Abstract
Objective
Is to investigate possible factors predicting success of ablation for 3700 MBq radioactive iodine 131 in patients with differentiated thyroid cancer (DTC) following near total thyroidectomy.
Methods
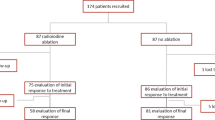
This retrospective study enrolled 272 patients between 2000 and 2014. The success or failure of ablation was assessed 6 months after given the dose and our criteria for complete successful remnant ablation defined as: Negative 131I whole body scan with no residual functioning thyroid tissue or distant functioning metastases and stimulated thyroglobulin (Tg) level less than 2 ng/ml. Different clinical and pathological factors, such as age, gender, tumor histology, grade and variants, size of primary malignant lesion, stage, and risk assessment according to the American (ATA) and European Thyroid Association (ETA) guidelines, associated pathology, tumor mutifocality, lymph node (LN) metastases and their number, invasiveness of the tumor (capsular invasion of the nodule, extra-thyroidal extension, and vascular invasion), baseline stimulated Tg level, and pre-ablative diagnostic scan were assessed.
Results
There were 185 successful ablations (68 %). The baseline-stimulated Tg measured before the ablation was the only independent predictor of ablation success in multivariate analysis (P < 0.0001) with odds ratio (OR) of 2.64 (95 % CI: 1.54–4.54) and the optimal cutoff for this was 3.8 ng/mL. On the univariate analysis, LN metastases was predictor of ablation failure (P value = 0.03).
Conclusion
Baseline-stimulated Tg level is clinically important and had a significant predictive value for successful ablation; therefore, higher pre-ablation Tg should potentially be incorporated in the decision making for 131I dosage or other treatment. In accordance with other studies, this is also applicable to cervical lymph nodal involvement and thyroid capsule invasion.

Similar content being viewed by others
References
Dohan O, Carrasco N. Thyroidal iodide transport and thyroid cancer. Cancer Treat Res. 2004;122:221–36.
Mazzaferri EL, Kloos RT. Clinical review 128: Current approaches to primary therapy for papillary and follicular thyroid cancer. J Clin Endocrinol Metab. 2001;86:1447–63.
Kukulska A, Krajewska J, Gawkowska-Suwinska M, Puch Z, Paliczka-Cieslik E, Roskosz J, et al. Radioiodine thyroid remnant ablation in patients with differentiated thyroid carcinoma (DTC): prospective comparison of long-term outcomes of treatment with 30, 60 and 100 mCi. Thyroid Res. 2010;3:9.
Verburg FA, de Keizer B, Lips CJ, Zelissen PM, de Klerk JM. Prognostic significance of successful ablation with radioiodine of differentiated thyroid cancer patients. Eur J Endocrinol. 2005;152:33–7.
Bal CS, Kumar A, Pant GS. Radioiodine dose for remnant ablation in differentiated thyroid carcinoma: a randomized clinical trial in 509 patients. J Clin Endocrinol Metab. 2004;89:1666–73.
Mazzaferri EL. An overview of the management of papillary and follicular thyroid carcinoma. Thyroid. 1999;9:421–7.
Sawka AM, Thephamongkhol K, Brouwers M, Thabane L, Browman G, Gerstein HC. Clinical review 170: a systematic review and metaanalysis of the effectiveness of radioactive iodine remnant ablation for well-differentiated thyroid cancer. J Clin Endocrinol Metab. 2004;89:3668–76.
Wagieh SM, El-Refaei SM, Salem SS, Al-Shiekh EA, Al-Ghamdy HA, Al-Juhani NR. Impact of histopathology of non-neoplastic thyroid tissue on ablation outcome in patients with papillary thyroid cancer. Nucl Med Commun. 2011;32:597–604.
Koch W, Knesewitsch P, Tatsch K, Hahn K. Stunning effects in radioiodine therapy of thyroid carcinoma: existence, clinical effects and ways out. Nuklearmedizin. 2003;42:10–4.
Arturi F, Russo D, Schlumberger M, du Villard JA, Caillou B, Vigneri P, et al. Iodide symporter gene expression in human thyroid tumors. J Clin Endocrinol Metab. 1998;83:2493–6.
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC cancer staging manual. 7th ed. New York: Springer; 2010. p. 87–96.
Pacini F, Schlumberger M, Dralle H, Elisei R, Smit JW, Wiersinga W, et al. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur J Endocrinol. 2006;154:787–803.
Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19:1167–214.
Zidan J, Hefer E, Iosilevski G, Drumea K, Stein ME, Kuten A, et al. Efficacy of I131 ablation therapy using different doses as determined by postoperative thyroid scan uptake in patients with differentiated thyroid cancer. Int J Radiat Oncol Biol Phys. 2004;59:1330–6.
Hackshaw A, Harmer C, Mallick U, Haq M, Franklyn JA. 131I activity for remnant ablation in patients with differentiated thyroid cancer: a systematic review. J Clin Endocrinol Metab. 2007;92:28–38.
Maxon HR, Englaro EE, Thomas SR, Hertzberg VS, Hinnefeld JD, Chen LS, et al. Radioiodine-131 therapy for well-differentiated thyroid cancer—a quantitative radiation dosimetric approach: outcome and validation in 85 patients. J Nucl Med. 1992;33:1132–6.
