Abstract
Ebola virus (EBOV) infection is a widespread infection that has created a bad memory in Africa. In the 2014 and 2015 outbreak, more than 28,000 infections were reported by the World Health Organization, with about 11,300 deaths in Guinea, Liberia, and Sierra Leone. Heat shock protein A5 (HSPA5), termed also GRP78, is a host cell chaperone protein responsible for the unfolded protein response in the endoplasmic reticulum. Under stress, HSPA5 is upregulated and becomes cell-surface exposed. Recent studies report the association of cell-surface HSPA5 with EBOV glycoproteins GP1 and GP2. In this study, structural and sequence analysis and molecular docking are used to predict the possible binding site between the cell-surface HSPA5 and EBOV GP1. The results show a promising binding site that supports the hypothesis of HSPA5 selectivity for binding to a specific peptide sequence (pep42). This study paves the way to suggest possible inhibitors to stop viral association with cell-surface receptors and subsequently reduce viral infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ebola virus (EBOV) is one of the re-emerging viruses with a high mortality rate of up to 90% (Bhattacharyya and Hope 2011; Shurtleff et al. 2014). EBOV belongs to the filovirus family and affects the liver (Pallesen et al. 2016). EBOV requires different host factors during the life cycle (Cantoni and Rossman 2018; Ibrahim et al. 2019). The 2015 outbreak of EBOV caused more than 10,000 deaths with hemorrhagic fever as the main characteristic effect (El gohary et al. 2019). The ease with which the virus spread (via body fluids) and its high mortality rate made EBOV a global health threat of international concern (El gohary et al. 2019). The current development in drug design to eradicate the virus, using direct-acting antivirals (DAA), has reduced the momentum of viral spreading (Elfiky 2019; Gonzalez-Grande et al. 2016; Yang et al. 2011).
The upregulation of specific cellular proteins that mediate the alleviation mechanisms to reduce stress is induced by the unfolded protein response (UPR) mechanism in stressed cells. Heat shock proteins (HSP), the chaperones, are among those proteins that are upregulated under stress, as in viral infection or some types of cancers (Ibrahim et al. 2019). Glucose-regulated protein 78 (GRP78), a member named HSPA5 (Kampinga et al. 2009) of the HSP70 chaperone family is termed the master of the UPR mechanism in the lumen of the endoplasmic reticulum (ER) (Gething and Sambrook 1992; Ibrahim et al. 2019; Lee 2005; Li and Lee 2006; Quinones et al. 2008; Rao et al. 2002). Under cellular stressors, HSPA5 releases activating transcription factor 6 (ATF6), protein kinase RNA-like endoplasmic reticulum kinase (PERK), and inositol-requiring enzyme 1 (IRE1) because of the accumulation of unfolded proteins. The released enzymes cause inhibition of protein synthesis and enhancement of refolding mechanisms (Ibrahim et al. 2019; Shen et al. 2002). Subsequently, HSPA5 is upregulated and succeeds in escaping the ER retention (detected in the cytoplasm and over the cell membrane (cell-surface HSPA5)) (Ibrahim et al. 2019; Wu et al. 2014). Cell-surface HSPA5 is susceptible to pathogen recognition by viral envelope glycoproteins or fungal coat proteins (Gebremariam et al. 2014; Ibrahim et al. 2019). Pep42, a cyclic 13-residues peptide (CTVALPGGYVRVC) targets specifically cell-surface HSPA5 in vivo (Kim et al. 2006). It is used to deliver the chemotherapeutic, doxorubicin, to cancer cells presenting cell-surface HSPA5 (Ibrahim et al. 2019; Martin et al. 2010). Additionally, HSPA5 is reported to be associated with some viral proteins like spike protein in coronaviruses and E6 in human papillomavirus while the binding site was predicted (Elfiky 2020; Ibrahim et al. 2020). It was reported that HSPA5 is associated with EBOV in ER (Shurtleff et al. 2014). It was suggested that HSPA5 could be used as a drug target to stop EBOV infection (Shurtleff et al. 2014). In addition, EBOV glycoproteins GP1 and GP2 have been shown to accumulate in the ER of the infected cells, causing stress response (Bhattacharyya and Hope 2011).
