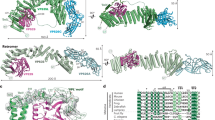
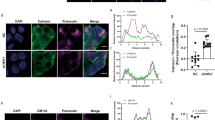
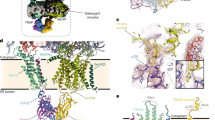
Abstract
Coat protein complex II (COPII) plays an essential role in the export of cargo molecules such as secretory proteins, membrane proteins, and lipids from the endoplasmic reticulum (ER). In yeast, the COPII machinery is critical for cell viability as most COPII knockout mutants fail to survive. In mice and fish, homozygous knockout mutants of most COPII genes are embryonic lethal, reflecting the essentiality of the COPII machinery in the early stages of vertebrate development. In humans, COPII mutations, which are often hypomorphic, cause diseases having distinct clinical features. This is interesting as the fundamental cellular defect of these diseases, that is, failure of ER export, is similar. Analyses of humans and animals carrying COPII mutations have revealed clues to why a similar ER export defect can cause such different diseases. Previous reviews have focused mainly on the deficit of secretory or membrane proteins in the final destinations because of an ER export block. In this review, we also underscore the other consequence of the ER export block, namely ER stress triggered by the accumulation of cargo proteins in the ER.


Similar content being viewed by others
References
Adams EJ, Chen XW, O'Shea KS, Ginsburg D (2014) Mammalian COPII coat component SEC24C is required for embryonic development in mice. J Biol Chem 289:20858–20870. https://doi.org/10.1074/jbc.M114.566687
Anselstetter V, Horstmann HJ, Heimpel H (1977) Congenital dyserythropoietic anaemia, types I and II: aberrant pattern of erythrocyte membrane proteins in CDA II, as revealed by two-dimensional polyacrylamide gel electrophoresis. Br J Haematol 35:209–215. https://doi.org/10.1111/j.1365-2141.1977.tb00577.x
Antonny B, Madden D, Hamamoto S, Orci L, Schekman R (2001) Dynamics of the COPII coat with GTP and stable analogues. Nat Cell Biol 3:531–537. https://doi.org/10.1038/3507850035078500
Bailly E, Walton A, Borg JP (2018) The planar cell polarity Vangl2 protein: from genetics to cellular and molecular functions. Semin Cell Dev Biol, 81:62–70. https://doi.org/10.1016/j.semcdb.2017.10.030
Baines AC, Adams EJ, Zhang B, Ginsburg D (2013) Disruption of the Sec24d gene results in early embryonic lethality in the mouse. PLoS One 8:e61114. https://doi.org/10.1371/journal.pone.0061114
Barlowe C, Schekman R (1993) SEC12 encodes a guanine-nucleotide-exchange factor essential for transport vesicle budding from the ER. Nature 365:347–349. https://doi.org/10.1038/365347a0
Barlowe C et al (1994) COPII: a membrane coat formed by sec proteins that drive vesicle budding from the endoplasmic reticulum. Cell 77:895–907. https://doi.org/10.1016/0092-8674(94)90138-4
Bi X, Corpina RA, Goldberg J (2002) Structure of the Sec23/24-Sar1 pre-budding complex of the COPII vesicle coat. Nature 419:271–277. https://doi.org/10.1038/nature01040
Bianchi P et al (2009) Congenital dyserythropoietic anemia type II (CDAII) is caused by mutations in the SEC23B gene. Hum Mutat 30:1292–1298. https://doi.org/10.1002/humu.21077
Boyadjiev SA et al (2006) Cranio-lenticulo-sutural dysplasia is caused by a SEC23A mutation leading to abnormal endoplasmic-reticulum-to-Golgi trafficking. Nat Genet 38:1192–1197. https://doi.org/10.1038/ng1876
Boyadjiev SA et al (2011) Cranio-lenticulo-sutural dysplasia associated with defects in collagen secretion. Clin Genet 80:169–176. https://doi.org/10.1111/j.1399-0004.2010.01550.x
