diseases of cultivated and wild plants of Khusestan. Gondishapoor University 28 pp.
Ikten C, Catal M, Yol E, Ustun R, Furat S, Toker C, Uzun B (2014) Molecular identification, characterization and transmission of phytoplasmas associated with sesame phyllody in Turkey. Eur J Plant Pathol 139:217–229
Jamshidi E, Jafarpour B, Rouhani H, Salehi M (2014) Association of members of clover proliferation (16SrVI) and pigeon pea witches’ broom (16SrIX) phytoplasma groups with tomato big bud disease in Iran. Iranian. J Plant Pathol 50:77–89
Kersting U (1993) Symptomatology, etiology and transmission of sesame phyllody in Turkey. J Turk Phytopathol 22:47–54
Khan AJ, Bottner K, Al-Saadi N, Al-Subhi AM, Lee I-M (2007a) Identification of phytoplasma associated with witches’ broom and virescence diseases of sesame in Oman. Bull Insectol 60:133–134
Khan MS, Raj SK, Snehi SK (2007b) First report of molecular identification of ‘Candidatus Phytoplasma asteris’ affecting sesame (Sesamum indicum) cultivation in India. J Pl Pathol 89:301–305
Klein M (1977) Sesame phyllody in Israel. Phytopathology 88:165–171
Kolte, SJ 1985. Diseases of annual edible oil seed crops. Vol. II. CRS Press 135 pp
Lee I-M, Gundersen-Rindal DE, Davis RE, Bartoszyk IM (1998) Revised classification scheme of phytoplasmas based on RFLP analyses of 16SrRNA and ribosomal protein gene sequences. Int J Syst Bacteriol 48:1153–1169
Lee I-M, Davis RE, Gundersen-Rindal DE (2000) Phytoplasma: phytopathogenic mollicutes. Annu Rev Microbiol 54:221–255
Lee I-M, Gundersen-Rindal D, Davis RE, Bottner KD, Marcone C, Seemüller E (2004) Candidatus Phytoplasma asteris’, a novel taxon associated with aster yellows and related diseases. Int J Syst Bacteriol 54:1037–1048
Lee I-M, Bottner KD, Zhao Y, Davis RE, Harrison NA (2010) Phylogenetic analysis and delineation of phytoplasmas based on SecY gene sequences. Int J Syst Evol Microbiol 60:2887–2897
Lee I-M, Bottner-Parker KD, Zhao Y, Bertaccini A, Davis RE (2012) Differentiation and classification of phytoplasmas in the pigeon pea witches’-broom group (16SrIX): an update based on multiple gene sequence analysis. Int J Syst Evol Microbiol 62:2279–2285
Madhupriya, Rao GP, Kumar A, Baranwal VK (2015) Classification of sesame phytoplasma strain in India at 16Sr subgroup level. J Plant Pathol 3:523–528
Manjunatha N, Prameela HA, Rangaswamy KT, Palanna KB, Wickramaaracgchi WART (2012) Phyllody phytoplasma infecting sesame in South India. Phytopath Moll 2:29–32
McGibbon, TD (1924) Annual report of the Economic Botanist, Burma for the year ending 30th June, 5 pp.