Zaman M, Toor R, Kamal S, Maqbool M, Habib S, Niaz K. A randomized clinical trial comparing 50 mCi and 100 mCi of iodine-131 for ablation of differentiated thyroid cancers. JPMA J Pak Med Assoc. 2006;56:353–6.
Golger A, Fridman TR, Eski S, Witterick IJ, Freeman JL, Walfish PG. Three-week thyroxine withdrawal thyroglobulin stimulation screening test to detect low-risk residual/recurrent well-differentiated thyroid carcinoma. J Endocrinol Invest. 2003;26:1023–31.
Tamilia M, Al-Kahtani N, Rochon L, Hier MP, Payne RJ, Holcroft CA, et al. Serum thyroglobulin predicts thyroid remnant ablation failure with 30 mCi iodine-131 treatment in patients with papillary thyroid carcinoma. Nucl Med Commun. 2011;32:212–20.
Bernier MO, Morel O, Rodien P, Muratet JP, Giraud P, Rohmer V, et al. Prognostic value of an increase in the serum thyroglobulin level at the time of the first ablative radioiodine treatment in patients with differentiated thyroid cancer. Eur J Nucl Med Mol Imaging. 2005;32:1418–21.
Kendler DB, Vaisman F, Corbo R, Martins R, Vaisman M. Preablation stimulated thyroglobulin is a good predictor of successful ablation in patients with differentiated thyroid cancer. Clin Nucl Med. 2012;37:545–9.
Caballero-Calabuig E, Cano-Terol C, Sopena-Monforte R, Reyes-Ojeda D, Abreu-Sanchez P, Ferrer-Rebolleda J, et al. Influence of the thyroid remnant in the elevation of the serum thyroglobulin after thyroidectomy in differentiated thyroid carcinoma. Importance of the diagnostic iodine total-body scanning. Eur J Nucl Med Mol Imaging. 2008;35:1449–56.
Stanojevic M, Savin S, Cvejic D, Djukic A, Jeremic M, Zivancevic Simonovic S. Comparison of the influence of thyroglobulin antibodies on serum thyroglobulin values from two different immunoassays in post surgical differentiated thyroid carcinoma patients. J Clin Lab Anal. 2009;23:341–6.
Verkooijen RB, Verburg FA, van Isselt JW, Lips CJ, Smit JW, Stokkel MP. The success rate of I-131 ablation in differentiated thyroid cancer: comparison of uptake-related and fixed-dose strategies. Eur J Endocrinol. 2008;159:301–7.
Rosario PW, Reis JS, Barroso AL, Rezende LL, Padrao EL, Fagundes TA. Efficacy of low and high 131I doses for thyroid remnant ablation in patients with differentiated thyroid carcinoma based on post-operative cervical uptake. Nucl Med Commun. 2004;25:1077–81.
Hee MP, Stephen KG. Stunning: untoward effect of 131I thyroid imaging prior to radioablation therapy. In: Leonard W, Douglas VN, editors. Thyroid cancer a comprehensive guide to clinical management. Totowa: Humana Press Inc; 2006. p. 337–45.
Meller B, Gaspar E, Deisting W, Czarnocka B, Baehre M, Wenzel BE. Decreased radioiodine uptake of FRTL-5 cells after (131)I incubation in vitro: molecular biological investigations indicate a cell cycle-dependent pathway. Eur J Nucl Med Mol Imaging. 2008;35:1204–12.
Park HM. Stunned thyroid after high-dose I-131 imaging. Clin Nucl Med. 1992;17(6):501–2.
Huic D, Medvedec M, Dodig D, Popovic S, Ivancevic D, Pavlinovic Z, et al. Radioiodine uptake in thyroid cancer patients after diagnostic application of low-dose 131I. Nucl Med Commun. 1996;17:839–42.
Leger FA, Izembart M, Dagousset F, Barritault L, Baillet G, Chevalier A, et al. Decreased uptake of therapeutic doses of iodine-131 after 185-MBq iodine-131 diagnostic imaging for thyroid remnants in differentiated thyroid carcinoma. Eur J Nucl Med. 1998;25:242–6.
Yeung HW, Humm JL, Larson SM. Radioiodine uptake in thyroid remnants during therapy after tracer dosimetry. J Nucl Med. 2000;41:1082–5.
Kao CH, Yen TC. Stunning effects after a diagnostic dose of iodine-131. Nuklearmedizin. 1998;37:30–2.
Karam M, Gianoukakis A, Feustel PJ, Cheema A, Postal ES, Cooper JA. Influence of diagnostic and therapeutic doses on thyroid remnant ablation rates. Nucl Med Commun. 2003;24:489–95.
Bajen MT, Mane S, Munoz A, Garcia JR. Effect of a diagnostic dose of 185 MBq 131I on postsurgical thyroid remnants. J Nucl Med. 2000;41:2038–3042.
Morris LF, Waxman AD, Braunstein GD. The nonimpact of thyroid stunning: remnant ablation rates in 131I-scanned and nonscanned individuals. J Clin Endocrinol Metab. 2001;86:3507–11.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Husseini, M.a.E. Implication of different clinical and pathological variables in patients with differentiated thyroid cancer on successful ablation for 3700 MBq 131I: a single Egyptian institutional experience over 14 years. Ann Nucl Med 30, 468–476 (2016). https://doi.org/10.1007/s12149-016-1084-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-016-1084-9