In this work, the binding site between cell-surface HSPA5 and viral GP1 protein is predicted based on sequence, and thus, Pep42 is an example of a class of drugs that may reduce EBOV infections. Protein/peptide and protein/protein docking are employed to explore such binding using the protein/protein docking software “HADDOCK” which utilizes solvation and molecular dynamics simulation (MDS) in refining the interacting residues (binding site) for the interactions formed to be trustful (van Dijk and Bonvin 2006). In addition, HPEPDOCK constructs different possible conformations of the peptide and tests the binding affinity of each of the predicted structures against the target protein (Zhou et al. 2018).
Materials and methods
Nine solved structures for EBOV glycoproteins are found in the Protein Data Bank (Berman et al. 2003) with the following codes: 5JQ3, 6QD7, 6QD8, 6MAM, 6EA7, 5KEL, 5KEN, 6EA5, and 5KEM with a resolution of 2.23, 3.10, 3.30, 4.10, 4.25, 4.30, 4.30, 4.75, and 5.50 Å, respectively (Ehrhardt et al. 2019; Pallesen et al. 2016; West et al. 2018, 2019). Four of these structures are solved by x-ray crystallography (5JQ3, 6MAM, 6EA7, and 6EA5), while the rest are solved by cryo-electron microscopy. All the solved structures have been released during the past 3 years. The PDB files and their corresponding FASTA sequence files are downloaded from the Protein Data Bank database (PDB). By the aid of PyMOL software (DeLano 2002), water, ions, and protein chains other than EBOV GP1 are removed from the PDB files. The same is done with the only available wild-type and full-length HSPA5 (x-ray crystallography solved structure PDB ID: 5E84 with 2.99 Å resolution) in the open conformation (Yang et al. 2015; Yang et al. 2017). It is important to use the open conformation of the HSP5A in which the SBDα and SBDβ are apart from each other, and the substrate affinity to SBDβ is higher (Chiappori et al. 2016). The available sequences for EBOV GP1 found in the protein database of the National Center for Biotechnology Information (NCBI) are downloaded (ten sequences found in the identical protein group database), and Clustal Omega is utilized to perform multiple sequence alignment (MSA) with the Pep42 sequence CTVALPGGYVRVC (Sievers et al. 2011). ESpript 3.0 is used to represent the MSA (Gouet et al. 1999). The C121–C135 region of EBOV GP1 protein that fits well with Pep42 (33.3% identity) is analyzed by the ProtScale web server (ExPASy Bioinformatics Resource Portal) (Garg et al. 2016; Gasteiger et al. 2005). PyMOL is used to superimpose all the solved structures to show the structural conservation of the C121–C135 region of EBOV GP1.
HADDOCK web server (van Dijk and Bonvin 2006) was utilized to test the recognition of the full-length EBOV GP1 (PDB ID: 5JQ3: A, 6QD7: A, 6QD8: A, 6MAM: G, 6EA7: A, 5KEL: A, 5KEN: A, 6EA5: A, and 5KEM: A) by HSPA5 SDBβ (PDB ID: 5E84: A). For HSPA5, the active residues are selected to be I426, T428, V429, V432, T434, F451, S452, V457, and I459 (Yang et al. 2015). In addition, EBOV GP1 active residues are chosen to be the C121–C135 region. Protein-ligand interaction profiler (PLIP) web server (Salentin et al. 2015) was selected to assess the binding pattern after docking. Pep42 cyclic peptide (CTVALPGGYVRVC) model was built using I-TASSER web server (Zhang 2008). The protein/peptide docking software HPEPDOCK (Zhou et al. 2018) and HADDOCK were utilized to dock the cyclic Pep42 model into the binding site of HSPA5. A blind rigid docking scheme is used in HPEPDOCK without the binding site determination for HSPA5 to scan the possible binding sites. The easy interface of HADDOCK is utilized to dock the cyclic Pep42 peptide (all residues are treated as active) into HSPA5 SBDβ (I426, T428, V429, V432, T434, F451, S452, V457, and I459 are selected to be the active residues).