Mostafavi M (1970) Green flowering of sesamum. Iran J Plant Path 5:36–37
Motamedi E, Salehi M, Sabbagh SK, Salari M (2016) Differentiation and classification of phytoplasmas associated with almond and GF-677 witches’ broom diseases using rRNA and rp genes. J Phytopathol 164:185–192
Nabi S, Madhupriya, Dubey DK, Rao GP, Baranwal VK, Sharma P (2015a) Characterization of phytoplasmas associated with sesame (Sesamum indicum) phyllody disease in North India utilizing multilocus genes and RFLP analysis. Indian Phytopathol 68:112–119
Nabi S, Madhupriya, Kumar Dubey D, Rao GP, Baranwal VK, Sharma P (2015b) Molecular characterization of ‘Candidatus Phytoplasma asteris’ subgroup I-B associated with sesame phyllody disease and identification of its natural vector and weed reservoir in India. Austral Plant Pathol 44:289–247
Nakashima K, Hayashi T, Chaleeprom W, Wongkaew P, Sirithorn P (1995) Detection of DNA of phytoplasmas associated with phyllody disease of sesame in Thailand. Ann Phytopathol Soc Jpn 61:519–528
Rao GP, Nabi SU, Madhupriya (2015) Overview on a century progress of research on sesame phyllody disease. Phytopath Moll 5:74–83
Salehi M, Izadpanah K (1992) Etiology and transmission of sesame phylloy in Iran. J Phytopathol 135:37–47
Salehi M, Heydarnejad J, Izadpanah K (2005) Molecular characterization and grouping of 35 phytoplasmas from central and southern provinces of Iran. Iran J Plant Pathol 41:62–64
Salehi M, Izadpanah K, Heydarnejad J (2006) Characterization of a new almond witches’ broom phytoplasma in Iran. J Phytopathol 154:386–391
Salehi M, Izadpanah K, Nejat N, Siampour M (2007a) Partial characterization of phytoplasmas associated with lettuce and wild lettuce phyllodies in Iran. Plant Pathol 65:669–670
Salehi M, Izadpanah K, Siampour M (2007b) Characterization of a phytoplasma associated with cabbage yellows in Iran. Plant Dis 91:625–630
Salehi M, Izadpanah K, Siampour M, Firuz R, Salehi E (2009) Molecular characterization and transmission of safflower phyllody phytoplasma in Iran. J Plant Pathol 91:453–458
Salehi M, Izadpanah K, Siampour M (2011) Occurrence, molecular characterization and vector transmission of a phytoplasma associated with rapeseed phyllody in Iran. J Phytopathol 159:100–105
Salehi E, Salehi M, Taghavi SM, Izadpanah K (2014) A 16SrII-D phytoplasma strain associated with tomato witches’ broom in Bushehr province, Iran. J Crop Protect 3:377–388
Salehi M, Esmailzadeh SA, Salehi E (2015a) Characterisation of a phytoplasma associated with sunflower phyllody in Fars, Isfahan and Yazd provinces of Iran. New Dis Repr 31:6
Salehi M, Siampour M, Esmailzadeh Hosseini SA, Bertaccini A (2015b) Characterization and vector identification of phytoplasmas associated with cucumber and squash phyllody in Iran. Bull Insectol 68:311–319
Schneider B, Seemueller E, Smart CD, Kirkpatrick BC (1995) Phylogenetic classification of plant pathogenic mycoplasma-like organisms or phytoplasmas. In: Razin S, Tully JG (eds) In: molecular and diagnostic procedures in mycoplasmology, vol Vol. 1. Academic Press, San Diego, pp. 369–380
Seemüller E, Marcone C, Lauer U, Ragozzino A, Göschl M (1998) Current status of molecular classification of the phytoplasmas. J Plant Pathol 80:3–26
Sertkaya G, Martini M, Musetti R, Osler R (2007) Detection and molecular characterization of phytoplasmas infecting sesame and solanaceous crops in Turkey. Bull Insectol 60:141–142
Tamimi KM, Fattah FA, Al-Hamdany MA (1989) Shoot apex fasciation in Sesamum indicum associated with mycoplasma-like organisms. Plant Pathol 38:300–304
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Tohidi Z, Salehi M, Ghasemi S, Khanchezar A, Shahamiri M (2015) Association of a 16SrIX-C phytoplasma with eggplant phyllody in Iran. J Crop protect 4:247–256
Tseng YW, Deng WL, Chang CJ, Huang JW, Jan FJ (2014) First report on the association of a 16SrII-A phytoplasma with sesame (Sesamum indicum) exhibiting abnormal stem curling and phyllody in Taiwan. Plant Dis 98:990
Weiss EA (2000) Oilseed crops, 2nd edn. Blackwell Science, Oxford 131-164 pp
Win NKK, Back C, Jung HY (2010) Phyllody phytoplasma infecting sesame in Myanmar belongs to group 16SrI and subgroup 16SrI-B. Trop Pl Pathol 35:310–313
Zhang YP, Uyemoto JK, Kirkpatrick BC (1998) A small-scale procedure for extracting nucleic acids from woody plants infected with various phytoplasmas for PCR assay. J Virol Methods 71:45–50
Zhao Y, Wei W, Lee I-M, Shao J, Suo X, Davis RE (2009) Construction of an interactive online phytoplasma classification tool, iPhyClassifier, and its application in analysis of the peach X-disease phytoplasma group (16SrIII. Int J Syst Evol Microbiol 59:2582–2593
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salehi, M., Esmailzadeh Hosseini, S.A., Salehi, E. et al. Genetic diversity and vector transmission of phytoplasmas associated with sesame phyllody in Iran. Folia Microbiol 62, 99–109 (2017). https://doi.org/10.1007/s12223-016-0476-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-016-0476-5