Abstract
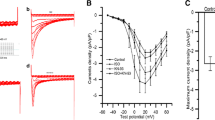
The L-type calcium channel plays a pivotal role in the regulation of a wide range of cellular processes, including membrane excitability, Ca2+ homeostasis, protein phosphorylation, and gene regulation. Alterations in the density or function of the L-type calcium channel have been implicated in a variety of cardiovascular diseases. Our previous study found that acute restraint stress could cause an enhancement of the L-type calcium current (I Ca-L), which correlated with an up-regulation of activation characters of the calcium channel. In this study, we observed the change of I Ca-L in rat ventricular myocytes under chronic restraint stress using the whole-cell patch-clamp technique and further explored its modulation mechanisms. The results showed that chronic restraint stress could also enhance I Ca-L, but increased I Ca-L was not accompanied by an alteration of the characteristics of activation and inactivation of the L-type calcium channel. Furthermore, results from reverse-transcription polymerase chain reaction and Northern blot showed that the abundance of α1c subunit messenger RNA of the L-type calcium channel in the ventricle was increased significantly after chronic stress, and Western blot analysis revealed the amount of α1c subunit protein also was elevated. These results suggest that the L-type calcium channel is involved in stress-induced cardiomyocyte injury, and the up-regulated expression of the L-type calcium channel α1c subunit might contribute to the I Ca-L change under chronic stress, which is different from the regulation mechanism of acute restraint stress that mostly relates to an alteration in protein kinase A-dependent channel activation. Thus, it would provide a new insight into the mechanism of cardiomyocyte injury induced by stress.




Similar content being viewed by others
Abbreviations
- I Ca-L :
-
L-type calcium current
- RT-PCR:
-
reverse-transcription polymerase chain reaction
- PKA/C:
-
protein kinase A /C
- [Ca2+]i :
-
intracellular calcium
- ER:
-
endoplasmic reticulum
- NE:
-
norepinephrine
References
Aggarwal R, Boyden PA (1995) Diminished calcium and barium currents in myocytes surviving in the epicardial border zone of the 5-day infracted canine heart. Circ Res 77:1180–1191
Balke CW, Shorofsky SR (1998) Alterations in calcium handling in cardiac hypertrophy and heart failure. Cardiovasc Res 37:290–299
Bean BP (1985) Two kinds of calcium channels in canine atrial cells. J Gen Physiol 86:1–30
Berridge MJ, Lipp P, Bootman MD (2000) The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol 1:11–21
Bers DM (1991) Ca influx via sarcolemmal Ca channels. In: Bers DM (ed) Excitation-contraction coupling and cardiac contractile force. Kluwer, Norwell, MA, pp 49–70
Bers DM (2002) Cardiac excitation–contraction coupling. Nature 415:198–205
Bodi I, Mikala G, Koch SE, Akhter SA, Schwartz A (2005) The L-type calcium channel in the heart: the beat goes on. J Clin Invest 115:3306–3317
Brosenitsch TA, Salgado Commissariat D, Kunze DL, Katz DM (1998) A role for L-type calcium channels in developmental regulation of transmitter phenotype in primary sensory neurons. Neurosci 18:1047–1055
Campbell DL, Strauss HC (1995) Regulation of calcium channels in the heart. Adv Second Messenger Phosphoprot Res 30:25–88
Catterall WA (1995) Structure and function of voltage-gated ion channels. Annu Rev Biochem 64:493–501
Catterall WA (2000) Structure and regulation of voltage-gated Ca2+ channels. Annu Rev Cell Dev Biol 16:521–555
Christ T, Boknik P, Wöhrl S et al (2004) L-type Ca2+ current downregulation in chronic human atrial fibrillation is associated with increased activity of protein phosphatases. Circulation 110:2651–2657
Communal C, Singh K, Pimentel DR, Colucci WS (1998) Norepinephrine stimulates apoptosis in adult rat ventricular myocytes by activation of the beta-adrenergic pathway. Circulation 98:1329–1334
Das DK, Engelman RM, Maulik N (1999) Oxygen free radical signaling in ischemic preconditioning. In: Das DK (ed) Heart in stress . Ann NY Acad Sci 874:49–65
