Abstract
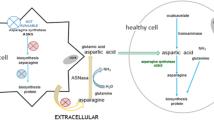
Patients with acute lymphoblastic leukemia (ALL), who develop antiasparaginase antibodies without clinical allergic reactions (“silent inactivation”) during l-asparaginase (l-Asp) treatment, have poor outcomes. Ammonia is produced by hydrolysis of asparagine by l-Asp. We postulated that plasma ammonia level might reflect the biological activity of l-Asp. Five children with ALL treated according to the Tokyo Children’s Cancer Study Group (TCCSG) protocol were enrolled. Plasma ammonia levels were analyzed immediately and 1 h after incubation at room temperature and “ex vivo ammonia production” was defined as increase in ammonia concentration. Ex vivo ammonia production well correlated with l-Asp activity (r = 0.882, P < 0.01, n = 23). It always exceeded 170 μg/dL (170–345 μg/dL) in induction therapy. We found 3 patients whose ammonia production was negligible during later phases of therapy. Antiasparaginase antibody was detected and l-Asp activity decreased in these patients. Ex vivo ammonia production is a surrogate marker of l-Asp biological activity.




Similar content being viewed by others
References
Muller HJ, Boos J. Use f l-asparaginase in childhood ALL. Crit Rev Oncol Hematol. 1998;28:97–113.
Woo MH, Hak LJ, Storm MC, Evans WE, SandLund JT, Rivera GK, et al. Anti-asparaginase antibodies following E. coli asparaginase therapy in pediatric acute lymphoblastic leukemia. Leukemia. 1998;12:1527–33.
Muller HJ, Beier R, Loning L, Bulteers-Sawatzki R, Dorffel W, Maass E, et al. Pharmacokinetics of native Escherichia coli asparaginase (Asparaginase medac) and hypersensitivity reactions in ALL-BFM 95 reinduction treatment. Br J Haematol. 2001;114:794–9.
Oettgen HF, Stephenson PA, Schwartz MK, et al. Toxicity of E. coli l-asparaginase in man. Cancer. 1970;25:253–78.
Woo MH, Hak LJ, Storm MC, SandLund JT, Ribeiro RC, Rivera GK, et al. Hypersensitivity or development of antibodies to asparagines does not impact treatment outcome of childhood acute lymphoblastic leukemia. J Clin Oncol. 2000;18:1525–32.
Cheung N-KV, Chau IY, Coccia PF. Antibody response to Escherichia coli l-asparaginase: prognostic significance and clinical utility of antibody measurement. Am J Pediatr Hematol Oncol. 1986;8:99–104.
Tsukimoto I, Matsui J, Iwashita H, Shigeta K, Suzuki H, Hashimoto T. Improved measurements of anti-l-asparaginase IgG antibody and its clinical applications. Jpn J Clin Hematol. 1992;33:9–24. (in Japanese).
Panosyan EH, et al. Asparaginase antibody and asparaginase activity in children with high-risk acute lymphoblastic leukemia children’s cancer group study CCG-1961. J Pediatr Hematol Oncol. 2004;26:217–26.
Laterza OF, Gerhardt G, Sokoll LJ. Measurement of plasma ammonia is affected in patients receiving asparaginase therapy. Clin Chem. 2003;49:1710–1.
Manabe A, Ohara A, Hasegawa D, Koh K, Saito T, Kiyokawa N, et al. Significance of the complete clearance of peripheral blast after 7 days of prednisolone treatment in children with acute lymphoblastic leukemia: the Tokyo Children’s Cancer Study Group Study L99-15. Haematologica. 2008;93:1155–60.
Rizzari C, Zucchetti M, Conter V, Diomede L, Bruno A, Gavazzi L, et al. l-Asparagine depletion and l-asparaginase activity in children with acute lymphoblastic leukemia receiving i.m. or i.v. Erwinia C. or E. coli l-asparaginase as first exposure. Ann Oncol. 2000;11:189–93.
Gentili D, Zucchetti M, Conter V, Masera G, D’Incalci M. Determination of l-asparagine in biological samples in the presence of l-asparaginase. J Chromatogr B Biomed Appl. 1994;657:47–52.
Tsurusawa M, Chin M, Iwai A, Nomura K, Maeda H, Taga T, et al. l-Asparagine depletion levels and l-asparaginase activity in plasma of children with acute lymphoblastic leukemia under asparaginase treatment. Cancer Chemother Pharmacol. 2004;53:204–8.
Avrramis VI, Panosyan EH. Pharmacokinetic/pharmacodynamic relationships of asparaginase formulations. Clin Pharmacokinet. 2005;44:367–93.
Zalewska-szewczyk B, Andrzejewski W, Mlynarski W, Jedrychowska-danska K, Witas H, Bodalski J. The anti-asparagines antibodies correlate with l-asparaginase activity and may affect outcome of childhood acute lymphoblastic leukemia. Leukemia Lymphoma. 2007;48:931–6.
Patel N, Krishnan S, Offman MN, Krol M, Moss CX, Leighton C, et al. A dyad of lymphoblastic lysosomal cysteine proteases degrades the antileukemic drug l-asparaginase. J Clin Invest. 2009;119:1964–73.
Steiner M, Attarbaschi A, Kastner U, Dworzak M, Haas OA, Gadner H, et al. Distinct fluctuations of ammonia levels during asparaginase therapy for childhood acute leukemia. Pediatr Blood Cancer. 2007;15:640–2.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Watanabe, S., Miyake, K., Ogawa, C. et al. The ex vivo production of ammonia predicts l-asparaginase biological activity in children with acute lymphoblastic leukemia. Int J Hematol 90, 347–352 (2009). https://doi.org/10.1007/s12185-009-0419-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-009-0419-x