Abstract
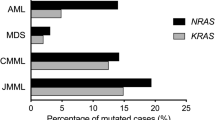
Mutations in RAS are frequent in acute myeloid leukemia (AML), and are thought to contribute to leukemogenesis in a subset of patients; however, their prognostic significance has not been firmly established. One hundred and fifty-seven pediatric patients with AML were analyzed for NRAS and KRAS mutations around hot spots at codons 12, 13, and 61. Twenty-nine patients (18.5%) had an activating mutation of RAS. We found KRAS mutations to be more frequent than NRAS mutations (18/29, 62.1% of patients with RAS mutation), in contrast to previous reports (18–40%). The frequency of RAS mutation was higher in French-American-British types M4 and M5 than other types (P = 0.02). There were no significant differences in other clinical manifestations or distribution in cytogenetic subgroups, or aberrations of other genes, including KIT mutation, FLT3-ITD, and MLL-PTD, between patients with and without RAS mutations. No significant differences were observed in the 3-year overall survival and disease-free survival; however, the presence of RAS mutation was related to late relapse. The occurrence of clinical events at relatively late period should be monitored for in AML patients with mutations in RAS.


Similar content being viewed by others
References
Gibson BE, Wheatley K, Hann IM, Stevens RF, Webb D, Hills RK, et al. Treatment strategy and long-term results in paediatric patients treated in consecutive UK AML trials. Leukemia. 2005;19:2130–8.
Lie SO, Abrahamsson J, Clausen N, Forestier E, Hasle H, Hovi L, et al. Long term results in children with AML: NOPHO-AML Study Group—report of three consecutive trials. Leukemia. 2005;19:2090–100.
Creutzig U, Zimmermann M, Lehrnbecher T, Graf N, Hermann J, Niemeyer CM, et al. Less toxicity by optimizing chemotherapy, but not by addition of granulocyte colony-stimulating factor in children and adolescents with acute myeloid leukemia: results of AML-BFM 98. J Clin Oncol. 2006;24:4499–506.
Lange BJ, Smith FO, Feusner J, Barnard DR, Dinndorf P, Feig S, et al. Outcomes in CCG-2961, a children’s oncology group phase 3 trial for untreated pediatric acute myeloid leukemia: a report from the children’s oncology group. Blood. 2008;111:1044–53.
Tsukimoto I, Tawa A, Horibe K, Tabuchi K, Kigasawa H, Tsuchida M, et al. Risk-stratified therapy and the intensive use of cytarabine improves the outcome in childhood acute myeloid leukemia: The AML99 trial from the Japanese Childhood AML Cooperative Study Group. J Clin Oncol. 2009;27:4007–13.
Rubnitz JE, Inaba H, Dahl G, Ribeiro RC, Bowman WP, Taub J, et al. Minimal residual disease-directed therapy for childhood acute myeloid leukaemia: results of the AML 02 multicentre trial. Lancet Oncol. 2010;11:543–52.
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G, et al. The importance of diagnostic cytogenetics on outcome of AML: analysis of 1612 patients entered into the MRC AML 10 trial. The Medical Research Council Adult and Children’s Leukaemia Working Parties. Blood. 1998;92:2322–33.
Speck NA, Gilliland DG. Core binding factors in haematopoiesis and leukaemia. Nat Rev Cancer. 2002;2:502–13.
Kelly LM, Kutok JL, Williams IR, Boulton CL, Amaral SM, Curley DP, et al. PML/RARalpha and FLT3-ITD induce an APL-like disease in a mouse model. Proc Natl Acad Sci USA. 2002;99:8283–8.
Grisolano JL, O’Neal J, Cain J, Tomasson MH. An activated receptor tyrosine kinase, TEL/PDGFbetaR, cooperates with AML1/ETO to induce acute myeloid leukemia in mice. Proc Natl Acad Sci USA. 2003;100:9506–11.
Chan IT, Gilliland DG. Oncogenic K-ras in mouse model of myeloproliferative disease and acute myeloid leukemia. Cell Cycle. 2004;3:536–7.
Janssen JW, Steenvoorden AC, Collard JG, Nusse R. Oncogene activation in human myeloid leukemia. Cancer Res. 1985;45:3262–7.
Kitayama H, Tsujimura T, Matsumura I, Oritani K, Ikeda H, Ishikawa J, et al. Neoplastic transformation of normal hematopoietic cells by constitutively activating mutations of c-kit receptor tyrosine kinase. Blood. 1996;88:995–1004.
Hayakawa F, Towatari M, Kiyoi H, Tanimoto M, Kitamura T, Saito H, et al. Tandem-duplicated Flt3 constitutively activates STAT5 and MAP kinase and introduces autonomous cell growth in IL-3-dependent cell lines. Oncogene. 2000;19:624–31.