Demaurex N, Distelhorst C (2003) Apoptosis—the calcium connection. Science 300:65–67
Feuerstein GZ, Young PR (2000) Apoptosis in cardiac diseases: stress- and mitogen-activated signaling pathways. Cardiovasc Res 45:560–569
Galea LAM, Mcewen BS, Tanapat P et al (1997) Sex differences in dendritic atrophy of CA3 pyramidal neurons in response to chronic restraint stress. Neurosci 81:689–697
Gao F, Gong B, Christopher TA, Lopez BL, Karasawa A, Ma XL (2001) Anti-apoptotic effect of benidipine, a long-lasting vasodilating calcium antagonist, in ischaemic/reperfused myocardial cells. Br J Pharmacol 132:869–878
Goldenberg I, Grossman E, Jacobson KA, Shneyvays V, Shainberg A (2001) Angiotensin II-induced apoptosis in rat cardiomyocyte culture: a possible role of AT1 and AT2 receptors. J Hypertens 19:1681–1689
Hajnoczky G, Csordas G, Madesh M, Pacher P (2000) Control of apoptosis by IP3 and ryanodine receptor driven calcium signals. Cell Calcium 28:349–363
Herzig S, Neumann J (2000) Effects of serine/threonine protein phosphatases on ion channels in excitable membranes. Physiol Rev 80:173–210
Isenberg G, Klockner U (1982) Calcium tolerant ventricular myocytes prepared by preincubation in a “KB” medium. Pflugers Arch 395:6–18
Kamp TJ, Hell JW (2000) Regulation of L-type calcium channels by protein kinase A and protein kinase C. Circ Res 22:1095–1102
Klockner U, Isenberg G (1994) Intracellular pH Modulates the Availability of Vascular L-type Ca2+ Channels. J Gen Physiol 103:647–663
Marty A, Neher E (1995) Tight-seal whole-cell recording. In: Sakmann B, Neher E (eds) Single-channel recording, 2nd edn. Plenum, New York, pp 31–52
Moosmang S, Lenhardt P, Haider N, Hofmann F, Wegener JW (2005) Mouse models to study L-type calcium channel function. Pharmacol Ther 106:347–355
Mukherjee R, Spinale FG (1998) L-type calcium channel abundance and function with cardiac hypertrophy and failure: A review. J Mol Cell Cardiol 30:1899–1916
Muth JN, Bodi I, Lewis W, Varadi G, Schwartz A (2001) A Ca2+-dependent transgenic model of cardiac hypertrophy: A role for protein kinase Calpha. Circulation 103:140–147
Schultz D, Mikala G, Yatani A et al (1993) Cloning, chromosomal localization, and functional expression of the 1 subunit of the L-type voltage-dependent calcium channel from normal human heart. Proc Natl Acad Sci USA 90:6228–6232
Schwartz AR, Gerin W, Davidson KW, Picking TG, Brosschot JF, Thayer JF, Christenfeld N, Linden W (2003) Toward a causal model of cardiovascular responses to stress and the development of cardiovascular disease. Psychosom Med 65:22–35
Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T, Korsmeyer SJ (2003) BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science 300:135–159
Timothy JK, He JQ (2002) L-type Ca2+ channels gaining respect in heart failure. Cardiovasc Res 91:451–453
Trump BF, Berezesky IK (1995) Calcium-mediated cell injury and cell death. FASEB 9:219–228
Van Wagoner DR, Pond AL, Lamorgese M, Rossie SS, McCarthy PM, Nerbonne JM (1999) Atrial L-type Ca2+ currents and human atrial fibrillation. Circ Res 85:428–436
Xu J, Ma Q, Duan HF, Qian LJ (2003) Effects of stress on L-type calcium channels of rat ventricular myocytes. Chin J Applied Physiol 19:216–219
Yue L, Feng J, Gaspo R, Li GR, Wang Z, Nattel S (1997) Ionic remodeling underlying action potential changes in a canine model of atrial fibrillation. Circ Res 81:512–525
Zhao Y, Wang WY, Qian LJ (2007) Hsp70 may protect cardiomyocytes from stress -induced injury by inhibiting Fas-mediated apoptosis. Cell Stress Chaperones 12:83–95
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (NNSFC; 30430590) and the National High Technology Research Development Program of China (2006AA02Z4C8).
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Zhao and J. Xu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, Y., Xu, J., Gong, J. et al. L-type calcium channel current up-regulation by chronic stress is associated with increased α1c subunit expression in rat ventricular myocytes. Cell Stress and Chaperones 14, 33–41 (2009). https://doi.org/10.1007/s12192-008-0052-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-008-0052-2