Chang HJ, Jesch SA, Gaspar ML, Henry SA (2004) Role of the unfolded protein response pathway in secretory stress and regulation of INO1 expression in Saccharomyces cerevisiae. Genetics 168:1899–1913. https://doi.org/10.1534/genetics.104.032961
Chen XW et al (2013) SEC24A deficiency lowers plasma cholesterol through reduced PCSK9 secretion. eLife 2:e00444. https://doi.org/10.7554/eLife.00444
Cox JS, Walter P (1996) A novel mechanism for regulating activity of a transcription factor that controls the unfolded protein response. Cell 87:391–404. https://doi.org/10.1016/s0092-8674(00)81360-4
Espenshade P, Gimeno RE, Holzmacher E, Teung P, Kaiser CA (1995) Yeast SEC16 gene encodes a multidomain vesicle coat protein that interacts with Sec23p. J Cell Biol 131:311–324. https://doi.org/10.1083/jcb.131.2.311
Fath S, Mancias JD, Bi X, Goldberg J (2007) Structure and organization of coat proteins in the COPII cage. Cell 129:1325–1336. https://doi.org/10.1016/j.cell.2007.05.036
Fawcett TW, Martindale JL, Guyton KZ, Hai T, Holbrook NJ (1999) Complexes containing activating transcription factor (ATF)/cAMP-responsive-element-binding protein (CREB) interact with the CCAAT/enhancer-binding protein (C/EBP)-ATF composite site to regulate Gadd153 expression during the stress response. Biochem J 339(Pt 1):135–141
Fromme JC et al (2007) The genetic basis of a craniofacial disease provides insight into COPII coat assembly. Dev Cell 13:623–634. https://doi.org/10.1016/j.devcel.2007.10.005
Gaddam D, Stevens N, Hollien J (2013) Comparison of mRNA localization and regulation during endoplasmic reticulum stress in Drosophila cells. Mol Biol Cell 24:14–20. https://doi.org/10.1091/mbc.E12-06-0491
Garbes L et al (2015) Mutations in SEC24D, encoding a component of the COPII machinery, cause a syndromic form of osteogenesis imperfecta. Am J Hum Genet 96:432–439. https://doi.org/10.1016/j.ajhg.2015.01.002
Georges A et al (2011) Molecular analysis and intestinal expression of SAR1 genes and proteins in Anderson’s disease (chylomicron retention disease). Orphanet J Rare Dis 6:1. https://doi.org/10.1186/1750-1172-6-1
Greene ND, Copp AJ (2014) Neural tube defects. Annu Rev Neurosci 37:221–242. https://doi.org/10.1146/annurev-neuro-062012-170354
Halliday M, Radford H, Sekine Y, Moreno J, Verity N, le Quesne J, Ortori CA, Barrett DA, Fromont C, Fischer PM, Harding HP, Ron D, Mallucci GR (2015) Partial restoration of protein synthesis rates by the small molecule ISRIB prevents neurodegeneration without pancreatic toxicity. Cell Death Dis 6:e1672. https://doi.org/10.1038/cddis.2015.49
Halperin D, Kadir R, Perez Y, Drabkin M, Yogev Y, Wormser O, Berman EM, Eremenko E, Rotblat B, Shorer Z, Gradstein L, Shelef I, Birk R, Abdu U, Flusser H, Birk OS (2019) SEC31A mutation affects ER homeostasis, causing a neurological syndrome. J Med Genet 56:139–148. https://doi.org/10.1136/jmedgenet-2018-105503
Harding HP, Zhang Y, Bertolotti A, Zeng H, Ron D (2000) Perk is essential for translational regulation and cell survival during the unfolded protein response. Mol Cell 5:897–904
Haze K, Yoshida H, Yanagi H, Yura T, Mori K (1999) Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol Biol Cell 10:3787–3799. https://doi.org/10.1091/mbc.10.11.3787
Hetz C, Russelakis-Carneiro M, Maundrell K, Castilla J, Soto C (2003) Caspase-12 and endoplasmic reticulum stress mediate neurotoxicity of pathological prion protein. EMBO J 22:5435–5445. https://doi.org/10.1093/emboj/cdg537
Hicke L, Schekman R (1989) Yeast Sec23p acts in the cytoplasm to promote protein transport from the endoplasmic reticulum to the Golgi complex in vivo and in vitro. EMBO J 8:1677–1684
