Abstract
Objective
Although [18F]-FDG is a useful oncologic PET tracer, FDG uptake is known to be low in a certain type of hepatocellular carcinoma (HCC). [18F]-fluoroacetate (18F-FACE) is an [18F] fluorinated acetate, which is known to be converted into fatty acids, incorporated in membrane and is expected to be a promising oncologic PET tracer. The aim of this study was to evaluate the usefulness of 18F-FACE as an oncologic PET tracer in preclinical study in healthy volunteers and in patients with liver tumors.
Methods
Twenty-four healthy volunteers (age 48.2 ± 12.9 years old; 15 male and 9 female) and ten patients with liver tumor (age 72.1 ± 7.0 years old; 6 male and 4 female) were included. We performed whole-body static PET/CT scan using 18F-FACE (n = 34) and 18F-FDG (n = 5 for volunteers, n = 8 for patients) on each day, respectively. Qualitative analysis and quantitative analysis of tumors (5 HCCs, 1 cholangiocellular carcinoma, 4 metastatic tumors from colon cancer and P-NET) were performed using SUVmax and tumor-to-normal liver ratio (TNR).
Results
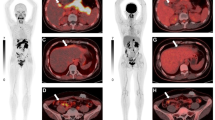
In healthy volunteers, 18F-FACE was metabolically stable in vivo and its biodistribution was almost similar to blood pool, basically uniformly independent of age and gender during PET scan time (up to 3 h). Normal physiological uptake of 18F-FACE at each organ including liver (SUVmean 1.8 ± 0.2) was lower than that of blood pool (SUVmean 2.3 ± 0.3) at 1 h after injection. Chronic inflammatory uptake around femur of post-operative state of femoral osteotomy and faint uptake of benign hemangioma were observed in a case of healthy volunteer. 18F-FACE (SUVmax 2.7 ± 0.6, TNR 1.5 ± 0.4) of liver tumors was significantly lower than those of 18F-FDG uptake (6.5 ± 4.2, 2.6 ± 1.7, respectively). In qualitative analysis, 18F-FDG was positive in 4 tumors (3 HCCs, 1 CCC) and negative in the other 6 tumors, while 18F-FACE was also positive in 4 tumors which were the same tumors with positive 18F-FDG uptake.
Conclusions
Biodistribution of 18F-FACE was appropriate for oncologic imaging. Tumor 18F-FACE uptake was positive in four patients with HCC and CCC, but the uptake pattern was similar to 18F-FDG. Further evaluation was needed.





Similar content being viewed by others
References
Poeppel TD, Krause BJ, Heusner TA, Boy C, Bockisch A, Antoch G. PET/CT for the staging and follow-up of patients with malignancies. Eur J Radiol. 2009;70(3):382–92.
Metser U, Even-Sapir E. Increased (18)F-fluorodeoxyglucose uptake in benign, nonphysiologic lesions found on whole-body positron emission tomography/computed tomography (PET/CT): accumulated data from four years of experience with PET/CT. Semin Nucl Med. 2007;37(3):206–22.
Shreve PD, Anzai Y, Wahl RL. Pitfalls in oncologic diagnosis with FDG PET imaging: physiologic and benign variants. Radiographics. 1999;19(1):61–77 (Review).
Delbeke D, Martin WH, Sandler MP, Chapman WC, Wright JK Jr, Pinson CW. Evaluation of benign vs malignant hepatic lesions with positron emission tomography. Arch Surg. 1998;133:510–5.
Shiomi S, Nishiguchi S, Ishizu H, Iwata Y, Sasaki N, Tamori A, et al. Usefulness of positron emission tomography with fluorine-18-fluorodeoxyglucose for predicting outcome in patients with hepatocellular carcinoma. Am J Gastroenterol. 2001;96:1877–80.
Trojan J, Schroeder O, Raedle J, Baum RP, Herrmann G, Jacobi V, et al. Fluorine-18 FDG positron emission tomography for imaging of hepatocellular carcinoma. Am J Gastroenterol. 1999;94:3314–9.
Khan MA, Combs CS, Brunt EM, Lowe VJ, Wolverson MK, Solomon H, et al. Positron emission tomography scanning in the evaluation of hepatocellular carcinoma. J Hepatol. 2000;32:792–7.
