Abstract
Objectives
Attenuation correction (AC) is crucial for ensuring the quantitative accuracy of positron emission tomography (PET) imaging. However, obtaining accurate μ-maps from brain-dedicated PET scanners without AC acquisition mechanism is challenging. Therefore, to overcome these problems, we developed a deep learning-based PET AC (deep AC) framework to synthesize transmission computed tomography (TCT) images from non-AC (NAC) PET images using a convolutional neural network (CNN) with a huge dataset of various radiotracers for brain PET imaging.
Methods
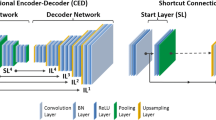
The proposed framework is comprised of three steps: (1) NAC PET image generation, (2) synthetic TCT generation using CNN, and (3) PET image reconstruction. We trained the CNN by combining the mixed image dataset of six radiotracers to avoid overfitting, including [18F]FDG, [18F]BCPP-EF, [11C]Racropride, [11C]PIB, [11C]DPA-713, and [11C]PBB3. We used 1261 brain NAC PET and TCT images (1091 for training and 70 for testing). We did not include [11C]Methionine subjects in the training dataset, but included them in the testing dataset.
Results
The image quality of the synthetic TCT images obtained using the CNN trained on the mixed dataset of six radiotracers was superior to those obtained using the CNN trained on the split dataset generated from each radiotracer. In the [18F]FDG study, the mean relative PET biases of the emission-segmented AC (ESAC) and deep AC were 8.46 ± 5.24 and − 5.69 ± 4.97, respectively. The deep AC PET and TCT AC PET images exhibited excellent correlation for all seven radiotracers (R2 = 0.912–0.982).
Conclusion
These results indicate that our proposed deep AC framework can be leveraged to provide quantitatively superior PET images when using the CNN trained on the mixed dataset of PET tracers than when using the CNN trained on the split dataset which means specific for each tracer.








Similar content being viewed by others
References
Phelps ME. PET: molecular imaging and its biological applications. New York: Springer; 2012.
Watanabe M, Shimizu K, Omura T, Takahashi M, Kosugi T, Yoshikawa E, et al. A new high-resolution PET scanner dedicated to brain research. IEEE Trans Nucl Sci. 2002;49(3):634–9.
Watanabe M, Saito A, Isobe T, Ote K, Yamada R, Moriya T, et al. Performance evaluation of a high-resolution brain PET scanner using four-layer MPPC DOI detectors. Phys Med Biol. 2017;62(17):7148–66.
Tashima H, Yoshida E, Iwao Y, Wakizaka H, Maeda T, Seki C, et al. First prototyping of a dedicated PET system with the hemisphere detector arrangement. Phys Med Biol. 2019;64(6):065004.
Berker Y, Li Y. Attenuation correction in emission tomography using the emission data—a review. Med Phys. 2016;43(2):807–32.
Rezaei A, Defrise M, Bal G, Michel C, Conti M, Watson C, et al. Simultaneous reconstruction of activity and attenuation in time-of-flight PET. IEEE Trans Med Imaging. 2012;31(12):2224–33.
Kobayashi T, Kitamura K. A solution for scaling problem in joint estimation of activity and attenuation. In: 2017 IEEE nuclear science symposium and medical imaging conference (NSS/MIC); 2017. https://doi.org/10.1109/NSSMIC.2017.8532856.
Marinelli M, Positano V, Tucci F, Neglia D, Landini L. Automatic PET-CT image registration method based on mutual information and genetic algorithms. Sci World J. 2012;2012:567067. https://doi.org/10.1100/2012/567067.
Hofmann M, Bezrukov I, Mantlik F, Aschoff P, Steinke F, Beyer T, et al. MRI-based attenuation correction for whole-body PET/MRI: quantitative evaluation of segmentation- and atlas-based methods. J Nucl Med. 2011;52(9):1392–9.
Hashimoto F, Ohba H, Ote K, Teramoto A, Tsukada H. Dynamic PET image denoising using deep convolutional neural networks without prior training datasets. IEEE Access. 2019;7:96594–603.