Higashio H, Kohno K (2002) A genetic link between the unfolded protein response and vesicle formation from the endoplasmic reticulum. Biochem Biophys Res Commun 296:568–574. https://doi.org/10.1016/s0006-291x(02)00923-3
Hollien J, Lin JH, Li H, Stevens N, Walter P, Weissman JS (2009) Regulated Ire1-dependent decay of messenger RNAs in mammalian cells. J Cell Biol 186:323–331. https://doi.org/10.1083/jcb.200903014
Jones B et al (2003) Mutations in a Sar1 GTPase of COPII vesicles are associated with lipid absorption disorders. Nat Genet 34:29–31. https://doi.org/10.1038/ng1145ng1145
Khoriaty R et al (2018) Functions of the COPII gene paralogs SEC23A and SEC23B are interchangeable in vivo. Proc Natl Acad Sci U S A 115:E7748–E7757. https://doi.org/10.1073/pnas.1805784115
Kimmig P, Diaz M, Zheng J, Williams CC, Lang A, Aragón T, Li H, Walter P (2012) The unfolded protein response in fission yeast modulates stability of select mRNAs to maintain protein homeostasis. 1:e00048–eLife. https://doi.org/10.7554/eLife.00048
Krupp M, Marquardt JU, Sahin U, Galle PR, Castle J, Teufel A (2012) RNA-Seq Atlas—a reference database for gene expression profiling in normal tissue by next-generation sequencing. Bioinformatics 28:1184–1185. https://doi.org/10.1093/bioinformatics/bts084
Kung LF et al (2011) Sec24p and Sec16p cooperate to regulate the GTP cycle of the COPII coat. EMBO J 31:1014–1027. https://doi.org/10.1038/emboj.2011.444
Kurihara T, Hamamoto S, Gimeno RE, Kaiser CA, Schekman R, Yoshihisa T (2000) Sec24p and Iss1p function interchangeably in transport vesicle formation from the endoplasmic reticulum in Saccharomyces cerevisiae. Mol Biol Cell 11:983–998. https://doi.org/10.1091/mbc.11.3.983
Lang MR, Lapierre LA, Frotscher M, Goldenring JR, Knapik EW (2006) Secretory COPII coat component Sec23a is essential for craniofacial chondrocyte maturation. Nat Genet 38:1198–1203. https://doi.org/10.1038/ng1880
Lee MC, Orci L, Hamamoto S, Futai E, Ravazzola M, Schekman R (2005) Sar1p N-terminal helix initiates membrane curvature and completes the fission of a COPII vesicle. Cell 122:605–617. https://doi.org/10.1016/j.cell.2005.07.025
Lee CS et al (2017) A chemical chaperone improves muscle function in mice with a RyR1 mutation. 8:14659–Nat Commun. https://doi.org/10.1038/ncomms14659
Lek M et al (2016) Analysis of protein-coding genetic variation in 60,706 humans. Nature 536:285–283. https://doi.org/10.1038/nature19057
Levic DS, Minkel JR, Wang WD, Rybski WM, Melville DB, Knapik EW (2015) Animal model of Sar1b deficiency presents lipid absorption deficits similar to Anderson disease. J Mol Med (Berl) 93:165–176. https://doi.org/10.1007/s00109-014-1247-x
Lord C et al (2011) Sequential interactions with Sec23 control the direction of vesicle traffic. Nature 473:181–186. https://doi.org/10.1038/nature09969
Ma W, Goldberg E, Goldberg J (2017) ER retention is imposed by COPII protein sorting and attenuated by 4-phenylbutyrate. eLife 6. https://doi.org/10.7554/eLife.26624
Matsuoka K, Orci L, Amherdt M, Bednarek SY, Hamamoto S, Schekman R, Yeung T (1998) COPII-coated vesicle formation reconstituted with purified coat proteins and chemically defined liposomes. Cell 93:263–275
Merte J, Jensen D, Wright K, Sarsfield S, Wang Y, Schekman R, Ginty DD (2010) Sec24b selectively sorts Vangl2 to regulate planar cell polarity during neural tube closure. Nat Cell Biol 12:41–46; sup pp 41-48. https://doi.org/10.1038/ncb2002
Miller EA, Beilharz TH, Malkus PN, Lee MC, Hamamoto S, Orci L, Schekman R (2003) Multiple cargo binding sites on the COPII subunit Sec24p ensure capture of diverse membrane proteins into transport vesicles. Cell 114:497–509
