Abstract
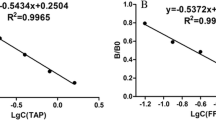
Polymyxin B (PMB), a member of polypeptide antibiotics, is widely used for the treatment of infection in animals such as cattle, sheep, pigs, and chickens. However, it is toxic on the kidneys and nervous system, and polymyxin resistance is increasingly reported, which leaves a serious threat to human health. Therefore, it is essential to establish rapid methods for detecting PMB with high sensitivity and specificity. In this study, an anti-PMB polyclonal antibody (pAb) was obtained by immunizing New Zealand white rabbits with PMB conjugated with glycosylated bovine serum albumin (GBSA). Indirect competitive enzyme-linked immunosorbent assay (ic-ELISA) and indirect competitive chemiluminescent enzyme immunoassay (ic-CLEIA) were developed. Under the optimal conditions, inhibitory concentrations (IC50) of PMB were 257.1 ng/mL (ic-ELISA) and 250.8 ng/mL (ic-CLEIA); the limits of detection (LOD) were 17.4 ng/mL (ic-ELISA) and 14.5 ng/mL (ic-CLEIA), respectively. Cross-reactivity of the pAb toward polymyxin E (PME) was 257.1%, and no response was found with other antibiotics. The recovery rates in spiked meat samples were 77.4~106.1% (ic-ELISA) and 84.1~107.1% (ic-CLEIA), respectively.





Similar content being viewed by others
References
Bergen PJ, Landersdorfer CB, Lee HJ, Li J, Nation RL (2012a) Old’ antibiotics for emerging multidrug-resistant bacteria. Curr Opin Infect Dis 25:626–633
Bergen PJ, Landersdorfer CB, Zhang J, Zhao M, Lee HJ, Nation RL, Li J (2012b) Pharmacokinetics and pharmacodynamics of ‘old’ polymyxins: what is new? Diagn Microbiol Infec Dis 74:213–223
Burkin MA, Galvidis IA (2010) Development of a competitive indirect ELISA for the determination of lincomycin in milk, eggs, and honey. J Agric Food Chem 58:9893–9898
Cai Y, Lee W, Kwa AL (2015) Polymyxin B versus colistin: an update. Expert Rev Anti Infect Ther 13:419–427
Chepyala D, Tsai IL, Sun HY, Lin SW, Kuo CH (2015) Development and validation of a high-performance liquid chromatography-fluorescence detection method for the accurate quantification of colistin in human plasma. J Chromatogr B 980:48–54
Cui L, He J, Hao R, Zhang X, Du E, Li X (2016) Preparation of a chicken scFv to analyze gentamicin residue in animal derived food products. Anal Chem 88:4092
Gales AC, Reis AO, Jones RN (2001) Contemporary assessment of antimicrobial susceptibility testing methods for polymyxin B and colistin: review of available interpretative criteria and quality control guidelines. J Clin Microbiol 39:183–190
Guo YB, Chen LP, Cao HW, Wang N, Zheng J, Xiao GX (2007) Polymyxin B antagonizing biological activity of lipopolysaccharide. Chin J Traumatol 10:180–183
He J, Hu J, Thirumalai D, Schade R, Du E, Zhang X (2015) Development of indirect competitive ELISA using egg yolk-derived immunoglobulin (IgY) for the detection of gentamicin residues. J Environ Sci Health B 51:8–13
Hee KH, Yee K, Leaw J, Ong JL, Lee LS (2017) Development and validation of liquid chromatography tandem mass spectrometry method quantitative determination of polymyxin B1, polymyxin B2, polymyxin B3 and isoleucine-polymyxin B1 in human plasma and its application in clinical studies. J Pharm Biomed Anal 140:91–97
Kadar B, Kocsis B, Nagy K, Szabo D (2013) The renaissance of polymyxins. Curr Med Chem 20:3759–3773
Landman D, Georgescu C, Martin DA, Quale J (2008) Polymyxins revisited. Clin Microbiol Rev 21:449–465
Li Y, Liu L, Song S, Kuang H, Xu C (2018) A rapid and semi-quantitative gold nanoparticles based strip sensor for polymyxin B sulfate residues. Nanomaterials 8:144
