Abstract
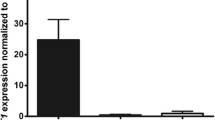
Dupuytren’s contracture (DC) is a fibroproliferative disorder of unknown etiology characterized by a scar-like contracture that develops in the palm and/or digits. We have previously reported that the eta subunit of the chaperonin containing T-complex polypeptide (CCT-eta) is increased in fibrotic wound healing, and is essential for the accumulation of α-smooth muscle actin (α-SMA) in fibroblasts. The purpose of this study was to determine if CCT-eta is similarly implicated in the aberrant fibrosis seen in DC and to investigate the role of CCT-eta in the behavior of myo/fibroblasts in DC. Fibroblasts were obtained from DC-affected palmar fascia, from adjacent phenotypically normal palmar fascia in the same DC patients (PF), and from non-DC palmar fascial tissues in patients undergoing carpal tunnel (CT) release. Inherent contractility in these three populations was examined using fibroblast-populated collagen lattices (FPCLs) and by cell traction force microscopy. Expression of CCT-eta and α-SMA protein was determined by Western blot. The effect of CCT-eta inhibition on the contractility of DC cells was determined by deploying an siRNA versus CCT-eta. DC cells were significantly more contractile than both matching palmar fascial (PF) cells and CT cells in both assays, with PF cells demonstrating an intermediate contractility in the FPCL assay. Whereas α-SMA protein was significantly increased only in DC cells compared to PF and CT cells, CCT-eta protein was significantly increased in both PF and DC cells compared to CT cells. siRNA-mediated depletion of CCT-eta inhibited the accumulation of both CCT-eta and α-SMA protein in DC cells, and also significantly decreased the contractility of treated DC cells. These observations suggest that increased expression of CCT-eta appears to be a marker for latent and active disease in these patients and to be essential for the increased contractility exhibited by these fibroblasts.






Similar content being viewed by others
References
Badalamente MA, Hurst LC (2007) Efficacy and safety of injectable mixed collagenase subtypes in the treatment of Dupuytren’s contracture. J Hand Surg Am 32:767–774
Badalamente MA, Hurst LC, Hentz VR (2002) Collagen as a clinical target: nonoperative treatment of Dupuytren’s disease. J Hand Surg Am 27:788–798
Bell E, Ivarsson B, Merrill C (1979) Production of a tissue-like structure by contraction of collagen lattices by human fibroblasts of different proliferative potential in vitro. Proc Natl Acad Sci 76:1274–178
Beningo KA, Wang YL (2002) Flexible substrata for the detection of cellular traction forces. Trends Cell Biol 12:79–84
Beningo KA, Lo CM, Wang YL (2002) Flexible polyacrylamide substrata for the analysis of mechanical interactions at cell-substratum adhesions. Methods Cell Biol 69:325–339
Bisson MA, Mudera V, McGrouther DA, Grobbelaar AO (2004) The contractile properties and responses to tensional loading of Dupuytren’s disease-derived fibroblasts are altered: a cause of the contracture? Plast Reconstr Surg 113:611–621, discussion 622–624
Butler JP, Tolic-Norrelykke IM, Fabry B, Fredberg JJ (2002) Traction fields, moments, and strain energy that cells exert on their surroundings. Am J Physiol Cell Physiol 282:C595–C605
Cass DL, Sylvester KG, Yang EY, Crombleholme TM, Adzick NS (1997) Myofibroblast persistence in fetal sheep wounds is associated with scar formation. J Pediatr Surg 32:1017–1021
Degreef I, Steeno P, De Smet PL (2008) A survey of clinical manifestations and risk factors in women with Dupuytren’s disease. Acta Orthop Belg 74:456–460
Hindocha S, Stanley JK, Watson JS, Bayat A (2008) Revised Tubiana’s staging system for assessment of disease severity in Dupuytren’s disease—preliminary clinical findings. Hand 3:80–86
Hinz B (2007) Formation and function of the myofibroblast during tissue repair. J Invest Dermatol 127:526–537
Howard JC, Varallo VM, Ross DC, Roth JH, Faber KJ, Alman B, Gan BS (2003) Elevated levels of beta-catenin and fibronectin in three-dimensional collagen cultures of Dupuytren’s disease cells are regulated by tension in vitro. BMC Musculoskelet Disord 4:16
Hurst LC, Badalamente MA (1999) Nonoperative treatment of Dupuytren’s disease. Hand Clin 15:97–107
Hurst LC, Badalamente MA, Hentz VR, Hotchkiss RN, Kaplan FT, Meals RA, Smith TM, Rodzvilla J, CORD I Study Group (2009) Injectable collagenase Clostridium histolyticum for Dupuytren’s contracture. N Engl J Med 361:968–979
Kobus K, Wojcicki P, Dydymski T, Wegrzyn M, Hamlawi F (2007) Evaluation of treatment results of patients with Dupuytren’s contracture—our clinical experience. Ortop Traumatol Rehabil 9:134–140
Koulikovska M, Podskochy A, Fagerholm P (2005) The expression pattern of the subunit of chaperonin containing T-complex polypeptide 1 and its substrate, alpha-smooth muscle actin, during corneal wound healing. Acta Opthalmol Scand 83:543–548