Results and discussion
Multiple sequence alignment
The cyclic peptide Pep42 was reported to selectively target cell-surface HSPA5 (which appears in some types of cancer) and used as a drug carrier for the anti-cancer agent, doxorubicin (Ibrahim et al. 2019; Yoneda et al. 2008). Figure 1a shows part of the multiple sequence alignment (MSA) between the Pep42 sequence CTVALPGGYVRVC and the ten EBOV GP1 sequences downloaded from the NCBI protein database. A complete MSA is found in the supplementary file (Fig. S1). Five identical residues (compared with the Pep42 sequence) were found in the MSA of EBOV GP1 sequences, which are C121, A124, P126, G128, and C135 (the numbering is based on PDB ID: 5JQ3). Interestingly, both C121 and C135 form disulfide bonds (see the green numbers at the bottom of Fig. 1a) with C147 and C108, respectively. The presence of these disulfides suggests its contribution to the folding of GP1 and GP2, as mentioned in earlier work (Cantoni and Rossman 2018). Moreover, the cyclic peptide (Pep42) can target CS-HSPA5. The C121–C135 region of EBOV GP1 is found in the β-turn region and the loop separating between the β7 and β8 secondary structure (see the top panel of Fig. 1a).
a Multiple sequence alignment (MSA) between Pep42 and EBOV GP1 sequences downloaded from the NCBI protein database. The full MSA is presented in the supplementary fig. S1. Alignment is made using the Clustal Ω web server and represented by ESpript 3 software. Red highlights indicate identical residues found, while residues written in red are conserved. b Hydrophobicity plot (Kyte and Doolittle) for both Pep42 (orange) and EBOV GP1 (C121–C135 region) (gray) peptides. c The surface representation of both the Pep42 cyclic peptide and EBOV GP1 C121–C135 region. Hydrophobic residues are colored red, hydrophilic residues in green, and residues with weak hydrophilicity are colored yellow
Structural superposition of the solved structures for EBOV GP1 (PDB IDs: 5JQ3, 6QD7, 6QD8, 6MAM, 6EA7, 5KEL, 5KEN, 6EA5, and 5KEM) found in the Protein Data Bank. The C121–C135 region in all structures is represented in red cartoons for clarity. Hydrophobic residues L122, I129, and F132 are labeled and represented in lines for the 5JQ3 structure. The right panel shows a 90° rotated view with both cartoon and surface representation to show the surface accessibility of the C121–C135 region
Figure 1b shows the hydrophobicity as a function of amino acid residues of both the Pep42 and C121–C135 regions of EBOV GP1 sequences. Kyte and Doolittle hydropathic parameters (Kyte and Doolittle 1982) are used by ProtScale web server of the ExPASy bioinformatics resource portal. A high degree of similarity in the hydrophobicity values for the EBOV GP1 C121–C135 region supports the MSA data. The sequence, and thus the structural similarity of EBOV GP1 compared with Pep42, indicates its ability to bind cell-surface HSPA5 substrate-binding domain β (SBDβ). In addition, Fig. 1c shows the molecular surface of the peptide Pep42 and the C121–C135 region of EBOV GP1. Three colors are used to represent the residues: highly hydrophobic residues in red (such as C, I, L, A, and V), low hydrophilicity residues in yellow (such as G, P, and T), and hydrophilic residues in green (such as D and R). As shown, the two structures show some hydrophobic patch that was reported previously to be the target regions for HSPA5. These hydrophobic residues of EBOV GP1 (C121, L122, A124, A125, I129, F132, and C135) are suggested to be the binding site for cell-surface HSPA5.
Structural conservation
Figure 2a shows the structural conservation of the EBOV GP1 C121–C135 region (red-colored cartoon) in all the solved structures (colored cartoons) found in the Protein Data Bank (PDB IDs: 5JQ3, 6QD7, 6QD8, 6MAM, 6EA7, 5KEL, 5KEN, 6EA5, and 5KEM). The root mean square deviation (RMSD) values of the superposition range from 0.35 Å up to 1.9 Å. As shown in the cartoon (lower) and surface representation (upper) of Fig. 2b (90° rotation on the x-axis of Fig. 2a), the C121–C135 region is surface exposed (encircled). It can interact with chaperone proteins with its hydrophobic residues L122, I129, and F132 (see the enlarged panel of Fig. 2a for 5JQ3)
EBOV GP1–HSPA5 docking
Table 1 lists the established interactions upon docking EBOV GP1 solved structures (PDB codes are listed) into HSPA5 SBDβ structure and the docking scores of HADDOCK (average values with the standard deviations calculated for the top-ranked cluster for each EBOV GP1 solved structure). Mainly, two types of interactions are established between EBOV GP1 and HSPA5, H-bonding and hydrophobic interactions. The 6EA5 structure only has two salt bridges formed (E124:K460 and E127 K447 for EBOV GP1–HSPA5). On average, six H-bonds are formed, and four hydrophobic interactions are established upon docking EBOV GP1 into HSPA5.
