Abstract
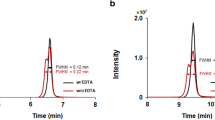
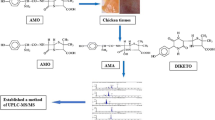
An amoxicillin stability study was performed under different pH (1, 3 and 5) and temperature (4 °C, 22 °C, 37 °C and 55 °C) conditions and for incurred samples stored at −20 °C with the goals of better understanding amoxicillin degradation and characterising its main degradation products (amoxicilloic acid and amoxicillin diketopiperazine). The analytical methodology used consisted of a simple extraction using phosphate buffer (pH 8) with sodium chloride followed by a sample clean-up using OASIS® HLB cartridges and a liquid chromatography–tandem mass spectrometric analysis. Amoxicillin was found to be greatly unstable at temperatures above 22 °C for all pH values studied, so it was recommended that biological samples should be frozen at temperatures below −70 °C until analysis of the amoxicillin residues.





Similar content being viewed by others
References
Agresti A (1992) Stat Sci 7:131
Alves RR, Ferreira MB, Santos AS, Pregal AL, Pedro E, Melo A, Santos MC, Barbosa MP (2005) Rev Port Imunoalerg 13:259
Antignac J-P, le Bizec B, Monteau F, André F (2003) Anal Chim Acta 483:325
Blanca M, Vega JM, Garcia J, Carmona MJ, Terados S, Avila MJ, Miranda A, Juarez C (1990) Clin Experim Allergy 20:475
Bogialli S, Capitolino V, Curini R, di Corcia A, Nazzari M, Sergi M (2004) J Agric Food Chem 52:3286
Commission Decision 2002/657/EC of 12 August (2002) Off J Eur Commun L221:8
de Baere S, Cherlet M, Baert KP, de Backer P (2002) Anal Chem 74:1393
de Baere S, Wassink P, Croubels S, de Boever S, Baert K, de Backer P (2005) Anal Chim Acta 529:221
Erah PO, Goddard AF, Barrett DA, Shaw PN, Spiller RC (1997) J Antim Chemoth 39:5
Fontanals N, Marcé RM, Borrull F (2005) Trends Anal Chem 24:394
Granellik K, Branzell C (2007) Anal Chim Acta 586:289
ISO 8466–1 (1990) Water quality—calibration and evaluation of analytical methods and estimation of performance characteristics. Part 1: Statistical evaluation of the linear calibration function. International Organization for Standardization (ed), Geneve
Okamoto T, Yoshiyama H, Nakazawa T, Park I-D, Chang MW, Yanai H, Okita K, Shirai M (2002) J Antimicr Chemoth 50:849
Popelka P, Nagy J, Germuška R, Marcinćák S, Jevinová P, de Rijk A (2005) Food Addit Contam 22:557
Regulation (EU) No. 37/2010 of the Council of 22 December 2009. Off J Eur Commun L15, 1 (2010)
Reyns T, Cherlet M, de Baere S, de Backer P, Croubels S (2008) J Chromatogr B 861:108
Sousa JC (2005) Manual de antibióticos antibacterianos. Fernando Pessoa University (ed), Oporto
Valvo L, Ciranni E, Alimenti R, Alimonti S, Draisci R, Giannetti L, Lucentini L (1998) J Chromatogr A 797:311
Verdon E, Fuselier R, Hurtaud-Pessel D, Pierrick Couëdor P, Cadieu N, Laurentie M (2000) J Chromatogr A 882:135
Vorona M, Veinberg G, Liepinsh E, Kažoka H, Andrejeva G, Lukevics E (2009) Chem Heteroc Comp 45:752
Weigel S, Kallenborn R, Hühnerfuss H (2004) J Chromatogr A 1023:183
Wibawa JJD, Fowkes D, Shaw PN, Barrett DA (2002) J Chromatogr B 774:141
Acknowledgements
A special thanks to GlaxoSmithKline (UK) for providing the standards of amoxicillin’s degradation products.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Freitas, A., Barbosa, J. & Ramos, F. Determination of Amoxicillin Stability in Chicken Meat by Liquid Chromatography–Tandem Mass Spectrometry. Food Anal. Methods 5, 471–479 (2012). https://doi.org/10.1007/s12161-011-9267-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-011-9267-4