Miyake M et al (2017) Ligand-induced rapid skeletal muscle atrophy in HSA-Fv2E-PERK transgenic mice. PLoS One 12:e0179955. https://doi.org/10.1371/journal.pone.0179955
Montcouquiol M, Rachel RA, Lanford PJ, Copeland NG, Jenkins NA, Kelley MW (2003) Identification of Vangl2 and Scrb1 as planar polarity genes in mammals. Nature 423:173–177. https://doi.org/10.1038/nature01618
Moreira TG et al (2015) Sec13 regulates expression of specific immune factors involved in inflammation in vivo. Sci Rep 5:17655. https://doi.org/10.1038/srep17655
Moreno JA et al (2013) Oral treatment targeting the unfolded protein response prevents neurodegeneration and clinical disease in prion-infected mice. Sci Transl Med 5:206ra138. https://doi.org/10.1126/scitranslmed.3006767
Mori K, Ma W, Gething MJ, Sambrook J (1993) A transmembrane protein with a cdc2+/CDC28-related kinase activity is required for signaling from the ER to the nucleus. cell 74:743–756. https://doi.org/10.1016/0092-8674(93)90521-q
Morishima N, Nakanishi K, Nakano A (2011) Activating transcription factor-6 (ATF6) mediates apoptosis with reduction of myeloid cell leukemia sequence 1 (Mcl-1) protein via induction of WW domain binding protein 1. J Biol Chem 286:35227–35235. https://doi.org/10.1074/jbc.M111.233502
Nakano A, Muramatsu M (1989) A novel GTP-binding protein, Sar1p, is involved in transport from the endoplasmic reticulum to the Golgi apparatus. J Cell Biol 109:2677–2691. https://doi.org/10.1083/jcb.109.6.2677
Nakano A, Brada D, Schekman R (1988) A membrane glycoprotein, Sec12p, required for protein transport from the endoplasmic reticulum to the Golgi apparatus in yeast. J Cell Biol 107:851–863. https://doi.org/10.1083/jcb.107.3.851
Niu X et al (2012) Sec13 safeguards the integrity of the endoplasmic reticulum and organogenesis of the digestive system in zebrafish. Dev Biol. https://doi.org/10.1016/j.ydbio.2012.05.004
Niu X, Hong J, Zheng X, Melville DB, Knapik EW, Meng A, Peng J (2014) The nuclear pore complex function of Sec13 protein is required for cell survival during retinal development. J Biol Chem 289:11971–11985. https://doi.org/10.1074/jbc.M114.547190
Novick P, Field C, Schekman R (1980) Identification of 23 complementation groups required for post-translational events in the yeast secretory pathway. cell 21:205–215
Ohisa S, Inohaya K, Takano Y, Kudo A (2010) sec24d encoding a component of COPII is essential for vertebra formation, revealed by the analysis of the medaka mutant, vbi. Dev Biol 342:85–95. https://doi.org/10.1016/j.ydbio.2010.03.016
Peretti N, Sassolas A, Roy CC, Deslandres C, Charcosset M, Castagnetti J, Pugnet-Chardon L, Moulin P, Labarge S, Bouthillier L, Lachaux A, Levy E, Department of Nutrition-Hepatogastroenterology, Hôpital Femme Mère Enfant, Bron, Université Lyon 1, Department of Pediatrics, CHU Sainte-Justine Research Center, Université de Montréal (2010) Guidelines for the diagnosis and management of chylomicron retention disease based on a review of the literature and the experience of two centers. Orphanet J Rare Dis 5:24. https://doi.org/10.1186/1750-1172-5-24
Pryer NK, Salama NR, Schekman R, Kaiser CA (1993) Cytosolic Sec13p complex is required for vesicle formation from the endoplasmic reticulum in vitro. J Cell Biol 120:865–875. https://doi.org/10.1083/jcb.120.4.865
Queisser W, Spiertz E, Jost E, Heimpel H (1971) Proliferation disturbances of erythroblasts in congenital dyserythropoietic anemia type I and II. Acta Haematol 45:65–76. https://doi.org/10.1159/000208608
