Abstract
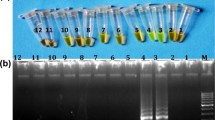
Meat fraud and adulteration is a growing concern in the marketplace today. Species identification in food product is among primary duties of food office control. In this study, a one-step, species-specific, sensitive, and rapid loop-mediated isothermal amplification (LAMP) assay was developed for identification of tissues of buffalo origin. A set of six LAMP primers were designed by targeting mitochondrial cyt b gene. The reaction temperature of LAMP and component of reaction mixture were optimized as 64 °C amplification temperature, 6 U/μL concentration of Bst DNA polymerase, 3 mM concentration of MgSO4, 0.4 mM concentration of dNTP, 0.6 M concentration of betaine, and 1:6 ratio of outer and inner primer pair. Successful amplifications were confirmed using SYBR Green I dye and agarose gel electrophoresis. The specificity of assay was tested with DNA of cattle, goat, sheep, and pig where amplification was observed exclusively in buffalo. The analytical sensitivity of the LAMP assay for buffalo DNA detection was 0.1 pg. The applicability check of developed assay carried out on known/coded samples and binary meat admixture substantiated the accuracy of assay. Supernatant of tissue exposed to Phire Animal Tissue Direct PCR master mix treatment was observed rich enough with DNA of target species and successfully amplified under optimized LAMP reaction. The comparison of results on the samples undergone pre-standardized species-specific PCR taken as gold standard shown complete match with developed species-specific LAMP assay. Hence, the developed species-specific LAMP assays are effective in detection of tissue of buffalo origin.





Similar content being viewed by others
References
Abdullahi UF, Igwenagu E, Aliyu S, Muazu A, Naim R, Wan-Taib WR (2017) A rapid and sensitive loop-mediated isothermal amplification assay for detection of pork DNA based on porcine tRNA lys and ATPase 8 genes. Int Food Res J 24(4):1357–1361 http://www.ifrj.upm.edu.my/24%20(04)%202017/(1).pdf
Arslan AI, Ilhak M, Calicioglu M, Karahan M (2005) Identification of meats using random amplified polymorphic DNA (RAPD) technique. J Muscle Foods 16(1):37–45. https://doi.org/10.1111/j.1745-4573.2004.07504.x
Cho AR, Dong HJ, Cho S (2014) Meat species identification using loop-mediated isothermal amplification assay targeting species-specific mitochondrial DNA. Korean J Food Sci Anim Resour 34(6):799–807. https://doi.org/10.5851/kosfa.2014.34.6.799
Chum PY, Haimes JD, Andre CP, Kuusisto PK, Kelley ML (2012) Genotyping of plant and animal samples without prior DNA purification. J Vis Exp 67:e3844
Dooley JJ, Paine KE, Garrett SD, Brown HM (2004) Detection of meat species using TaqMan real-time PCR assays. Meat Sci 68:431–438. https://doi.org/10.1016/j.meatsci.2004.04.010
Duan YB, Yang Y, Wang YX, Liu CC, He LL, Zhou MG (2015) Development and application of loop-mediated isothermal amplification for detecting the highly benzimidazole-resistant isolates in Sclerotinia sclerotiorum. Sci Rep 5:17278. https://doi.org/10.1038/srep17278
Giordano G, Guarini P, Ferrari P, Biondi-Zoccai G, Schiavone B, Giordano A (2010) Beneficial impact on cardiovascular risk profile of water buffalo meat consumption. Eur J Clin Nutr 64(9):1000–1006. https://doi.org/10.1038/ejcn.2010.108
Girish PS, Anjaneyulu ASR, Viswas KN, Anand M, Rajkumar N, Shivakumar BM, Sharma B (2004) Sequence analysis of mitochondrial 12S rRNA gene can identify meat species. Meat Sci 66(3):551–556. https://doi.org/10.1016/S0309-1740(03)00158-X
Haider N, Nabulsi I, Al-Safadi B (2012) Identification of meat species by PCR-RFLP of the mitochondrial COI gene. Meat Sci 90(2):490–493. https://doi.org/10.1016/j.meatsci.2011.09.013
Jonker KM, Tilburg JJ, Hägele GH, Boer E (2008) Species identification in meat products using real-time PCR. Food Addit Contam 25(5):527–533. https://doi.org/10.1080/02652030701584041
Karabasanavar NS, Singh SP, Umapathi V, Kumar D, Patil G, Shebannavar SN (2011) A highly specific PCR assay for identification of raw and heat treated mutton (Ovis aries). Small Rumin Res 100(2):153–158. https://doi.org/10.1016/j.smallrumres.2011.07.009
Koh BRD, Kim JY, Na HM, Park SD, Kim YH (2011) Development of species-specific multiplex PCR assays of mitochondrial 12S rRNA and 16S rRNA for the identification of animal species. Korean J Vet Serv 34:417–428. https://doi.org/10.7853/kjvs.2011.34.4.417
Kumar A, Kumar RR, Sharma BD, Mendiratta SK, Sharma D, Gokulakrishnan P (2012) Species specific polymerase chain reaction (PCR) assay for identification of pig (Sus domesticus) meat. Afr J Biotechnol 11(89):15590–15595. https://doi.org/10.5897/AJB12.1753
Kumar A, Kumar RR, Sharma BD, Gokulakrishnan P, Mendiratta SK, Sharma D (2015a) Identification of species origin of meat and meat products on the DNA basis: a review. Crit Rev Food Sci Nutr 55(10):1340–1351. https://doi.org/10.1080/10408398.2012.693978
Kumar A, Kumar RR, Sharma BD, Mendiratta SK, Gokulakrishnan P, Kumar D, Sharma D (2015b) Authentication of goat (Capra hircus) meat using PCR amplification of mitochondrial cytochrome b gene. Small Rumin Res 131:17–20. https://doi.org/10.1016/j.smallrumres.2015.07.016