The primary residues of the EBOV GP1 that form H-bonds are A124, A125, P126, D/E127, G128, and R130. G128 forms only H-bonds (11 interactions), while A124, A125, D/E127, and R130 form both H-bonding and hydrophobic interactions with ratios 9:3, 5:4, 8:8, and 6:3, respectively. Moreover, the main interactions formed by P126 are hydrophobic (12 out of 13) interactions.
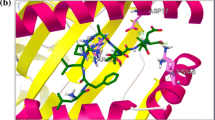
On the other hand, T428, V429, Q449, F451, S452, V453, K460, and V490 are the primary interacting residues from the HSPA5. S452 and K460 only interact through H-bonding (13 and 5 interactions, respectively). F451 and V490 form hydrophobic interactions only (6 interactions for each of them). Meanwhile, the other residues, T428, V429, Q449, and V453, form both H-bonds and hydrophobic contacts with EBOV GP1 (6:3, 4:3, 5:1, and 4:4, respectively). I426 and I459 both form few (3 and 4, respectively) hydrophobic interactions with EBOV GP1, while T458 show three H-bonds, and T434 forms two H-bonds and two hydrophobic contacts with HSPA5. Figure 3a and b show the docking pose of the EBOV GP1 structure (5JQ3) into HSPA5. In Fig. 3a, HSPA5 is represented in the green cartoon, while EBOV GP1 is represented in a cyan cartoon with the C121–C135 region in red. The residues involved in the interactions are in line representation and labeled in the enlarged panel. Figure 3b shows the molecular surface of HSPA5 and EBOV GP1 in cartoon representation. The docking pose shows that EBOV GP1 fits perfectly inside the substrate-binding domain β pocket of HSPA5 with a predicted binding affinity of − 89.4 ± 2.8 Kcal/mol (HADDOCK score).
EBOV GP1–HSPA5 docking experiment using HADDOCK. a HSPA5 solved structure (5E84) is represented in a green-colored cartoon with the EBOV GP1 solved structure (5JQ3) depicted in the cyan cartoon. The C121–C135 region of EBOV GP1 is represented in a red cartoon for clarification. Amino acids involved in H-bonding or hydrophobic interactions are labeled and expressed in lines. b The same docking complex but showing the molecular surface representation of the HSPA5
Pep42 peptide docking versus EBOV GP1
Table 2 shows the docking analysis made by PLIP for the Pep42 peptide after docking into HSPA5 SBDβ. The docking was made by HADDOCK as for the EBOV GP1 docking and by the peptide/protein docking software, HPEPDOCK. Docking scores are listed in the table, along with the interactions that were established after each docking experiment. On average, three H-bonds were formed with S452, T458, and K460 of the HSPA5. Eight hydrophobic interactions were found in the HADDOCK trial with I426, T428, V429, F451, and I459 of the HSPA5, while only five hydrophobic contacts were established in the case of the HPEPDOCK experiment (T428, V429, V432, and I450 of the HSPA5). Compared with EBOV GP1, the peptide Pep42 has the same pattern of interactions with HSPA5 (S452 and K460 for H-bonding while T428, V429, and F451 for hydrophobic contacts). This illustrates the possibility of recognition of the EBOV GP1 for cell-surface HSPA5 overexpressed on cancer cells or cells under other stressors.
The present in silico study predicts the binding mode of EBOV GP1 into the HSPA5 chaperone. Molecular dynamics simulation is suggested as future work to study the dynamics at the binding site and to test some binding inhibitors.
Conclusion
The Ebola virus, EBOV, is one of the most deadly viral infections in West-African countries. The viral protein GP1 is the crucial host-cell recognizing protein that enables viral entry. HSPA5 overexpression is reported to increase the infectivity of EBOV. Inhibiting HSPA5/EBOV GP1 binding would help in lowering the viral infection. The present work suggests the binding site of the viral/host-cell receptor utilizing in silico methods. This work paves the way for exploring HSPA5/EBOV GP1 binding inhibitors. Further experimental work is required to prove the suggested binding site and to test some inhibitors.
