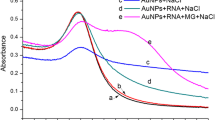
Abstract
To detect enrofloxacin (ENR) and ciprofloxacin (CIP) residues in fishery foods, a computer simulation was used to predict and analyze the binding sites existing aptamer specific to ENR and CIP. A new aptamer (SEQ.3) that can recognize ENR and CIP was screened and evaluated, and the dissociation constants (Kd) of SEQ.3 from ENR and CIP were determined to be 210.955 nM and 887.49 nM which are higher than other aptamers SEQ.4 and SEQ.5 by microscale thermophoresis (MST). Colorimetric detection method based on gold nanoparticles (AuNPs) was developed through optimization of temperature, incubation time, aptamer concentration, and other conditions. The results showed that the limit of defection (LOD) of the new aptamer to ENR and CIP were 1.89 nM and 6.84 nM, respectively, which were similar to that of the original aptamer. The recovery rate of the colorimetric method was between 87.8~102.18%. All the results implied that the developed colorimetric aptamer sensor has broad application prospects in food safety detection and environmental monitoring.





Similar content being viewed by others
References
Abnous K, Danesh NM, Alibolandi M, Ramezani M, Taghdisi SM, Emrani AS (2017) A novel electrochemical aptasensor for ultrasensitive detection of fluoroquinolones based on single-stranded DNA-binding protein. Sensors Actuators B Chem 240:100–106 https://www.sciencedirect.com/science/article/abs/pii/S0925400516313326
Alsager OA, Kumar S, Zhu B, Travas-Sejdic J, McNatty KP, Hodgkiss JM (2015) Ultrasensitive colorimetric detection of 17-estradiol: the effect of shortening DNA aptamer sequences. Anal Chem 87(8):4201–4209. https://doi.org/10.1021/acs.analchem.5b00335
Cai S, Yan J, Xiong H, Liu Y, Peng D, Liu Z (2018) Investigations on the interface of nucleic acid aptamers and binding targets. Analyst 143(22):5317–5338. https://doi.org/10.1039/c8an01467a
Cao Z, Wang S, Liu Z, Xue C, Mao X (2020) A rapid, easy, and sensitive method for detecting His-tag-containing chitinase based on ssDNA aptamers and gold nanoparticles. Food Chem 330(January):127230. https://doi.org/10.1016/j.foodchem.2020.127230
Chinnappan R, Rahamn AA, AlZabn R, Kamath S, Lopata AL, Abu-Salah KM, Zourob M (2020) Aptameric biosensor for the sensitive detection of major shrimp allergen, tropomyosin. Food Chem 314:126133. https://doi.org/10.1016/j.foodchem.2019.126133
Cui N, Cao L, Sui J, Lin H, Han X, Chen X, Xie H, Sun X (2020) Quick and convenient construction of lambda-cyhalothrin antigen for the generation of specific antibody. Anal Biochem 597(January):113669. https://doi.org/10.1016/j.ab.2020.113669
Dolati S, Ramezani M, Nabavinia MS, Soheili V, Abnous K, Taghdisi SM (2018) Selection of specific aptamer against enrofloxacin and fabrication of graphene oxide based label-free fluorescent assay. Anal Biochem 549(January):124–129. https://doi.org/10.1016/j.ab.2018.03.021
Du B, Wen F, Guo X, Zheng N, Zhang Y, Li S, Zhao S, Liu H, Meng L, Xu Q, Li M, Li F, Wang J (2019) Evaluation of an ELISA-based visualization microarray chip technique for the detection of veterinary antibiotics in milk. Food Control 106(April):106713. https://doi.org/10.1016/j.foodcont.2019.106713
Entzian C, Schubert T (2016) Studying small molecule-aptamer interactions using MicroScale Thermophoresis (MST). Methods 97:27–34. https://doi.org/10.1016/j.ymeth.2015.08.023