Hashimoto F, Ohba H, Ote K, Kakimoto A, Tsukada H, Ouchi Y. 4D deep image prior: dynamic PET image denoising using an unsupervised four-dimensional branch convolutional neural network. Phys Med Biol. 2021;66(1):015006.
Reader AJ, Corda G, Mehranian A, da Costa-Luis C, Ellis S, Schnabel JA. Deep learning for PET image reconstruction. IEEE Trans Radiat Plasma Med Sci. 2021;5(1):1–25.
Suzuki K. Overview of deep learning in medical imaging. Radiol Phys Technol. 2017;10(3):257–73.
Lee JS. A review of deep learning-based approaches for attenuation correction in positron emission tomography. IEEE Trans Radiat Plasma Med Sci. 2021;5(2):160–84.
Shiri I, Arabi H, Geramifar P, Hajianfar G, Ghafarian P, Rahmim A, et al. Deep-JASC: joint attenuation and scatter correction in whole-body 18F-FDG PET using a deep residual network. Eur J Nucl Med Mol Imaging. 2020;47(11):2533–48. https://doi.org/10.1007/s00259-020-04852-5.
Arabi H, Zeng G, Zheng G, Zaidi H. Novel adversarial semantic structure deep learning for MRI-guided attenuation correction in brain PET/MRI. Eur J Nucl Med Mol Imaging. 2019;46(13):2746–59.
Yang J, Park D, Gullberg GT, Seo Y. Joint correction of attenuation and scatter in image space using deep convolutional neural networks for dedicated brain 18F-FDG PET. Phys Med Biol. 2019;64(7):075019.
Liu F, Jang H, Kijowski R, Zhao G, Bradshaw T, McMillan AB. A deep learning approach for 18F-FDG PET attenuation correction. EJNMMI Phys. 2018;5(1):24. https://doi.org/10.1186/s40658-018-0225-8.
Armanious K, Hepp T, Küstner T, Dittmann H, Nikolaou K, La Fougère C, et al. Independent attenuation correction of whole body [18F]FDG-PET using a deep learning approach with generative adversarial networks. EJNMMI Res. 2020;10(1):53. https://doi.org/10.1186/s13550-020-00644-y.
Armanious K, Küstner T, Reimold M, Nikolaou K, La Fougère C, Yang B, et al. Independent brain 18F-FDG PET attenuation correction using a deep learning approach with generative adversarial networks. Hell J Nucl Med. 2019;22(3):179–86. https://doi.org/10.1967/s002449911053.
Dong X, Wang T, Lei Y, Higgins K, Liu T, Curran WJ, et al. Synthetic CT generation from non-attenuation corrected PET images for whole-body PET imaging. Phys Med Biol. 2019;64(21):215016.
Hu Z, Li Y, Zou S, Xue H, Sang Z, Liu X, et al. Obtaining PET/CT images from non-attenuation corrected PET images in a single PET system using Wasserstein generative adversarial networks. Phys Med Biol. 2020;65(21):215010.
Dong X, Lei Y, Wang T, Higgins K, Liu T, Curran WJ, et al. Deep learning-based attenuation correction in the absence of structural information for whole-body positron emission tomography imaging. Phys Med Biol. 2020;65(5):055011.
Hwang D, Kim KY, Kang SK, Seo S, Paeng JC, Lee DS, et al. Improving the accuracy of simultaneously reconstructed activity and attenuation maps using deep learning. J Nucl Med. 2018;59(10):1624–9.
Hwang D, Kang SK, Kim KY, Seo S, Paeng JC, Lee DS, et al. Generation of PET attenuation map for whole-body time-of-flight 18F-FDG PET/MRI using a deep neural network trained with simultaneously reconstructed activity and attenuation maps. J Nucl Med. 2019;60(8):1183–9.
Arabi H, Zaidi H. Deep learning-guided estimation of attenuation correction factors from time-of-flight PET emission data. Med Image Anal. 2020;64:101718.