Data availability
The docking structures are available upon request from the corresponding author
References
Berman H, Henrick K, Nakamura H (2003) Announcing the worldwide Protein Data Bank. Nat Struct Mol Biol 10:980–980
Bhattacharyya S, Hope TJ (2011) Full-length Ebola glycoprotein accumulates in the endoplasmic reticulum. Virol J 8:11
Cantoni D, Rossman JS (2018) Ebolaviruses: new roles for old proteins. PLoS Negl Trop Dis 12:e0006349. https://doi.org/10.1371/journal.pntd.0006349
Chiappori F, Merelli I, Milanesi L, Colombo G, Morra G (2016) An atomistic view of Hsp70 allosteric crosstalk: from the nucleotide to the substrate binding domain and back. Sci Rep 6:23474. https://doi.org/10.1038/srep23474
DeLano WL (2002) The PyMOL molecular graphics system. Delano Scientific, San Carlos
Ehrhardt SA et al (2019) Polyclonal and convergent antibody response to Ebola virus vaccine rVSV-ZEBOV. Nat Med 25:1589–1600. https://doi.org/10.1038/s41591-019-0602-4
El gohary MI, Eissa A, Elfiky A, Desouky AA (2019) Ebola Virus L Polymerase RdRp sequence and phylogenetic analysis. Egy J Biomed Eng Biophy 29–36. https://doi.org/10.21608/ejbbe.2019.17450.1027
Elfiky AA (2019) Novel guanosine derivatives as anti-HCV NS5b polymerase: a QSAR and molecular docking study. Med Chem 15:130–137
Elfiky AA (2020) Human papillomavirus E6 - host cell receptor, GRP78, binding site prediction. J Med Virol in press. https://doi.org/10.1002/jmv.25737
Garg VK, Avashthi H, Tiwari A, Jain PA, Ramkete PW, Kayastha AM, Singh VK (2016) MFPPI–Multi FASTA ProtParam Interface. Bioinformation 12:74
Gasteiger E, Hoogland C, Gattiker A, Duvaud S, Wilkins MR, Appel RD, Bairoch A (2005) Protein identification and analysis tools on the ExPASy server; (In) John M. Walker (ed): The Proteomics Protocols Handbook, Humana Press. pp. 571–607
Gebremariam T, Liu M, Luo G, Bruno V, Phan QT, Waring AJ, Edwards JE Jr, Filler SG, Yeaman MR, Ibrahim AS (2014) CotH3 mediates fungal invasion of host cells during mucormycosis. J Clin Invest 124:237–250
Gething M-J, Sambrook J (1992) Protein folding in the cell. Nature 355:33
Gonzalez-Grande R, Jimenez-Perez M, Gonzalez Arjona C, Mostazo Torres J (2016) New approaches in the treatment of hepatitis C. World J Gastroenterol 22:1421–1432. https://doi.org/10.3748/wjg.v22.i4.1421
Gouet P, Courcelle E, Stuart DI, Métoz F (1999) ESPript: analysis of multiple sequence alignments in PostScript. Bioinformatics 15:305–308
Ibrahim IM, Abdelmalek DH, Elfiky AA (2019) GRP78: A cell’s response to stress. Life Sci 226:156–163. https://doi.org/10.1016/j.lfs.2019.04.022
Ibrahim IM, Abdelmalek DH, Elshahat ME, Elfiky AA (2020) COVID-19 spike-host cell receptor GRP78 binding site prediction. J Inf. https://doi.org/10.1016/j.jinf.2020.02.026
Kampinga HH et al (2009) Guidelines for the nomenclature of the human heat shock proteins. Cell Stress Chaperones 14:105–111. https://doi.org/10.1007/s12192-008-0068-7
Kim Y, Lillo AM, Steiniger SC, Liu Y, Ballatore C, Anichini A, Mortarini R, Kaufmann GF, Zhou B, Felding-Habermann B, Janda KD (2006) Targeting heat shock proteins on cancer cells: selection, characterization, and cell-penetrating properties of a peptidic GRP78 ligand. Biochemistry 45:9434–9444
Kyte J, Doolittle RF (1982) A simple method for displaying the hydropathic character of a protein. J Mol Biol 157:105–132
Lee AS (2005) The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods 35:373–381
Li J, Lee AS (2006) Stress induction of GRP78/BiP and its role in cancer. Curr Mol Med 6:45–54
Martin S, Hill DS, Paton JC, Paton AW, Birch-Machin MA, Lovat PE, Redfern CP (2010) Targeting GRP78 to enhance melanoma cell death. Pigment Cell Melanoma Res 23:675–682