Kubota H, Hynes G, Carne A, Ashworth A, Willison K (1994) Identification of six Tcp-1-related genes encoding divergent subunits of the TCP-1-containing chaperonin. Curr Biol 4:89–99
Kubota H, Hynes G, Willison K (1995) The chaperonin containing T-complex polypeptide 1 (TCP-1). Multisubunit machinery assisting in protein folding and assembly in the eukaryotic cytosol. Eur J Biochem 230:3–16, Review
Makela EA, Jaroma H, Harju A, Anttila S, Vainio J (1991) Dupuytren’s contracture: the long-term results after day surgery. J Hand Surg 16B:272–274
McFarlane RM (1974) Patterns of diseased fascia in the fingers of Dupuytren’s contracture: displacement of the neurovascular bundle. Plast Reconstr Surg 54:31–44
McFarlane RM (2002) On the origin and spread of Dupuytren’s disease. J Hand Surg Am 27:385–390
Michou L, Lermusiaux JL, Teyssedou JP, Bardin T, Beaudreuil J, Petit-Teixeira E (2012) Genetics of Dupuytren’s disease. Joint Bone Spine 79:7–12
Rodrigo JJ, Niebauer JJ, Brown RL, Doyle JR (1976) Treatment of Dupuytren’s contracture. Long-term results after fasciotomy and fascial excision. J Bone Joint Surg 58A:380–387
Satish L, Abdulally A, Oswald D, Johnson S, Hu FZ, Post JC, Ehrlich GD, Kathju S (2008) Differential expression of chaperonin containing T-complex polypeptide (CCT) subunits during fetal and adult skin wound healing. Cell Stress Chaperones 13:527–533
Satish L, Johnson S, Abdulally A, Post JC, Ehrlich GD, Kathju S (2010a) Cloning and expression of rabbit CCT subunits eta and beta in healing cutaneous wounds. Cell Stress Chaperones 15:819–826
Satish L, Johnson S, Wang JH, Post JC, Ehrlich GD, Kathju S (2010b) Chaperonin containing T-complex polypeptide subunit eta (CCT-eta) is a specific regulator of fibroblast motility and contractility. PLoS One 20:e10063
Satish L, Gallo PH, Baratz ME, Johnson S, Kathju S (2011) Reversal of TGF-beta1 stimulation of alpha-smooth muscle actin and extracellular matrix components by cyclic AMP in Dupuytren’s-derived fibroblasts. BMC Musculoskeletal Disord 12:113
Satish L, LaFramboise WA, Johnson S, Vi L, Njarlangattil A, Raykha C, Krill-Burger JM, Gallo PH, O’Gorman DB, Gan BS, Baratz ME, Ehrlich GD, Kathju S (2012) Fibroblasts from phenotypically normal palmar fascia exhibit molecular profiles highly similar to fibroblasts from active disease in Dupuytren’s contracture. BMC Med Genomics 5:15
Shaw RB Jr, Chong AK, Zhang A, Hentz VR, Chang J (2007) Dupuytren’s disease: history, diagnosis, and treatment. Plast Reconstr Surg 120:44e–54e
Tomasek J, Rayan GM (1995) Correlation of alpha-smooth muscle actin expression and contraction in Dupuytren’s disease fibroblasts. J Hand Surg Am 20:450–455
Tonkin MA, Burke FD, Varian JP (1985) The proximal interphalangeal joint in Dupuytren’s disease. J Hand Surg 10B:358–364
Tse R, Howard J, Wu Y, Gan BS (2004) Enhanced Dupuytren’s disease fibroblast populated collagen lattice contraction is independent of endogenous active TGF-beta2. BMC Musculoskelet Disord 12:41
Verjee LS, Midwood K, Davidson D, Essex D, Sandison A, Nanchahal J (2009) Myofibroblast distribution in Dupuytren’s cords: correlation with digital contracture. J Hand Surg Am 34:1785–1794
Verjee LS, Midwood K, Davidson D, Eastwood M, Nanchahal J (2010) Post-transcriptional regulation of alpha-smooth muscle actin determines the contractile phenotype of Dupuytren’s nodular cells. J Cell Physiol 224:681–690
Wang XQ, Kravchuk O, Winterford C, Kimble RM (2011) The correlation of in vivo burn scar contraction with the level of alpha-smooth muscle actin expression. Burns 37:1367–1377
Watt AJ, Curtin CM, Hentz VR (2010) Collagenase injection as nonsurgical treatment of Dupuytren’s disease: 8-year follow up. J Hand Surg Am 35:534–539
Yang ZC, Chen JX, Lin J-S, Wang JH-C (2006) Determining substrate displacement and cell traction fields—a new approach. J Theor Biol 242:607–616
Zhang AY, Curtin CM, Hentz VR (2011) Flexor tendon rupture after collagenase injection for dupuytren contracture: case report. J Hand Surg Am 36:1323–1325
Acknowledgments
We acknowledge the support of the Department of Plastic Surgery at the University of Pittsburgh Medical Center, and the Allegheny-Singer Research Institute. We gratefully acknowledge the funding support from NIH (AR049921 and AR06139) for this work (JHW). We would also like to thank Yan Wu, Department of Biochemistry, University of Western Ontario, for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Satish, L., O’Gorman, D.B., Johnson, S. et al. Increased CCT-eta expression is a marker of latent and active disease and a modulator of fibroblast contractility in Dupuytren’s contracture. Cell Stress and Chaperones 18, 397–404 (2013). https://doi.org/10.1007/s12192-012-0392-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-012-0392-9