Bos JL. ras oncogenes in human cancer: a review. Cancer Res. 1989;49:4682–9.
Radich JP, Kopecky KJ, Willman CL, Weick J, Head D, Appelbaum F, et al. N-ras mutations in adult de novo acute myelogenous leukemia: prevalence and clinical significance. Blood. 1990;76:801–7.
Neubauer A, Dodge RK, George SL, Davey FR, Silver RT, Schiffer CA, et al. Prognostic importance of mutations in the ras proto-oncogenes in de novo acute myeloid leukemia. Blood. 1994;83:1603–11.
De Melo MB, Lorand-Metze I, Lima CS, Saad ST, Costa FF. N-RAS gene point mutations in Brazilian acute myelogenous leukemia patients correlate with a poor prognosis. Leuk Lymphoma. 1997;24:309–17.
Kiyoi H, Naoe T, Nakano Y, Yokota S, Minami S, Miyawaki S, et al. Prognostic implication of FLT3 and N-RAS gene mutations in acute myeloid leukemia. Blood. 1999;93:3074–80.
Meshinchi S, Stirewalt DL, Alonzo TA, Zhang Q, Sweetser DA, Woods WG, et al. Activating mutations of RTK/ras signal transduction pathway in pediatric acute myeloid leukemia. Blood. 2003;102:1474–9.
Stirewalt DL, Kopecky KJ, Meshinchi S, Appelbaum FR, Slovak ML, Willman CL, et al. FLT3, RAS, and TP53 mutations in elderly patients with acute myeloid leukemia. Blood. 2001;97:3589–95.
Ritter M, Kim TD, Lisske P, Thiede C, Schaich M, Neubauer A. Prognostic significance of N-RAS and K-RAS mutations in 232 patients with acute myeloid leukemia. Haematologica. 2004;89:1397–9.
Illmer T, Thiede C, Fredersdorf A, Stadler S, Neubauer A, Ehninger G, et al. Activation of the RAS pathway is predictive for a chemosensitive phenotype of acute myelogenous leukemia blasts. Clin Cancer Res. 2005;11:3217–24.
Bowen DT, Frew ME, Hills R, Gale RE, Wheatley K, Groves MJ, et al. RAS mutation in acute myeloid leukemia is associated with distinct cytogenetic subgroups but does not influence outcome in patients younger than 60 years. Blood. 2005;106:2113–9.
Goemans BF, Zwaan CM, Miller M, Zimmermann M, Harlow A, Meshinchi S, et al. Mutations in KIT and RAS are frequent events in pediatric core-binding factor acute myeloid leukemia. Leukemia. 2005;19:1536–42.
Bacher U, Haferlach T, Schoch C, Kern W, Schnittger S. Implications of NRAS mutations in AML: a study of 2502 patients. Blood. 2006;107:3847–53.
Coghlan DW, Morley AA, Matthews JP, Bishop JF. The incidence and prognostic significance of mutations in codon 13 of the N-RAS gene in acute myeloid leukemia. Leukemia. 1994;8:1682–7.
Shimada A, Taki T, Tabuchi K, Tawa A, Horibe K, Tsuchida M, et al. KIT mutations, and not FLT3 internal tandem duplication, are strongly associated with a poor prognosis in pediatric acute myeloid leukemia with t(8;21): a study of the Japanese Childhood AML Cooperative Study Group. Blood. 2006;107:1806–9.
Kobayashi R, Tawa A, Hanada R, Horibe K, Tsuchida M, Tsukimoto I. Extramedullary infiltration at diagnosis and prognosis in children with acute myeloid leukemia. Pediatr Blood Cancer. 2007;48:393–8.
Shimada A, Taki T, Tabuchi K, Taketani T, Hanada R, Tawa A, et al. Tandem duplications of MLL and FLT3 are correlated with poor prognoses in pediatric acute myeloid leukemia: a study of the Japanese Childhood AML Cooperative Study Group. Pediatr Blood Cancer. 2008;50:264–9.
Xu F, Taki T, Yang HW, Hanada R, Hongo T, Ohnishi H, et al. Tandem duplication of the FLT3 gene is found in acute lymphoblastic leukaemia as well as acute myeloid leukaemia but not in myelodysplastic syndrome or juvenile chronic myelogenous leukaemia in children. Br J Haematol. 1999;105:155–62.
Taketani T, Taki T, Sugita K, Furuichi Y, Ishii E, Hanada R, et al. FLT3 mutations in the activation loop of tyrosine kinase domain are frequently found in infant ALL with MLL rearrangements and pediatric ALL with hyperdiploidy. Blood. 2004;103:1085–8.
Kaplan E, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1957;53:457–81.