Arabi H, Bortolin K, Ginovart N, Garibotto V, Zaidi H. Deep learning-guided joint attenuation and scatter correction in multi-tracer neuroimaging studies. Hum Brain Mapp. 2020;41(13):3667–79.
Harada N, Nishiyama S, Kanazawa M, Tsukada H. Development of novel PET probes, [18F]BCPP-EF, [18F]BCPP-BF, and [11C]BCPP-EM for mitochondrial complex 1 imaging in the living brain. J Label Compd Radiopharm. 2013;56(11):553–61.
Boutin H, Chauveau F, Thominiaux C, Grégoire MC, James ML, Trebossen R, et al. 11C-DPA-713: a novel peripheral benzodiazepine receptor PET ligand for in vivo imaging of neuroinflammation. J Nucl Med. 2007;48(4):573–81.
Kaji S, Kida S. Overview of image-to-image translation by use of deep neural networks: denoising, super-resolution, modality conversion, and reconstruction in medical imaging. Radiol Phys Technol. 2019;12(3):235–48.
Hashimoto F, Kakimoto A, Ota N, Ito S, Nishizawa S. Automated segmentation of 2D low-dose CT images of the psoas-major muscle using deep convolutional neural networks. Radiol Phys Technol. 2019;12(2):210–5.
Hashimoto F, Ote K, Oida T, Teramoto A, Ouchi Y. Compressed-sensing magnetic resonance image reconstruction using an iterative convolutional neural network approach. Appl Sci. 2020;10(6):1902.
Hashimoto F, Ito M, Ote K, Isobe T, Okada H, Ouchi Y. Emis2Trans: attenuation correction for brain PET with many types of PET ligands using convolutional neural networks. In: 2019 IEEE nuclear science symposium and medical imaging conference (NSS/MIC); 2019. https://doi.org/10.1109/NSS/MIC42101.2019.9059961.
Ronneberger O, Fischer P, Brox T. U-net: convolutional networks for biomedical image segmentation. Lect Notes Comput Sci MICCAI. 2015;2015:234–41. https://doi.org/10.1007/978-3-319-24574-4_28.
Tanaka E, Kudo H. Subset-dependent relaxation in block-iterative algorithms for image reconstruction in emission tomography. Phys Med Biol. 2003;48(10):1405–22.
Holmes CJ, Hoge R, Collins L, Woods R, Toga AW, Evans AC. Enhancement of MR images using registration for signal averaging. J Comput Assist Tomogr. 1998;22(2):324–33.
Zaidi H, Hasegawa B. Determination of the attenuation map in emission tomography. J Nucl Med. 2003;44(2):291–315.
Kittler J, Illingworth J. Minimum error thresholding. Pattern Recogn. 1986;19(1):41–7.
Nakamoto Y, Osman M, Cohade C, Marshall LT, Links JM, Kohlmyer S, et al. PET/CT: comparison of quantitative tracer uptake between germanium and CT transmission attenuation-corrected images. J Nucl Med. 2002;43(9):1137–43.
Çiçek Ö, Abdulkadir A, Lienkamp SS, Brox T, Ronneberger O. 3D U-net: learning dense volumetric segmentation from sparse annotation. Lect Notes Comput Sci MICCAI. 2016;2016(3d):424–32. https://doi.org/10.1007/978-3-319-46723-8_49.
Acknowledgements
The authors would like to thank Dr. Norohiro Harada, Dr. Hideo Tsukada, and Dr. Hiroyuki Ohba from the Central Research Laboratory, Hamamatsu Photonics K.K for constructive advice. The authors also would like to thank the staff of Hamamatsu Medical Imaging Center and the staff of Hamamatsu Photonics K.K. for their support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
F.H., M.I., K.O, T.I., and H.O. are employees of Hamamatsu Photonics K.K. The company had no control over the interpretation, writing, or publication of this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hashimoto, F., Ito, M., Ote, K. et al. Deep learning-based attenuation correction for brain PET with various radiotracers. Ann Nucl Med 35, 691–701 (2021). https://doi.org/10.1007/s12149-021-01611-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-021-01611-w