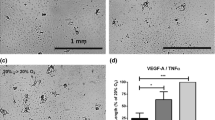
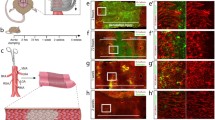
Abstract
A genome-wide microarray analysis of gene expression was carried out on human microvascular endothelial cells (HMEC-1) exposed to hyperbaric oxygen treatment (HBOT) under conditions that approximated clinical settings. Highly up-regulated genes included immediate early transcription factors (FOS, FOSB, and JUNB) and metallothioneins. Six molecular chaperones were also up-regulated immediately following HBOT, and all of these have been implicated in protein damage control. Pathway analysis programs identified the Nrf-2-mediated oxidative stress response as one of the primary responders to HBOT. Several of the microarray changes in the Nrf2 pathway and a molecular chaperone were validated using quantitative PCR. For all of the genes tested (Nrf2, HMOX1, HSPA1A, M1A, ACTC1, and FOS), HBOT elicited large responses, whereas changes were minimal following treatment with 100% O2 in the absence of elevated pressure. The increased expression of immediate early and cytoprotective genes corresponded with an HBOT-induced increase in cell proliferation and oxidative stress resistance. In addition, HBOT treatment enhanced endothelial tube formation on Matrigel plates, with particularly dramatic effects observed following two daily HBO treatments. Understanding how HBOT influences gene expression changes in endothelial cells may be beneficial for improving current HBOT-based wound-healing protocols. These data also point to other potential HBOT applications where stimulating protection and repair of the endothelium would be beneficial, such as patient preconditioning prior to major surgery.





Similar content being viewed by others
References
Ades EW, Candal FJ, Swerlick RA, George VG, Summers S, Bosse DC, Lawley TJ (1992) HMEC-1: establishment of an immortalized human microvascular endothelial cell line. J Invest Dermatol 99:683–690
Alex J, Laden G, Cale AR et al (2005) Pretreatment with hyperbaric oxygen and its effect on neuropsychometric dysfunction and systemic inflammatory response after cardiopulmonary bypass: a prospective randomized double-blind trial. J Thorac Cardiovasc Surg 130:1623–1630
Al-Waili NS, Butler GJ (2006) Effects of hyperbaric oxygen on inflammatory response to wound and trauma: possible mechanism of action. ScientificWorldJournal 6:425–441
Asakuno K, Isono M, Wakabayashi Y, Mori T, Hori S, Kohno K, Kuwano M (1995) The exogenous control of transfected c-fos gene expression and angiogenesis in cells implanted into the rat brain. Brain Res 702:23–31
Bell SG, Vallee BL (2009) The metallothionein/thionein system: an oxidoreductive metabolic zinc link. ChemBioChem 10:55–62
Cypser JR, Johnson TE (2002) Multiple stressors in Caenorhabditis elegans induce stress hormesis and extended longevity. J Gerontol A Biol Sci Med Sci 57:B109–B114
Du P, Kibbe WA, Lin SM (2008) lumi: a pipeline for processing Illumina microarray. Bioinformatics 24:1547–1548
Duzgun AP, Satir HZ, Ozozan O, Saylam B, Kulah B, Coskun F (2008) Effect of hyperbaric oxygen therapy on healing of diabetic foot ulcers. J Foot Ankle Surg 47:515–519
Efrati S, Gall N, Bergan J et al (2009) Hyperbaric oxygen, oxidative stress, NO bioavailability and ulcer oxygenation in diabetic patients. Undersea Hyperb Med 36:1–12
Gesell LB (2008) Hyperbaric oxygen therapy indications, 12th edn. Undersea and Hyperbaric Medical Society, Durham
Godman CA, Joshi R, Tierney BR et al (2008) HDAC3 impacts multiple oncogenic pathways in colon cancer cells with effects on Wnt and vitamin D signaling. Cancer Biol Ther 7:1570–1580
Gurdol F, Cimsit M, Oner-Iyidogan Y, Kocak H, Sengun S, Yalcinkaya-Demirsoz S (2009) Collagen synthesis, nitric oxide and asymmetric dimethylarginine in diabetic subjects undergoing hyperbaric oxygen therapy. Physiol Res 2009 Aug 12 (Epub ahead of print)
Hageman J (2008) The human HSP70/HSP40 chaperone family: a study on its capacity to combat proteotoxic stress. PhD thesis, Department of Cell Biology, University Medical Center Groningen, Groningen, p. 188
Ihaka R, Gentleman R (1996) R: a language for data analysis and graphics. J Comput Graph Stat 5:16
Li Q, Li J, Zhang L, Wang B, Xiong L (2007) Preconditioning with hyperbaric oxygen induces tolerance against oxidative injury via increased expression of heme oxygenase-1 in primary cultured spinal cord neurons. Life Sci 80:1087–1093
Liu ZJ, Velazquez OC (2008) Hyperoxia, endothelial progenitor cell mobilization, and diabetic wound healing. Antioxid Redox Signal 10:1869–1882
