Abstract
Objective
In patients with differentiated thyroid carcinoma (DTC) with a microscopically positive tumor margin, the optimal first dose of I-131 during adjuvant radioactive iodine (RAI) therapy to improve relapse-free survival (RFS) is unclear. Due to the limited number of hospital beds, 1110 MBq of I-131 is administered to such patients in Japan. This study primarily aimed to retrospectively determine the difference in RFS between high-dose (3700 MBq) and low-dose (1110 MBq) adjuvant RAI therapies in DTC patients with a microscopically positive tumor margin. The secondary aim was to investigate the background factors affecting RFS.
Methods
Forty-eight consecutive patients (15 men and 33 women) who underwent total thyroidectomy between April 2007 and December 2017 for DTC without gross residual tumors and distant metastasis, and who were diagnosed with a positive margin histopathologically, followed by RAI therapy, were enrolled. We retrospectively investigated initial I-131 dose, classifying it into high-dose and low-dose groups. The primary endpoint was RFS. Relapse was considered in the following cases: patients with visible recurrent tumor and/or metastasis on neck ultrasound and/or computed tomography, and patients without visible tumor, but with clearly increasing thyroglobulin levels on follow-up every 3–6 months. RFS outcomes were analyzed using the Kaplan–Meier method. Univariate analysis was performed using the log-rank test, and multivariate analysis was undertaken using the Cox proportional hazard model.
Results
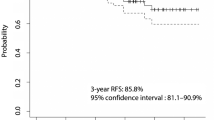
There were nine cases of recurrence (52.9%) in the low-dose group and seven in the high-dose group (22.5%) during follow-up. The estimated median RFS was 69.4 months in the low-dose group and 120.7 months in the high-dose group. High-dose administration was associated with improved RFS, as demonstrated by Kaplan–Meier survival curves (log-rank test [P = 0.009]). Patient factors associated with worse RFS included low-dose administration (hazard ratio [HR] = 91.9; 95% confidence interval [CI] = 7.66–1102.79); P < 0.001), T4 stage (HR = 1.88; 95% CI = 0.44–8.10; P = 0.015), and presence of central lymph node metastases (HR = 190; 95% CI = 3.80–9496; P = 0.009). The most common type of recurrence was lymph node metastasis.
Conclusion
Patients with microscopically positive tumor margins could benefit from RAI with high activities.


Similar content being viewed by others
References
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2015;26:1–133.
Tsang RW, Brierley JD, Simpson WJ, Panzarella T, Gospodarowicz MK, Sutcliffe SB. The effects of surgery, radioiodine, and external radiation therapy on the clinical outcome of patients with differentiated thyroid carcinoma. Cancer. 1998;82:375–88.
Bernier J, Cooper JS, Pajak TF, van Glabbeke M, Bourhis J, Forastiere A, et al. Defining risk levels in locally advanced head and neck cancers: a comparative analysis of concurrent postoperative radiation plus chemotherapy trials of the EORTC(#22931)and RTOG(#9501). Head Neck. 2005;27:843–50.
Chow S, Yau S, Kwan C, Poon P, Law S. Local and regional control in patients with papillary thyroid carcinoma: specific indications of external radiotherapy and radioactive iodine according to T and N categories in AJCC 6th edition. EndocrRelat Cancer. 2006;13:1159–72.
Lee NK, Kim CY, Baek SK, Jung KY. The role of adjuvant radiation therapy for locoregionally advanced papillary thyroid carcinoma. Oncology. 2016;90:209–14.
Wang LY, Ghossein R, Palmer FL, Nixon IJ, Tuttle RM, Shaha AR, et al. Microscopic positive margins in differentiated thyroid cancer is not an independent predictor of local failure. Thyroid. 2015;25:993–8.
Elisei R, Molinaro E, Agate L, Bottici V, Masserini L, Ceccarelli C, et al. Are the clinical and pathological features of differentiated thyroid carcinoma really changed over the last 35 years? Study on 4187 patients from a single Italian institution to answer this question. J ClinEndocrinolMetab. 2010;95:1516–27.
Ruel E, Thomas S, Dinan M, Perkins JM, Roman SA, Sosa JA. Adjuvant radioactive iodine therapy is associated with improved survival for patients with intermediate-risk papillary thyroid cancer. J ClinEndocrinolMetab. 2015;100:1529–36.
National Comprehensive Cancer Network, Inc. Practice Guidelines in Oncology-Thyroid Carcinoma v.2. 2007; https://www.nccn.org/professionals/physician_gls/PDF/thyroid.pdf. Accessed 30 May 2017.
Pacini F, Brianzoni E, Durante C, Elisei R, Ferdeghini M, Fugazzola L, et al. Recommendations for post-surgical thyroid ablation in differentiated thyroid cancer: a 2015 position statement of the Italian Society of Endocrinology. J Endocrinol Invest. 2016;39:341–7.
The Japanease Society of Nuclear Medicine in Oncology and Imunology-Survey on actual situation of the commissioning status of RAI treatment beds in the therapy for thyroid cancer (v.4). https://www.jstage.jst.go.jp/article/kakuigaku/54/1/54_rp.1778/_pdf/-char/ja (in Japanese). Accessed 21 Jul 2017
American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules, and Differentiated Thyroid Cancer, Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19:1167–214.
Hong CM, Ahn BC, Park JY, Jeong SY, Lee SW, Lee J. Prognostic implications of microscopic involvement of surgical resection margin in patients with differentiated papillary thyroid cancer after high-dose radioactive iodine ablation. Ann Nucl Med. 2012;26:311–8.
Hart DM, Zago S, Leboulleux S, Mirghani H, Deandreis D, Baudin E, et al. Resection margins and prognosis in locally invasive thyroid cancer. Head Neck. 2014;36:1034–8.
Mercado CE, Drew PA, Morris CG, Dziegielewski PT, Mendenhall WM, Amdur RJ. Positive surgical margins in favorable-stage differentiated thyroid cancer. Am J ClinOncol. 2018;41:1168–71.
Saito W, Tanaka K, Ogura H, Kishino E, Sugawara S, Yamamoto M, et al. Analyses of the outcome of the patients with the local microscopic residual of papillary thyroid carcinoma. Off J Jpn Ass Endocr Surgeon JpnSoc Thyroid Surg. 2016;33:254–8.
Sanabria A, Rojas A, Arevalo J, Kowalski LP, Nixon I. Microscopically positive surgical margins and local recurrence in thyroid cancer. A meta-analysis. Eur J SurgOncol. 2019;45:1310–6.
Ito Y, Higashiyama T, Takamura Y, Miya A, Kobayashi K, Matsuzuka F, et al. Risk factors for recurrence to the lymph node in papillary thyroid carcinoma patients without preoperatively detectable lateral node metastasis: validity of prophylactic modified radical neck dissection. World J Surg. 2007;31:2085–91.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Watanabe, K., Igarashi, T., Uchiyama, M. et al. Relapse-free survival after adjuvant radioactive iodine therapy in patients with differentiated thyroid carcinoma with a microscopically positive tumor margin. Ann Nucl Med 34, 920–925 (2020). https://doi.org/10.1007/s12149-020-01523-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-020-01523-1