Jeng LB, Changlai SP, Shen YY, Lin CC, Tsai CH, Kao CH. Limited value of 18F-2-deoxyglucose positron emission tomography to detect hepatocellular carcinoma in hepatitis B virus carriers. Hepatogastroenterology. 2003;50:2154–6.
Hatano E, Ikai I, Higashi T, Teramukai S, Torizuka T, Saga T, et al. Preoperative positron emission tomography with fluorine-18-fluorodeoxyglucose is predictive of prognosis in patients with hepatocellular carcinoma after resection. World J Surg. 2006;30:1736–41.
Seo S, Hatano E, Higashi T, Hara T, Tada M, Tamaki N, et al. Fluorine-18 fluorodeoxyglucose positron emission tomography predicts tumour differentiation, P-glycoprotein expression, and outcome after resection in hepatocellular carcinoma. Clin Cancer Res. 2007;13:427–33.
Ho CL, Yu SC, Yeung DW. 11C-acetate PET imaging in hepatocellular carcinoma and other liver masses. J Nucl Med. 2003;44(2):213–21.
Pike VW, Eakins MN, Allan RM, Selwyn AP. Preparation of [1-11C]acetate—an agent for the study of myocardial metabolism by positron emission tomography. Int J Appl Radiat Isot. 1982;33(7):505–12.
Swinnen JV, Van Veldhoven PP, Timmermans L, De Schrijver E, Brusselmans K, et al. Fatty acid synthase drives the synthesis of phospholipids partitioning into detergent-resistant membrane microdomains. Biochem Biophys Res Commun. 2003;302(4):898–903.
Howard BV, Howard WJ. Lipids in normal and tumor cells in culture. Prog Biochem Pharmacol. 1975;10:135–66.
Nishii R, Tong W, Wendt R 3rd, Soghomonyan S, Mukhopadhyay U, et al. Pharmacokinetics, metabolism, biodistribution, radiation dosimetry, and toxicology of (18)F-fluoroacetate ((18)F-FACE) in non-human primates. Mol Imaging Biol. 2012;14(2):213–24.
Lindhe O, Sun A, Ulin J, Rahman O, Långström B, et al. [(18)F]Fluoroacetate is not a functional analogue of [(11)C]acetate in normal physiology. Eur J Nucl Med Mol Imaging. 2009;36(9):1453–9.
Ho CL, Cheung MK, Chen S, Cheung TT, Leung YL, et al. [18F]fluoroacetate positron emission tomography for hepatocellular carcinoma and metastases: an alternative tracer for [11C]acetate? Mol Imaging. 2012;11(3):229–39.
Cheung TT, Chan SC, Ho CL, et al. Can positron emission tomography with the dual tracers [11 C] acetate and [18 F] fludeoxyglucose predict microvascular invasion in hepatocellular carcinoma? Liver Transpl. 2011;17(10):1218–25.
Cheung TT, Ho CL, Lo CM, et al. 11C-acetate and 18F-FDG PET/CT for clinical staging and selection of patients with hepatocellular carcinoma for liver transplantation on the basis of Milan criteria: surgeon’s perspective. J Nucl Med. 2013;54(2):192–200.
Ponde DE, Dence CS, Oyama N, et al. 18F-fluoroacetate: a potential acetate analog for prostate tumor imaging—in vivo evaluation of 18F-fluoroacetate versus 11C-acetate. J Nucl Med. 2007;48(3):420–8.
Clarke DD. Fluoroacetate and fluorocitrate: mechanism of action. Neurochem Res. 1991;16(9):1055–8.
Morrison JF, Peters RA. Biochemistry of fluoroacetate poisoning the effect of fluorocitrate on purified aconitase. Biochem J. 1954;58:473–99.
Muir D, Berl S, Clarke DD. Acetate and fluoroacetate as possible markers for glial metabolism in vivo. Brain Res. 1986;380(2):336–40.
Marik J, Ogasawara A, Martin-McNulty B, Ross J, Flores JE, et al. PET of glial metabolism using 2-18F-fluoroacetate. J Nucl Med. 2009;50(6):982–90.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takemoto, K., Hatano, E., Nishii, R. et al. Assessment of [18F]-fluoroacetate PET/CT as a tumor-imaging modality: preclinical study in healthy volunteers and clinical evaluation in patients with liver tumor. Ann Nucl Med 28, 371–380 (2014). https://doi.org/10.1007/s12149-014-0823-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-014-0823-z