Kumar D, Kumar RR, Kumar A, Mendiratta SK, Lalawampuii H, Kumbhar V, Choudhary A, Kumari S, Rana P (2017) A PCR Assay for Identification of Buffalo Origin of Tissue by Amplification of the mt D-loop. Gene J Anim Res 7(1):7–14. https://doi.org/10.5958/2277-940X.2017.00002.X
Mane BG, Mendiratta SK, Tiwari AK, Bhilegaokar KN (2012) Detection of adulteration of meat and meat products with buffalo meat employing polymerase chain reaction assay. Food Anal Methods 5(2):296–300. https://doi.org/10.1007/s12161-011-9237-x
Nagamine KTH, Notomi T (2002) Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol Cell Probes 16(3):223–229. https://doi.org/10.1006/mcpr.2002.0415
Njiru ZK (2012) Loop-mediated isothermal amplification technology: towards point of care diagnostics. PLoS Negl Trop Dis 6:e1572. https://doi.org/10.1371/journal.pntd.0001572
Notomi T, Okayama H, Masubuchi H, Yonekawa T, Watanabe K, Amino N, Hase T (2000) Loop-mediated isothermal amplification of DNA. Nucleic Acids Res 28(12):63–69. https://doi.org/10.1093/nar/28.12.e63
Notomi T, Mori Y, Tomita N, Kanda H (2015) Loop-mediated isothermal amplification (LAMP): principle features and future prospects. J Microbiol 53(1):1–5. https://doi.org/10.1007/s12275-015-4656-9
Rahmati S, Julkapli NM, Yehye WA, Basirun WJ (2016) Identification of meat origin in food products–a review. Food Control. https://doi.org/10.1016/j.foodcont.2016.04.013
Ran G, Ren L, Han X, Liu X, Li Z, Pang D, Ouyang H, Tang X (2016) Development of a rapid method for the visible detection of pork DNA in halal products by loop-mediated isothermal amplification. Food Anal Methods 9(3):565–570. https://doi.org/10.1007/s12161-015-0246-z
Rees WA, Korte YT, Von J, Hippel PH (1993) Betaine can eliminate the base pair composition dependence of DNA melting. Biochem 32(1):137–144. https://doi.org/10.1021/bi00052a019
Rojas M, Gonzalez I, Pavon M, Pegels N, Hernández P, Garcia T, Martín R (2011) Mitochondrial and nuclear markers for the authentication of partridge meat and the specific identification of red-legged partridge meat products by polymerase chain reaction. Poult Sci 90(1):211–222. https://doi.org/10.3382/ps.2010-00895
Santos CG, Melo VS, Amaral JS, Estevinho L, Oliveira MB, Mafra I (2012) Identification of hare meat by a species-specific marker of mitochondrial origin. Meat Sci 90:836–841. https://doi.org/10.1016/j.meatsci.2011.10.018
Soares S, Amaral JS, Oliveira MBP, Mafra I (2014) Quantitative detection of soybean in meat products by a TaqMan real-time PCR assay. Meat Sci 98(1):41–46. https://doi.org/10.1016/j.meatsci.2014.04.002
Stamoulis P, Stamatis C, Saradou T, Mamuris Z (2010) Development and application of molecular markers for poultry meat identification in food chain. Food Control 21:1061–1065. https://doi.org/10.1016/j.foodcont.2009.12.027
Tomita N, Mori Y, Kanda H, Notomi T (2008) Loop-mediated isothermal amplification (LAMP) of gene sequences and simple visual detection of products. Nat Protoc 3(5):877–882. https://doi.org/10.1038/nprot.2008.57
Vaithiyanathan S, Kulkarni VV (2016) Species identification of cattle and buffalo fat through PCR assay. J Food Sci Technol 53(4):2077–2082. https://doi.org/10.1007/s13197-016-2198-8
Wang Z, Gu Q, Sun H, Li H, Sun B, Liang X, Yuan Y, Liu R, Shi Y (2014) One-step reverse transcription loop mediated isothermal amplification assay for sensitive and rapid detection of Cucurbit chlorotic yellows virus. J Virol Methods 195:63–66. https://doi.org/10.1016/j.jviromet.2013.08.037
Yang L, Fu S, Peng X, Li L, Song T, Li L. (2014) Identification of pork in meat products using real-time loop mediated isothermal amplification. Biotechnol and Biotechnol Equip 28(5):882–888. https://doi.org/10.1080/13102818.2014.963789
Acknowledgments
Authors would like to acknowledge The Director, Indian Veterinary Research Institute and Head, Division of Livestock Products Technology, Indian Veterinary Research Institute, Izatnagar, Bareilly for providing necessary facilities and financial assistance to complete this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Sarita Kumari declares that she has no conflict of interest. R. R. Kumar declares that he has no conflict of interest. S. K. Mendiratta declares that he has no conflict of interest. Deepak Kumar declares that he has no conflict of interest. Arun Kumar declares that he has no conflict of interest. Dhananjay Kumar declares that he has no conflict of interest. Preeti Rana declares that she has no conflict of interest. Jyoti Jawla declares that she has no conflict of interest.
Ethics Approval
Not applicable.
Informed Consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kumari, S., Kumar, R.R., Mendiratta, S.K. et al. On-Site Detection of Tissues of Buffalo Origin by Loop-Mediated Isothermal Amplification (LAMP) Assay Targeting Mitochondrial Gene Sequences. Food Anal. Methods 13, 1060–1068 (2020). https://doi.org/10.1007/s12161-020-01718-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01718-2