Abstract
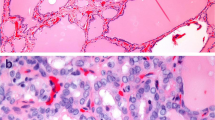
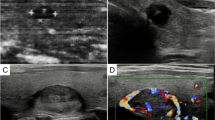
Collision tumor is the occurrence of two histologically and morphologically distinct tumors within the same organ with no histological admixture. Collision tumors of the thyroid are extremely rare constituting < 1% of all thyroid tumors. Clinical profiles and pathological features of Medullary thyroid carcinoma (MTC) and Papillary thyroid carcinoma (PTC) presenting as Collision tumors of thyroid, diagnosed between 2009 and 2019, at a tertiary care cancer center were retrospectively analyzed. Collision tumors comprised 4.7% of all MTC cases diagnosed over 10 years. A total of 21 cases (11males, 11 females, M:F = 1) were retrieved with the mean age of patients being 45.33 years (range 26–77 years). More than half of PTCs involved the right lobe of the thyroid (66.6%). About half (53.4%) of MTCs affected the left lobe. Imaging done pre-operatively failed to identify the smaller second tumor in 60% of the cases with both tumours in separate lobes. Pre-operative FNAC showed only MTC in all 8 cases in which it was done. Papillary microcarcinoma (m-PTC) was seen in 85.7% cases, with one case of multifocal m-PTC. MTC (mean size 3.12 cm), on an average, was 3 times larger than the PTC (mean size 0.91 cm). The histological variants of MTC included-oncocytic (1/21, 4.7%), spindle cell (1/21, 4.7%), epithelial (3/21, 14.2%) and classical (16/21, 76.2%) and of PTC included classic PTC (12/21, 57.14%), Hurthle cell (2/21, 9.52%), tall cell (1/21, 4.76%) and follicular variant of PTC (6/21, 28.57%). The microscopic extrathyroidal extension (ETE) due to MTC and PTC component was 42.8% and 9.5% respectively. Lymph node metastasis was seen in 16 (76.2%) cases; 87.5% (14/16) of which were contributed by MTC, 12.5% (2/16) by PTC alone, and 12.5% (2/16) cases showed metastasis from both MTC and PTC. MTC had a higher stage than PTC in 85.5% of cases. Collision tumors of the thyroid are exceedingly rare, and possibly underdiagnosed due to variation in sampling techniques, especially of the grossly “normal lobe”. The low incidence in our cohort is in favor of the “Chance theory” of co-occurrence. This diagnosis is important due to its therapeutic and prognostic implications.



Similar content being viewed by others
References
Brandwein-Gensler M, Urken M, Wang B. Collision tumor of the thyroid: a case report of metastatic liposarcoma plus papillary thyroid carcinoma. Head Neck. 2004;26(7):637–41. https://doi.org/10.1002/hed.20024.
Goyal R, Parwani AV, Gellert L, Hameed O, Giannico GA. A collision tumor of papillary renal cell carcinoma and oncocytoma: case report and literature review. Am J Clin Pathol. 2015;144(5):811–6. https://doi.org/10.1309/AJCPQ0P1YHDBZUFL.
Kim VM, Goicochea L, Fang SH. Case report: collision tumour of colon leiomyosarcoma and adenocarcinoma. J Clin Diagn Res. 2016;10(6):PD03-4. https://doi.org/10.7860/JCDR/2016/16949.7956.
Kitagawa H, Kaneko M, Kano M, Ibuki Y, Amatya VJ, Takeshima Y, Hirabayashi N, Hirota S. Coexistence of gastrointestinal stromal tumor and leiomyosarcoma of the stomach presenting as a collision tumor: a case report and review of literature. Pathol Int. 2018;68(5):313–7. https://doi.org/10.1111/pin.12662.
McGregor SM, Schoolmeester JK, Lastra RR. Collision signet-ring stromal tumor and steroid cell tumor of the ovary: report of the first case. Int J Gynecol Pathol. 2017;36(3):261–4. https://doi.org/10.1097/PGP.0000000000000321.
Abbi KK, Hameed MK, Jiang Y, De Las Casas LE, Schwann TA. Pulmonary collision tumor consisting of adenocarcinoma and typical carcinoid-a case report and review of literature. Am J Ther. 2014;21(6):e234–8. https://doi.org/10.1097/MJT.0b013e318293b0b0.
Sizemore GW. Medullary carcinoma of the thyroid gland. Semin Oncol. 1987;14(3):306–14.