Liu T, Gao L, Zhao J, Cao Y, Tang Y, Miao P (2017) A polymyxin B-silver nanoparticle colloidal system and the application of lipopolysaccharide analysis. Analyst 143:1053–1058. https://doi.org/10.1039/C7AN01788J
Lu J, Xu Y, Chu JS, Xie L (2011) Chemiluminescence enzyme immunoassay for the determination of zearalenone in corn. Food Ferment Ind 37:160–165
Olaitan AO, Morand S, Rolain JM (2014) Mechanisms of polymyxin resistance: acquired and intrinsic resistance in bacteria. Front Microbiol 5:643
Orwa JA, Van GA, Roets E, Hoogmartens J (2000) Liquid chromatography of polymyxin B sulphate. J Chromatogr A 870:237–243
Pendela M, Adams EJ (2004) Development of a liquid chromatographic method for ear drops containing neomycin sulphate, polymyxin B sulphate and dexamethasone sodium phosphate. J Pharm Biomed Anal 36:751–757
Ronco C, Klein DJ (2014) Polymyxin B hemoperfusion: a mechanistic perspective. Crit Care 18:309
Saita T, Yoshida M, Nakashima M, Matsunaga H, Fujito H, Mori M (1999) A highly sensitive ELISA for the quantification of polymyxin B sulfate in human serum. Biol Pharm Bull 22:1257–1261
Sitzlar B, Vajravelu RK, Jury L, Donskey CJ, Jump RL (2012) Environmental decontamination with ultraviolet radiation to prevent recurrent Clostridium difficile infection in 2 roommates in a long-term care facility. Infect Control Hosp Epidemiol 33:534–536
Storm DR, And KSR, Swanson PE (1977) Polymyxin and related peptide antibiotics. Annu Rev Biochem 46:723–763
Vaara M (2010) Polymyxins and their novel derivatives. Curr Opin Microbiol 13:574–581
Velkov T, Roberts KD, Nation RL, Thompson PE, Li J (2013) Pharmacology of polymyxins: new insights into an ‘old’ class of antibiotics. Future Microbiol 8:711–724
Wan EC, Ho C, Sin DW, Wong YC (2006) Detection of residual bacitracin A, colistin A, and colistin B in milk and animal tissues by liquid chromatography tandem mass spectrometry. Anal Bioanal Chem 385:181–188
Yu F, Yu S, Yu L, Li Y, Wu Y, Zhang H, Qu L, Harrington PB (2014) Determination of residual enrofloxacin in food samples by a sensitive method of chemiluminescence enzyme immunoassay. Food Chem 149:71–75
Yu S, Yang B, Yan L, Dai Q (2017) Sensitive detection of α-onotoxin GI in human plasma using a solid-phase extraction column and LC-MS/MS. Toxins 9:235
Funding
This work was supported by the Key Program for International S&T Cooperation Project of Shaanxi Province (2017KW-ZD-10), the strategic program UID/BIA/04050/2013 (POCI-01-0145-FEDER-007569) funded by national funds through the FCT I. P., the Ministério da Ciência, Tecnologia e Ensino Superior (MCTES), the ERDF through the COMPETE2020 - Programa Operacional Competitividade e Internacionalização (POCI), and the Ministry of Education and Science of the Russian Federation, unique project identifier RFMEFI60417X0198.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Long Xu declares that he has no conflict of interest. Maksim Burkin declares that he has no conflict of interest. Sergei Eremin declares that he has no conflict of interest. Alberto C. P. Dias declares that he has no conflict of interest. Xiaoying Zhang declares that he has no conflict of interest.
Ethical Approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Informed Consent
Informed consent was obtained from all individual participants included in the study (in case humans are involved).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, L., Burkin, M., Eremin, S. et al. Development of Competitive ELISA and CLEIA for Quantitative Analysis of Polymyxin B. Food Anal. Methods 12, 1412–1419 (2019). https://doi.org/10.1007/s12161-019-01477-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-019-01477-9