Pallesen J, Murin CD, de Val N, Cottrell CA, Hastie KM, Turner HL, Fusco ML, Flyak AI, Zeitlin L, Crowe JE Jr, Andersen KG, Saphire EO, Ward AB (2016) Structures of Ebola virus GP and sGP in complex with therapeutic antibodies. Nat Microbiol 1:16128. https://doi.org/10.1038/nmicrobiol.2016.128
Quinones QJ, de Ridder GG, Pizzo SV (2008) GRP78: a chaperone with diverse roles beyond the endoplasmic reticulum. Histol Histopathol. 23(11):1409–1416. https://doi.org/10.14670/HH-23.1409
Rao RV et al (2002) Coupling endoplasmic reticulum stress to the cell death program: role of the ER chaperone GRP78. FEBS Lett 514:122–128
Salentin S, Schreiber S, Haupt VJ, Adasme MF, Schroeder M (2015) PLIP: fully automated protein–ligand interaction profiler. Nucleic Acids Res 43:W443–W447
Shen J, Chen X, Hendershot L, Prywes R (2002) ER stress regulation of ATF6 localization by dissociation of BiP/GRP78 binding and unmasking of Golgi localization signals. Dev Cell 3:99–111
Shurtleff AC, Costantino JA, Tritsch SR, Retterer C, Spurgers KB, Bavari S (2014) HSPA5 is an essential host factor for Ebola virus infection. Antivir Res 109:171–174
Sievers F et al (2011) Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal omega. Mol Syst Biol 7:539
van Dijk AD, Bonvin AM (2006) Solvated docking: introducing water into the modelling of biomolecular complexes. Bioinformatics 22:2340–2347. https://doi.org/10.1093/bioinformatics/btl395
West BR, Moyer CL, King LB, Fusco ML, Milligan JC, Hui S, Saphire EO (2018) Structural basis of pan-ebolavirus neutralization by a human antibody against a conserved, yet cryptic epitope. MBio:9. https://doi.org/10.1128/mBio.01674-18
West BR et al (2019) Structural basis of broad ebolavirus neutralization by a human survivor antibody. Nat Struct Mol Biol 26:204–212. https://doi.org/10.1038/s41594-019-0191-4
Wu C-T et al (2014) Glucose-regulated protein 78 mediates hormone-independent prostate cancer progression and metastasis through maspin and COX-2 expression. Tumor Biol 35:195–204
Yang PL, Gao M, Lin K, Liu Q, Villareal VA (2011) Anti-HCV drugs in the pipeline. Curr Opin Virol 1:607–616. https://doi.org/10.1016/j.coviro.2011.10.019
Yang J, Nune M, Zong Y, Zhou L, Liu Q (2015) Close and allosteric opening of the polypeptide-binding site in a human Hsp70 chaperone BiP. Structure 23:2191–2203. https://doi.org/10.1016/j.str.2015.10.012
Yang J et al (2017) Conformation transitions of the polypeptide-binding pocket support an active substrate release from Hsp70s. Nat Commun 8:1201. https://doi.org/10.1038/s41467-017-01310-z
Yoneda Y, Steiniger SC, Capkova K, Mee JM, Liu Y, Kaufmann GF, Janda KD (2008) A cell-penetrating peptidic GRP78 ligand for tumor cell-specific prodrug therapy. Bioorg Med Chem Lett 18:1632–1636. https://doi.org/10.1016/j.bmcl.2008.01.060
Zhang Y (2008) I-TASSER server for protein 3D structure prediction. BMC Bioinformatics 9:40
Zhou P, Jin B, Li H, Huang S-Y (2018) HPEPDOCK: a web server for blind peptide–protein docking based on a hierarchical algorithm. Nucleic Acids Res 46:W443–W450
Author information
Authors and Affiliations
Contributions
A. E. designed the research study, performed the docking calculations, analyzed the data, and wrote the paper. A. E. has read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declares that he has no competing interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 23 kb).
Rights and permissions
About this article
Cite this article
Elfiky, A.A. Ebola virus glycoprotein GP1—host cell-surface HSPA5 binding site prediction. Cell Stress and Chaperones 25, 541–548 (2020). https://doi.org/10.1007/s12192-020-01106-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-020-01106-z