Jain M, Verma D, Thomas S, Chauhan R. Mixed medullary - papillary carcinoma thyroid: an uncommon variant of thyroid carcinoma. J Lab Phys. 2014;6(2):133–5. https://doi.org/10.4103/0974-2727.141520.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, Pacini F, Randolph GW, Sawka AM, Schlumberger M, Schuff KG, Sherman SI, Sosa JA, Steward DL, Tuttle RM, Wartofsky L. 2015 American Thyroid Association Management Guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26(1):1–133. https://doi.org/10.1089/thy.2015.0020.
Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR, Winchester DP. The Eighth Edition AJCC Cancer Staging Manual Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67(2):93–9.
Mishra V, Kowtal P, Rane P, Sarin R. Genetic risk association of CDKN1A and RET gene SNPs with medullary thyroid carcinoma: results from the largest MTC cohort and meta-analysis. Cancer Med. 2019;8(13):6151–61. https://doi.org/10.1002/cam4.2443.
Liu J, Singh B, Tallini G, Carlson DL, Katabi N, Shaha A, Tuttle RM, Ghossein RA. Cancer. 2006;107(6):1255–64. https://doi.org/10.1002/cncr.22138.
Nikiforov YE, Seethala RR, Tallini G, Baloch ZW, Basolo F, Thompson LD, Barletta JA, Wenig BM, Al Ghuzlan A, Kakudo K, Giordano TJ, Alves VA, Khanafshar E, Asa SL, El-Naggar AK, Gooding WE, Hodak SP, Lloyd RV, Maytal G, Mete O, Nikiforova MN, Nosé V, Papotti M, Poller DN, Sadow PM, Tischler AS, Tuttle RM, Wall KB, LiVolsi VA, Randolph GW, Ghossein RA. Nomenclature revision for encapsulated follicular variant of papillary thyroid carcinoma: a paradigm shift to reduce overtreatment of indolent tumors. JAMA Oncol. 2016;2(8):1023–9. https://doi.org/10.1001/jamaoncol.2016.0386.
Volante M, Hunt JL, Komminoth P, et al. Mixed medullary and follicular thyroid carcinoma. In: Lloyd RV, Osamura RY, Kloppel G, Rosai J, editors., et al., World Health Organization (WHO) classification of tumours of endocrine organs. 4th ed. WHO International Agency for Research on Cancer (IARC) Press: Lyon; 2017. p. 108–16.
Walvekar RR, Kane SV, D’Cruz AK. Collision tumor of the thyroid: follicular variant of papillary carcinoma and squamous carcinoma. World J Surg Oncol. 2006;19(4):65. https://doi.org/10.1186/1477-7819-4-65.
Sung CT, Shetty A, Menias CO, Houshyar R, Chatterjee S, Lee TK, Tung P, Helmy M, Lall C. Collision and composite tumors; radiologic and pathologic correlation. Abdom Radiol (NY). 2017;42(12):2909–26. https://doi.org/10.1007/s00261-017-1200-x.
Lamberg BA, Reissel P, Stenman S, Koivuniemi A, Ekbolm M, Mäkinen J, Franssila K. Concurrent medullary and papillary thyroid carcinoma in the same thyroid lobe and in siblings. Acta Med Scand. 1981;209(5):421–4. https://doi.org/10.1111/j.0954-6820.1981.tb11620.x.
Tanaka T, Yoshimi N, Kanai N, Mori H, Nagai K, Fujii A, Sakata S, Tokimitsu N. Simultaneous occurrence of medullary and follicular carcinoma in the same thyroid lobe. Hum Pathol. 1989;20(1):83–6. https://doi.org/10.1016/0046-8177(89)90208-6.
Pastolero GC, Coire CI, Asa SL. Concurrent medullary and papillary carcinomas of thyroid with lymph node metastases. A collision phenomenon. Am J Surg Pathol. 1996;20(2):245–50. https://doi.org/10.1097/00000478-199602000-00014.
Lax SF, Beham A, Kronberger-Schönecker D, Langsteger W, Denk H. Coexistence of papillary and medullary carcinoma of the thyroid gland-mixed or collision tumour? Clinicopathological analysis of three cases. Virchows Arch. 1994;424(4):441–7. https://doi.org/10.1007/BF00190568.
Rossi S, Fugazzola L, De Pasquale L, Braidotti P, Cirello V, Beck-Peccoz P, Bosari S, Bastagli A. Medullary and papillary carcinoma of the thyroid gland occurring as a collision tumour: report of three cases with molecular analysis and review of the literature. Endocr Relat Cancer. 2005;12(2):281–9. https://doi.org/10.1677/erc.1.00901.