Guo X, Zhang L, Wang Z, Sun Y, Liu Q, Dong W, Hao A (2019) Fluorescent carbon dots based sensing system for detection of enrofloxacin in water solutions. Spectrochim Acta A Mol Biomol Spectrosc 219(February):15–22. https://doi.org/10.1016/j.saa.2019.02.017
Haiss W, Thanh NTK, Aveyard J, Fernig DG (2007) Determination of size and concentration of gold nanoparticles from UV-Vis spectra. Anal Chem 79(11):4215–4221. https://doi.org/10.1021/ac0702084
Heiat M, Najafi A, Ranjbar R, Latifi AM, Rasaee MJ (2016) Computational approach to analyze isolated ssDNA aptamers against angiotensin II. J Biotechnol 230:34–39. https://doi.org/10.1016/j.jbiotec.2016.05.021
Jia M, Sha J, Li Z, Wang W, Zhang H (2020) High affinity truncated aptamers for ultra-sensitive colorimetric detection of bisphenol A with label-free aptasensor. Food Chem 317(February):126459. https://doi.org/10.1016/j.foodchem.2020.126459
Kudłak B, Wieczerzak M (2020) Aptamer based tools for environmental and therapeutic monitoring: A review of developments, applications, future perspectives. Crit Rev Environ Sci Technol 50(8):816–867. https://doi.org/10.1080/10643389.2019.1634457
Lavaee P, Danesh NM, Ramezani M, Abnous K, Taghdisi SM (2017) Colorimetric aptamer based assay for the determination of fluoroquinolones by triggering the reduction-catalyzing activity of gold nanoparticles. Microchim Acta 184(7):2039–2045. https://doi.org/10.1007/s00604-017-2213-4
Liu X, He F, Zhang F, Zhang Z, Huang Z, Liu J (2020) Dopamine and melamine binding to gold nanoparticles dominates their aptamer-based label-free colorimetric sensing. Anal Chem 92:9370–9378. https://doi.org/10.1021/acs.analchem.0c01773
Mao Y, Fan T, Gysbers R, Tan Y, Liu F, Lin S, Jiang Y (2017) A simple and sensitive aptasensor for colorimetric detection of adenosine triphosphate based on unmodified gold nanoparticles. Talanta 168(February):279–285. https://doi.org/10.1016/j.talanta.2017.03.014
Marks HL, Pishko MV, Jackson GW, Coté GL (2014) Rational design of a bisphenol A aptamer selective surface-enhanced Raman scattering nanoprobe. Anal Chem 86(23):11614–11619. https://doi.org/10.1021/ac502541v
Mehlhorn A, Rahimi P, Joseph Y (2018) Aptamer-based biosensors for antibiotic detection: A review. Biosensors 8(2). https://doi.org/10.3390/bios8020054
Mirau PA, Smith JE, Chávez JL, Hagen JA, Kelley-Loughnane N, Naik R (2018) Structured DNA aptamer interactions with gold nanoparticles. Langmuir 34(5):2139–2146. https://doi.org/10.1021/acs.langmuir.7b02449
Ni H, Zhang S, Ding X, Mi T, Wang Z, Liu M (2014) Determination of enrofloxacin in bovine milk by a novel single-stranded DNA aptamer chemiluminescent enzyme immunoassay. Anal Lett 47(17):2844–2856. https://doi.org/10.1080/00032719.2014.924009
Qi X, Hu H, Yang Y, Piao Y (2018) Graphite nanoparticle as nanoquencher for 17β-estradiol detection using shortened aptamer sequence. Analyst 143(17):4163–4170. https://doi.org/10.1039/c8an00591e
Reinemann C, Freiin von Fritsch U, Rudolph S, Strehlitz B (2016) Generation and characterization of quinolone-specific DNA aptamers suitable for water monitoring. Biosens Bioelectron 77:1039–1047. https://doi.org/10.1016/j.bios.2015.10.069
Schax E, Lönne M, Scheper T, Belkin S, Walter JG (2015) Aptamer-based depletion of small molecular contaminants: a case study using ochratoxin A. Biotechnol Bioprocess Eng 20(6):1016–1025. https://doi.org/10.1007/s12257-015-0486-1
