Abstract
Purpose
The role of 18F-fluoride (18F-NaF) PET-CT for the detection of bone metastases in adults is well established and is considered superior to conventional bone scintigraphy. However, data pertaining use of 18F-NaF PET-CT in pediatric oncology is relatively sparse. The aim of the present study is to retrospectively analyze and share a single-center experience of 18F-NaF PET-CT in pediatric population and to provide preliminary information regarding imaging technique, feasibility of this modality in young patients and radiation dosimetry measurements in pediatric oncology cases.
Materials and methods
Twenty-four pediatric patients (mean age 8.0 ± 3.9) were included in the study for retrospective analysis. All patients were referred for primary staging or restaging for potential osseous metastatic disease and PET-CT scan was performed by injecting 2.2 MBq/kg (0.06 mCi/kg) of 18F-NaF.
Results
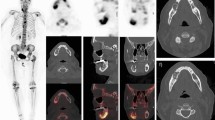
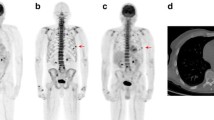
Nine patients were imaged for primary staging and in all cases increase osteoblastic activity was seen in the primary tumor and of these, metastatic bone disease was identified in 2/9 patients. In the restaging group comprising 15/24 patients, metastatic deposits were identified in 3/15 whilst no disease was seen in the remaining 12 patients. Patients were injected a mean dose of 90.35 ± 22.9 MBq with an estimated mean effective absorbed doses of 2.98 ± 0.75 mSv for 18F-NaF and 3.37 ± 2.4 mSv for CT alone. Mean cumulative effective dose of 18F-NaF PET-CT scan was 5.11 ± 2.7 mSv.
Conclusions
18F-NaF PET-CT may be a feasible alternative to 99mTc MDP for radionuclide bone scintigraphy in the evaluation of pediatric bone pathology. Due to its better pharmacokinetics, there is potential that osseous staging can be achieved with relatively low doses and with a similar radiation burden as with 99mTc-MDP imaging.




Similar content being viewed by others
References
Meyer JS, Nadel HR, Marina N, et al. Imaging guidelines for children with Ewing sarcoma and osteosarcoma: a report from the Children’s Oncology Group Bone Tumor Committee. Pediatr Blood Cancer. 2008;51:163–70.
Czernin J, Satyamurthy N, Schiepers C. Molecular mechanisms of bone 18F-NaF deposition. J Nucl Med. 2010;51:1826–9.
Derlin T, Tóth Z, Papp L, et al. Correlation of inflammation assessed by 18F-FDG PET, active mineral deposition assessed by18F-fluoride PET, and vascular calcification in atherosclerotic plaque: a dual-tracer PET/CT study. J Nucl Med. 2011;52:1020–7.
Grant FD, Fahey FH, Packard AB, et al. Skeletal PET with 18F-fluoride: applying new technology to an old tracer. J Nucl Med. 2008;49:68–78.
Behesht M, Mottaghy FM, Payche F, et al. 18 F-NaF PET/CT: EANM procedure guidelines for bone imaging. Eur J Nucl Med Mol Imaging. 2015;42:1767–77.
Radiation dose to patients from radiopharmaceuticals. Addendum 4 to ICRP Publication 53. ICRP Publication 106. Approved by the Commission in May 2013. Ann ICRP, 38, 1–197.
International Commission on Radiological Protection. ICRP Publication 80: radiation dose to patients from radiopharmaceuticals. Ann ICRP 2000;28(3):29.
Christner J, Kofler J, McCollough CH. Estimating effective dose for CT using dose-length product compared with using organ doses: consequences of adopting International Commission on Radiological Protection Publication 103 or Dual-Energy Scanning. AJR Am J Roentgenol. 2010;194:881–9.
American Cancer Society. Global cancer facts and figs. 2nd ed. Atlanta: American Cancer Society; 2011.
Ribeiro R, Pui CH. Treatment of acute lymphoblastic leukemia in low- and middle-income countries: challenges and opportunities. Leuk Lymphoma. 2008;49:373–6.