Brauckhoff M, Gimm O, Hinze R, Ukkat J, Brauckhoff K, Dralle H. Papillary thyroid carcinoma in patients with RET proto-oncogene germline mutation. Thyroid. 2002;12(7):557–61. https://doi.org/10.1089/105072502320288393.
Papi G, Corrado S, Pomponi MG, Carapezzi C, Cesinaro A, LiVolsi VA. Concurrent lymph node metastases of medullary and papillary thyroid carcinoma in a case with RET oncogene germline mutation. Endocr Pathol. 2003;14(3):269–76. https://doi.org/10.1007/s12022-003-0020-4.
Kim WG, Gong G, Kim EY, Kim TY, Hong SJ, Kim WB, Shong YK. Concurrent occurrence of medullary thyroid carcinoma and papillary thyroid carcinoma in the same thyroid should be considered as coincidental. Clin Endocrinol (Oxf). 2010;72(2):256–63. https://doi.org/10.1111/j.1365-2265.2009.03622.x.
Biscolla RP, Ugolini C, Sculli M, Bottici V, Castagna MG, Romei C, Cosci B, Molinaro E, Faviana P, Basolo F, Miccoli P, Pacini F, Pinchera A, Elisei R. Medullary and papillary tumors are frequently associated in the same thyroid gland without evidence of reciprocal influence in their biologic behavior. Thyroid. 2004;14(11):946–52. https://doi.org/10.1089/thy.2004.14.946.
Prades JM, Dumollard JM, Timoshenko A, Chelikh L, Michel F, Estour B, Martin C. Multinodular goiter: surgical management and histopathological findings. Eur Arch Otorhinolaryngol. 2002;259(4):217–21. https://doi.org/10.1007/s00405-002-0455-0.
Pelizzo MR, Bernante P, Toniato A, Fassina A. Frequency of thyroid carcinoma in a recent series of 539 consecutive thyroidectomies for multinodular goiter. Tumori. 1997;83(3):653–5.
Kasuga Y, Sugenoya A, Kobayashi S, Masuda H, Iida F. The outcome of patients with thyroid carcinoma and Graves’ disease. Surg Today. 1993;23(1):9–12. https://doi.org/10.1007/BF00308993.
Mazzaferri EL. Thyroid cancer and Graves’ disease. J Clin Endocrinol Metab. 1990;70(4):826–9. https://doi.org/10.1210/jcem-70-4-826.
Schwartz RW, Kenady DE, Bensema M, McGrath PC, Flueck J. Medullary thyroid cancer and Graves’ disease. Surgery. 1989;105(6):804–7.
Joshi P, Nair S, Nair D, Chaturvedi P. Incidence of occult papillary carcinoma of thyroid in Indian population: case series and review of literature. J Cancer Res Ther. 2014;10(3):693–5. https://doi.org/10.4103/0973-1482.138134.
Thomas CM, Asa SL, Ezzat S, Sawka AM, Goldstein D. Diagnosis and pathologic characteristics of medullary thyroid carcinoma-review of current guidelines. Curr Oncol. 2019;26(5):338–44. https://doi.org/10.3747/co.26.5539.
Wells SA, Asa SL, Dralle H, Elisei R, Evans DB, Gagel RF, Lee N, Machens A, Moley JF, Pacini F, Raue F, Frank-Raue K, Robinson B, Rosenthal MS, Santoro M, Schlumberger M, Shah M, Waguespack SG. American Thyroid Association Guidelines Task Force on Medullary Thyroid Carcinoma. Revised American Thyroid Association guidelines for the management of medullary thyroid carcinoma. Thyroid. 2015;25(6):567–610. https://doi.org/10.1089/thy.2014.0335.
Ryan N, Walkden G, Lazic D, Tierney P. Collision tumors of the thyroid: a case report and review of the literature. Head Neck. 2015;37(10):E125–9. https://doi.org/10.1002/hed.23936.
Funding
No funding obtained.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest to disclose.
Ethical Approval
The study complies with the medical ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thomas, A., Mittal, N., Rane, S.U. et al. Papillary and Medullary Thyroid Carcinomas Presenting as Collision Tumors: A Case Series of 21 Cases at a Tertiary Care Cancer Center. Head and Neck Pathol 15, 1137–1146 (2021). https://doi.org/10.1007/s12105-021-01323-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-021-01323-7