Roberg KJ, Crotwell M, Espenshade P, Gimeno R, Kaiser CA (1999) LST1 is a SEC24 homologue used for selective export of the plasma membrane ATPase from the endoplasmic reticulum. J Cell Biol 145:659–672. https://doi.org/10.1083/jcb.145.4.659
Ron D, Walter P (2007) Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol 8:519–529. https://doi.org/10.1038/nrm2199
Rudd PM, Merry AH, Wormald MR, Dwek RA (2002) Glycosylation and prion protein. Curr Opin Struct Biol 12:578–586. https://doi.org/10.1016/s0959-440x(02)00377-9
Saito K, Katada T (2015) Mechanisms for exporting large-sized cargoes from the endoplasmic reticulum. Cell Mol Life Sci 72:3709–3720. https://doi.org/10.1007/s00018-015-1952-9
Salama NR, Chuang JS, Schekman RW (1997) Sec31 encodes an essential component of the COPII coat required for transport vesicle budding from the endoplasmic reticulum. Mol Biol Cell 8:205–217. https://doi.org/10.1091/mbc.8.2.205
Sarmah S, Barrallo-Gimeno A, Melville DB, Topczewski J, Solnica-Krezel L, Knapik EW (2010) Sec24D-dependent transport of extracellular matrix proteins is required for zebrafish skeletal morphogenesis. PLoS One 5:e10367. https://doi.org/10.1371/journal.pone.0010367
Sato M, Sato K, Nakano A (2002) Evidence for the intimate relationship between vesicle budding from the ER and the unfolded protein response. Biochem Biophys Res Commun 296:560–567. https://doi.org/10.1016/s0006-291x(02)00922-1
Schwarz K et al (2009) Mutations affecting the secretory COPII coat component SEC23B cause congenital dyserythropoietic anemia type II. Nat Genet 41:936–940. https://doi.org/10.1038/ng.405
Shen J, Chen X, Hendershot L, Prywes R (2002) ER stress regulation of ATF6 localization by dissociation of BiP/GRP78 binding and unmasking of Golgi localization signals. Dev Cell 3:99–111
Sidrauski C, Acosta-Alvear D, Khoutorsky A, Vedantham P, Hearn BR, Li H, Gamache K, Gallagher CM, Ang KK, Wilson C, Okreglak V, Ashkenazi A, Hann B, Nader K, Arkin MR, Renslo AR, Sonenberg N, Walter P (2013) Pharmacological brake-release of mRNA translation enhances cognitive memory. eLife 2:e00498. https://doi.org/10.7554/eLife.00498
Sidrauski C et al (2015) Pharmacological dimerization and activation of the exchange factor eIF2B antagonizes the integrated stress response. eLife 4:e07314. https://doi.org/10.7554/eLife.07314
Siniossoglou S et al (1996) A novel complex of nucleoporins, which includes Sec13p and a Sec13p homolog, is essential for normal nuclear pores. cell 84:265–275. https://doi.org/10.1016/s0092-8674(00)80981-2
Stagg SM, LaPointe P, Razvi A, Gurkan C, Potter CS, Carragher B, Balch WE (2008) Structural basis for cargo regulation of COPII coat assembly. Cell 134:474–484. https://doi.org/10.1016/j.cell.2008.06.024
Stainier DYR, Raz E, Lawson ND, Ekker SC, Burdine RD, Eisen JS, Ingham PW, Schulte-Merker S, Yelon D, Weinstein BM, Mullins MC, Wilson SW, Ramakrishnan L, Amacher SL, Neuhauss SCF, Meng A, Mochizuki N, Panula P, Moens CB (2017) Guidelines for morpholino use in zebrafish. PLoS Genet 13:e1007000. https://doi.org/10.1371/journal.pgen.1007000
Tang BL, Kausalya J, Low DY, Lock ML, Hong W (1999) A family of mammalian proteins homologous to yeast Sec24p. Biochem Biophys Res Commun 258:679–684. https://doi.org/10.1006/bbrc.1999.0574
Tao J et al (2012) SEC23B is required for the maintenance of murine professional secretory tissues. Proc Natl Acad Sci U S A 109:E2001–E2009. https://doi.org/10.1073/pnas.1209207109
Townley AK, Feng Y, Schmidt K, Carter DA, Porter R, Verkade P, Stephens DJ (2008) Efficient coupling of Sec23-Sec24 to Sec13-Sec31 drives COPII-dependent collagen secretion and is essential for normal craniofacial development. J Cell Sci 121:3025–3034. https://doi.org/10.1242/jcs.031070