Voss SD. Pediatric oncology and the future of oncological imaging. Pediatr Radiol. 2011;41(Suppl 1):172–8.
Weiser DA, Kaste SC, Siegel MJ, Adamson PC. Imaging in childhood cancer: a Society for Pediatric Radiology and Children’s Oncology Group Joint Task Force Report. Pediatr Blood Cancer. 2013;60:1253–60.
Roberts CC, Daffner RH, Weissman BN, et al. ACR appropriateness criteria® on metastatic bone disease. J Am Coll Radiol. 2010;7:400–9.
Löfgren J, Mortensen J, Rasmussen SH, et al. A prospective study comparing 99mTc-hydroxyethylene-diphosphonate planar bone scintigraphy and whole-body SPECT/CT with 18F-fluoride PET/CT and 18F-fluoride PET/MRI for diagnosing bone metastases. Nucl Med. 2017;58:1778–85.
Frost ML, Blake GM, Cook GJ, et al. Differences in regional bone perfusion and turnover between lumbar spine and distal humerus: 18F-fluoride PET study of treatment-naive and treated postmenopausal women. Bone. 2009;45:942–8.
Blake GM, Park-Holohan SJ, Cook GJ, et al. Quantitative studies of bone with the use of 18F-fluoride and 99mTc-methylene diphosphonate. Semin Nucl Med. 2001;31:28–49.
Drubach LA, Sapp MV, Laffin S, Kleinman PK. Fluorine-18 NaF PET imaging of child abuse. Pediatr Radiol. 2008;38:776–9.
Laverick S, Bounds G, Wong WL. [18F]-Fluoride positron emission tomography for imaging condylar hyperplasia. Br J Oral Maxillofac Surg. 2009;47:196–9.
Small GR, Ruddy TD, Simion O, et al. Lessons from the Tc-99m shortage: implications of substituting Tl-201 for Tc-99m single-photon emission computed tomography. Circ Cardiovasc Imaging. 2013;6:683–91.
Lassmann M, Biassoni L, Monsieurs M, EANM Dosimetry and Paediatrics Committees, et al. The new EANM paediatric dosage card. Eur J Nucl Med Mol Imaging. 2009;36:540–1.
Lim R, Fahey FH, Drubach LA, et al. Early experience with fluorine-18 sodium fluoride bone PET in young patients with back pain. J Pediatr Orthop. 2007;27:277–82.
Segall G, Delbeke D, Stabin MG, et al. SNM practice guideline for sodium 18F-fluoride PET/CT bone scans 1.0. J Nucl Med. 2010;51:1813–20.
Marafi F, Esmail A, Rasheed R, et al. Novel weight-based dose threshold for 18F-NaF PET-CT imaging using advanced PET-CT systems: a potential tool for reducing radiation burden. Nucl Med Commun. 2017;38:764–70.
Chavhan GB, Caro-Dominguez P. Diffusion-weighted imaging in pediatric body magnetic resonance imaging. Pediatr Radiol. 2016;46:847–57.
Messiou C, Cook G, de Souza NM. Imaging metastatic bone disease from carcinoma of the prostate. Br J Cancer. 2009;101:1225–32.
Jambor I, Kuisma A, Ramadan S, et al. Prospective evaluation of planar bone scintigraphy, SPECT, SPECT/CT, 18F-NaF PET/CT and whole body 1.5T MRI, including DWI, for the detection of bone metastases in high risk breast and prostate cancer patients: SKELETA clinical trial. Acta Oncol. 2016;55:59–67.
Saif MW, Tzannou I, Makrilia N, et al. Role and cost effectiveness of PET/CT in management of patients with cancer. Yale J Biol Med. 2010;83:53–65.
Jones T, Townsend D. History and future technical innovation in positron emission tomography. J Med Imaging. 2017;4:011013.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no potential conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Usmani, S., Van den Wyngaert, T., Ahmed, N. et al. Technical feasibility, radiation dosimetry and clinical use of 18F-sodium fluoride (NaF) in evaluation of metastatic bone disease in pediatric population. Ann Nucl Med 32, 594–601 (2018). https://doi.org/10.1007/s12149-018-1279-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-018-1279-3