Milovanova TN, Bhopale VM, Sorokina EM, Moore JS, Hunt TK, Hauer-Jensen M, Velazquez OC, Thom SR (2009) Hyperbaric oxygen stimulates vasculogenic stem cell growth and differentiation in vivo. J Appl Physiol 106:711–728
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284:13291–13295
Ohtsuji M, Katsuoka F, Kobayashi A, Aburatani H, Hayes JD, Yamamoto M (2008) Nrf1 and Nrf2 play distinct roles in activation of antioxidant response element-dependent genes. J Biol Chem 283:33554–33562
Ong M (2008) Hyperbaric oxygen therapy in the management of diabetic lower limb wounds. Singapore Med J 49:105–109
Osburn WO, Kensler TW (2008) Nrf2 signaling: an adaptive response pathway for protection against environmental toxic insults. Mutat Res 659:31–39
Pahl HL, Baeuerle PA (1994) Oxygen and the control of gene expression. Bioessays 16:497–502
Ren P, Kang Z, Gu G et al (2008) Hyperbaric oxygen preconditioning promotes angiogenesis in rat liver after partial hepatectomy. Life Sci 83:236–241
Rothfuss A, Speit G (2002) Investigations on the mechanism of hyperbaric oxygen (HBO)-induced adaptive protection against oxidative stress. Mutat Res 508:157–165
Ryter SW, Alam J, Choi AM (2006) Heme oxygenase-1/carbon monoxide: from basic science to therapeutic applications. Physiol Rev 86:583–650
Sander AL, Henrich D, Muth CM, Marzi I, Barker JH, Frank JM (2009) In vivo effect of hyperbaric oxygen on wound angiogenesis and epithelialization. Wound Repair Regen 17:179–184
Schroder M, Kaufman RJ (2005) The mammalian unfolded protein response. Annu Rev Biochem 74:739–789
Shono T, Ono M, Izumi H, Jimi SI, Matsushima K, Okamoto T, Kohno K, Kuwano M (1996) Involvement of the transcription factor NF-kappaB in tubular morphogenesis of human microvascular endothelial cells by oxidative stress. Mol Cell Biol 16:4231–4239
Speit G, Dennog C, Eichhorn U, Rothfuss A, Kaina B (2000) Induction of heme oxygenase-1 and adaptive protection against the induction of DNA damage after hyperbaric oxygen treatment. Carcinogenesis 21:1795–1799
Suh JH, Shenvi SV, Dixon BM, Liu H, Jaiswal AK, Liu RM, Hagen TM (2004) Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc Natl Acad Sci U S A 101:3381–3386
Thackham JA, McElwain DL, Long RJ (2008) The use of hyperbaric oxygen therapy to treat chronic wounds: A review. Wound Repair Regen 16:321–330
Theodorakis NG, Drujan D, De Maio A (1999) Thermotolerant cells show an attenuated expression of Hsp70 after heat shock. J Biol Chem 274:12081–12086
Thom SR (2009) Oxidative stress is fundamental to hyperbaric oxygen therapy. J Appl Physiol 106:988–995
Yuan J, Handy RD, Moody AJ, Bryson P (2009) Response of blood vessels in vitro to hyperbaric oxygen (HBO): modulation of VEGF and NO(x) release by external lactate or arginine. Biochim Biophys Acta 1787:828–834
Yura T, Guisbert E, Poritz M, Lu CZ, Campbell E, Gross CA (2007) Analysis of sigma32 mutants defective in chaperone-mediated feedback control reveals unexpected complexity of the heat shock response. Proc Natl Acad Sci U S A 104:17638–17643
Zakkar M, Van der Heiden K, Luong LA et al (2009) Activation of Nrf2 in endothelial cells protects arteries from exhibiting a proinflammatory state. Arterioscler Thromb Vasc Biol 29:1851–1857
Zhang Q, Andersen ME (2007) Dose response relationship in anti-stress gene regulatory networks. PLoS Comput Biol 3:e24
Zhang Q, Chang Q, Cox RA, Gong X, Gould LJ (2008) Hyperbaric oxygen attenuates apoptosis and decreases inflammation in an ischemic wound model. J Invest Dermatol 128:2102–2112
Acknowledgements
We would like to thank OxyHeal Health Group for providing the funding and hyperbaric chamber for these studies. We would also like to extend our gratitude to Alexandria Thornton and Bryan Lawlor for their work during the early stages of the project. We would like to thank Advanced Turbine Services, LLC for donating some of the equipment used in this work. For technical support, we thank Dr. Carol Norris, Facility Scientist at the University of Connecticut FCCM Facility.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Gene list of biological functions for HBOT0 (XLS 50 kb)
Supplementary Table 2
Gene list of biological functions for HBOT24 (XLS 99 kb)
Rights and permissions
About this article
Cite this article
Godman, C.A., Chheda, K.P., Hightower, L.E. et al. Hyperbaric oxygen induces a cytoprotective and angiogenic response in human microvascular endothelial cells. Cell Stress and Chaperones 15, 431–442 (2010). https://doi.org/10.1007/s12192-009-0159-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-009-0159-0