Anderson JR, Cain KC, Gelber RD. Analysis of survival by tumor response. J Clin Oncol. 1983;1:710–9.
Neubauer A, Maharry K, Mrózek K, Thiede C, Marcucci G, Paschka P, et al. Patients with acute myeloid leukemia and RAS mutations benefit most from postremission high-dose cytarabine: a Cancer and Leukemia Group B Study. J Clin Oncol. 2008;26:4603–9.
Vogelstein B, Civin CI, Preisinger AC, Krischer JP, Steuber P, Ravindranath Y, et al. RAS gene mutations in childhood acute myeloid leukemia: a Pediatric Oncology Group study. Genes Chromosomes Cancer. 1990;2:159–62.
Farr C, Gill R, Katz F, Gibbons B, Marshall CJ. Analysis of ras gene mutations in childhood myeloid leukemia. Br J Haematol. 1991;77:323–7.
Lübbert M, Mirro J Jr, Kitchingman G, McCormick F, Mertelsmann R, Herrmann F, et al. Prevalence of N-ras mutations in children with myelodysplastic syndrome and acute myeloid leukemia. Oncogene. 1992;7:263–8.
Sheng XM, Kawamura M, Ohnishi H, Ida K, Hanada R, Kojima S, et al. Mutations of the RAS genes in childhood acute myeloid leukemia, myelodysplastic syndrome and juvenile myelocytic leukemia. Leuk Res. 1997;21:697–701.
Mahgoub N, Parker RI, Hosler MR, Close P, Winick NJ, Masterson M, et al. RAS mutations in pediatric leukemias with MLL gene rearrangements. Genes Chromosomes Cancer. 1998;21:270–5.
Nakagawa T, Saitoh S, Imoto S, Itoh M, Tsutsumi M, Hikiji K, et al. Multiple point mutation of N-ras and K-ras oncogenes in myelodysplastic syndrome and acute myelogenous leukemia. Oncology. 1992;49:114–22.
Shih LY, Liang DC, Huang CF, Chang YT, Lai CL, Lin TH, et al. Cooperating mutations of receptor tyrosine kinases and Ras genes in childhood core-binding factor acute myeloid leukemia and a comparative analysis on paired diagnosis and relapse samples. Leukemia. 2008;22:303–7.
Darley RL, Burnett AK. Mutant RAS inhibits neutrophil but not macrophage differentiation and allows continued growth of neutrophil precursors. Exp Hematol. 1999;27:1599–608.
De Filippi P, Zecca M, Lisini D, Rosti V, Cagioni C, Carlo-Stella C, et al. Germline mutation of the NRAS gene maybe responsible for the development of juvenile myelomonocytic leukaemia. Br J Haematol. 2009;147:706–9.
Oliveira JB, Bidère N, Niemela JE, Zheng L, Sakai K, Nix CP, et al. NRAS mutation causes a human autoimmune lymphoproliferative disease. Proc Natl Acad Sci USA. 2007;104:8953–8.
Acknowledgments
Committee members of the Japanese Childhood AML Cooperative Study Group who contributed data to this study include Akira Morimoto, Department of Pediatrics, Kyoto Prefectural University of Medicine; Hiromasa Yabe, Department of Pediatrics, Tokai University School of Medicine; Kazuko Hamamoto, Department of Pediatrics, Hiroshima Red Cross Hospital; Shigeru Tsuchiya, Department of Pediatric Oncology, Institute of Development, Aging and Cancer, Tohoku University; Yuichi Akiyama, Department of Pediatrics, National Hospital Organization Kyoto Medical Center; Hisato Kigasawa, Department of Hematology, Kanagawa Children’s Medical Center; Akira Ohara, First Department of Pediatrics, Toho University School of Medicine; Hideki Nakayama, Department of Pediatrics, Hamanomachi Hospital; Kazuko Kudo, Department of Pediatrics, Nagoya University Graduate School of Medicine; and Masue Imaizumi, Department of Hematology/Oncology, Miyagi Prefectural Children’s Hospital. This work was supported by a grant for Cancer Research and a grant for Research on Children and Families from the Ministry of Health, Labour, and Welfare of Japan, a Grant-in-Aid for Scientific Research (B, C) and Exploratory Research from the Ministry of Education, Culture, Sports, Science, and Technology of Japan, and by a Research grant for Gunma Prefectural Hospitals.
Conflict of interest
There is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Sano, H., Shimada, A., Taki, T. et al. RAS mutations are frequent in FAB type M4 and M5 of acute myeloid leukemia, and related to late relapse: a study of the Japanese Childhood AML Cooperative Study Group. Int J Hematol 95, 509–515 (2012). https://doi.org/10.1007/s12185-012-1033-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-012-1033-x