Soheili V, Taghdisi SM, Hassanzadeh Khayyat M, Fazly Bazzaz BBS, Ramezani M, Abnous K (2016) Colorimetric and ratiometric aggregation assay for streptomycin using gold nanoparticles and a new and highly specific aptamer. Microchim Acta 183(5):1687–1697. https://doi.org/10.1007/s00604-016-1798-3
Song SH, Gao ZF, Guo X, Chen GH (2019) Aptamer-based detection methodology studies in food safety. Food Anal Methods 2019(January):966–990. https://doi.org/10.1007/s12161-019-01437-3
Sun Y, Duan N, Ma P, Liang Y, Zhu X, Wang Z (2019) Colorimetric aptasensor based on truncated aptamer and trivalent DNAzyme for Vibrio parahemolyticus determination. J Agric Food Chem 67(8):2313–2320. https://doi.org/10.1021/acs.jafc.8b06893
Svobodová M, Skouridou V, Botero ML, Jauset-Rubio M, Schubert T, Bashammakh AS, El-Shahawi MS, Alyoubi AO, O’Sullivan CK (2017) The characterization and validation of 17β-estradiol binding aptamers. J Steroid Biochem Mol Biol 167:14–22. https://doi.org/10.1016/j.jsbmb.2016.09.018
Valenzano S, De Girolamo A, DeRosa MC, McKeague M, Schena R, Catucci L, Pascale M (2016) Screening and identification of DNA aptamers to tyramine using in vitro selection and high-throughput sequencing. ACS Comb Sci 18(6):302–313. https://doi.org/10.1021/acscombsci.5b00163
Wang S, Liu J, Dong Y, Su H, Tan T (2015) Conformational structure-dependent molecular recognition of two aptamers for tetracycline. RSC Adv 5(66):53796–53801. https://doi.org/10.1039/c5ra08050a
Wang S, Li W, Chang K, Liu J, Guo Q, Sun H, Jiang M, Zhang H, Chen J, Hu J (2017) Localized surface plasmon resonance-based abscisic acid biosensor using aptamer-functionalized gold nanoparticles. PLoS One 12(9):1–13. https://doi.org/10.1371/journal.pone.0185530
Yi H, Yan Z, Wang L, Zhou X, Yan R, Zhang D, Shen G, Zhou S (2019) Fluorometric determination for ofloxacin by using an aptamer and SYBR Green I. Microchim Acta 186(10). https://doi.org/10.1007/s00604-019-3788-8
Yuan M, Xiong Z, Fang B, Guo Z, Guo D, Lai W, Peng J (2020) Preparation of an antidanofloxacin monoclonal antibody and development of immunoassays for detecting danofloxacin in meat. ACS Omega 5:667–673. https://doi.org/10.1021/acsomega.9b03270
Funding
This study was supported by the Modern Agricultural Industry Technology System of China (grant numbers: CARS-47)
Author information
Authors and Affiliations
Contributions
Junyi Sha: methodology validation, formal analysis, investigation, writing–original draft. Hong Lin: conceptualization, methodology, supervision, writing–review and editing. Timira Vaileth: writing–review and editing. Jianxin Sui: conceptualization, methodology, supervision, writing–review and editing
Corresponding author
Ethics declarations
Conflict of Interest
Junyi Sha declares that she has no conflict of interest. Hong Lin declares that he has no conflict of interest. Vaileth Timira declares that she has no conflict of interest. Jianxin Sui declares that he has no conflict of interest
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sha, J., Lin, H., Timira, V. et al. The Construction and Application of Aptamer to Simultaneous Identification of Enrofloxacin and Ciprofloxacin Residues in Fish. Food Anal. Methods 14, 957–967 (2021). https://doi.org/10.1007/s12161-020-01937-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01937-7