Ulirsch JC, Lacy JN, An X, Mohandas N, Mikkelsen TS, Sankaran VG (2014) Altered chromatin occupancy of master regulators underlies evolutionary divergence in the transcriptional landscape of erythroid differentiation. PLoS Genet 10:e1004890. https://doi.org/10.1371/journal.pgen.1004890
Upton JP et al (2012) IRE1alpha cleaves select microRNAs during ER stress to derepress translation of proapoptotic Caspase-2. science 338:818–822. https://doi.org/10.1126/science.1226191
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. science 334:1081–1086. https://doi.org/10.1126/science.1209038
Wang M, Kaufman RJ (2016) Protein misfolding in the endoplasmic reticulum as a conduit to human disease. Nature 529:326–335. https://doi.org/10.1038/nature17041
Wang XZ, Harding HP, Zhang Y, Jolicoeur EM, Kuroda M, Ron D (1998) Cloning of mammalian Ire1 reveals diversity in the ER stress responses. EMBO J 17:5708–5717. https://doi.org/10.1093/emboj/17.19.5708
Wang B, Joo JH, Mount R, Teubner BJW, Krenzer A, Ward AL, Ichhaporia VP, Adams EJ, Khoriaty R, Peters ST, Pruett-Miller SM, Zakharenko SS, Ginsburg D, Kundu M (2018) The COPII cargo adapter SEC24C is essential for neuronal homeostasis. J Clin Invest 128:3319–3332. https://doi.org/10.1172/JCI98194
Wilmshurst JM et al (2010) RYR1 mutations are a common cause of congenital myopathies with central nuclei. Ann Neurol 68:717–726. https://doi.org/10.1002/ana.22119
Yang XY et al (2013) Mutations in the COPII vesicle component gene SEC24B are associated with human neural tube defects. Hum Mutat 34:1094–1101. https://doi.org/10.1002/humu.22338
Yeh KY, Lai CY, Lin CY, Hsu CC, Lo CP, Her GM (2017) ATF4 overexpression induces early onset of hyperlipidaemia and hepatic steatosis and enhances adipogenesis in zebrafish. Sci Rep 7:16362. https://doi.org/10.1038/s41598-017-16587-9
Yoneda T, Imaizumi K, Oono K, Yui D, Gomi F, Katayama T, Tohyama M (2001) Activation of caspase-12, an endoplastic reticulum (ER) resident caspase, through tumor necrosis factor receptor-associated factor 2-dependent mechanism in response to the ER stress. J Biol Chem 276:13935–13940. https://doi.org/10.1074/jbc.M010677200
Yoshida H, Matsui T, Yamamoto A, Okada T, Mori K (2001) XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. cell 107:881–891. https://doi.org/10.1016/s0092-8674(01)00611-0
Zanetti G, Prinz S, Daum S, Meister A, Schekman R, Bacia K, Briggs JA (2013) The structure of the COPII transport-vesicle coat assembled on membranes. eLife 2:e00951. https://doi.org/10.7554/eLife.00951
Zhu M, Tao J, Vasievich MP, Wei W, Zhu G, Khoriaty RN, Zhang B (2015) Neural tube opening and abnormal extraembryonic membrane development in SEC23A deficient mice. Sci Rep 5:15471. https://doi.org/10.1038/srep15471
Zinszner H et al (1998) CHOP is implicated in programmed cell death in response to impaired function of the endoplasmic reticulum. Genes Dev 12:982–995. https://doi.org/10.1101/gad.12.7.982
Acknowledgments
The authors thank Leah Wabel for proofreading the manuscript. This work was supported by the National Institute of General Medical Sciences of the National Institutes of Health under award number R01GM110373 to J.K.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lu, CL., Kim, J. Consequences of mutations in the genes of the ER export machinery COPII in vertebrates. Cell Stress and Chaperones 25, 199–209 (2020). https://doi.org/10.1007/s12192-019-01062-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-019-01062-3