Abstract
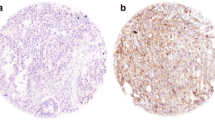
Acetylated tubulin (AT) expression has been proposed as a marker for sensitivity to taxane chemotherapy. We wanted to explore AT as a prognostic marker in squamous cell carcinoma of the head and neck (SCCHN). We assessed AT expression in archival tissue from our institutional tissue bank of primary SCCHN specimens. We also examined AT expression on pre-therapy tissues of patients with SCCHN receiving induction chemotherapy with docetaxel, cisplatin and 5FU (TPF IC). AT expression was assessed on archival cases of SCCHN with (N = 63) and without (N = 82) locoregional lymph node metastases (LNM). The predominant tumor site was oral cavity (52 %). Immunohistochemistry staining was based on staining intensity and percentage of tumor cells stained to create a weighted index (WI). A total of nine patients who received TPF IC were evaluable for response by RECIST and also had pre-therapy tissues available. A significant independent correlation between AT and tumor grade (p = 0.001) and primary location (p = 0.008) was noted. There was a trend of higher AT in patients with presence of LNM (p = 0.052) and a trend in improved OS for patients with an AT WI below the median compared to those above the median for patients with no LNM (p = 0.054). For patients treated with induction TPF, we observed an inverse correlation between AT expression and response to TPF IC (p = 0.0071). AT expression is correlated with tumor grade and primary site. There was an observed trend correlating AT with presence nodal metastases. The observed inverse correlation with response to taxane based chemotherapy needs validation in a larger sample size.



Similar content being viewed by others
References
Pignon JP, Bourhis J. Domenge C LD. Chemotherapy added to locoregional treatment for head and neck squamous-cell carcinoma: three meta-analyses of updated individual data. Lancet. 2000;355:949–55.
Pignon JP, Le MA, Bourhis J. Meta-analyses of chemotherapy in head and neck cancer (MACH-NC): an update. Int J Radiat Oncol Biol Phys. 2007;69:S112–4.
Posner MR, Hershock DM, Blajman CR, et al. Cisplatin and fluorouracil alone or with docetaxel in head and neck cancer. N Engl J Med. 2007;357:1705–15.
Vermorken JB, Remenar E, van Herpen C, et al. Cisplatin, fluorouracil, and docetaxel in unresectable head and neck cancer. N Engl J Med. 2007;357:1695–704.
Cohen EEW, Karrison T, Kocherginsky M, Huang CH, Agulnik M, Mittal BB, Yunus F, Samant S, Brockstein B, Raez LE, Mehra R, Kumar P, Ondrey FG, Seiwert TY, Villaflor VM, Haraf DJ, Vokes EE. DeCIDE: a phase III randomized trial of docetaxel (D), cisplatin (P), 5-fluorouracil (F) (TPF) induction chemotherapy (IC) in patients with N2/N3 locally advanced squamous cell carcinoma of the head and neck (SCCHN). ASCO 2013 meeting. J Clin Oncol 2012;30(Suppl):abstr 5500.
Haddad R, O’Neill A, Rabinowits G, Tishler R, Khuri F, Adkins D, Clark J, Sarlis N, Lorch J, Beitler JJ, Limaye S, Riley S, Posner M. Induction chemotherapy followed by concurrent chemoradiotherapy (sequential chemoradiotherapy) versus concurrent chemoradiotherapy alone in locally advanced head and neck cancer (PARADIGM): a randomised phase 3 trial. Lancet Oncol. 2013;14:257–64.
Marcus AI, O’Brate AM, Buey RM, et al. Farnesyltransferase inhibitors reverse taxane resistance. Cancer Res. 2006;66:8838–46.
Kauh J, Chanel-Vos C, Escuin D, et al. Farnesyl transferase expression determines clinical response to the docetaxel-lonafarnib combination in patients with advanced malignancies. Cancer. 2011;117(17):4049–59.
Marcus AI, Zhou J, O’Brate A, et al. The synergistic combination of the farnesyl transferase inhibitor lonafarnib and paclitaxel enhances tubulin acetylation and requires a functional tubulin deacetylase. Cancer Res. 2005;65:3883–93.
Tsuchida Y, Therasse P. Response evaluation criteria in solid tumors (RECIST): new guidelines. Med Pediatr Oncol. 2001;37:1–3.
Cox D. Regression models and life tables. J R Stat Soc Ser. 1972;B34:187–220.
Piperno G, LeDizet M, Chang XJ. Microtubules containing acetylated alpha-tubulin in mammalian cells in culture. J Gen Physiol. 1987;104:289–302.
Gefland VI, Bershadsky AD. Microtubule dynamics: mechanism, regulation, and function. Annu Rev Cell Biol. 1991;7:93–116.
Westermann S, Weber K. Post-translational modifications regulate microtubule function. Nat Rev Mol Cell Biol. 2003;4(12):938–48.
Wan CP, Letchford K, Jackson JK, Burt HM. The combined use of paclitaxel-loaded nanoparticles with a low-molecular-weight copolymer inhibitor of P-glycoprotein to overcome drug resistance. Int J Nanomedicine. 2013;8:379–91.
Baek JS, Cho CW. 2-Hydroxypropyl-β-cyclodextrin-modified SLN of paclitaxel for overcoming p-glycoprotein function in multidrug-resistant breast cancer cells. J Pharm Pharmacol. 2013;65:72–8.
Stordal B, Hamon M, McEneaney V, Roche S, Gillet JP, O’Leary JJ, Gottesman M, Clynes M. Resistance to paclitaxel in a cisplatin-resistant ovarian cancer cell line is mediated by P-glycoprotein. PLoS ONE. 2012;7:e40717.
Haber M, Burkhart CA, Regl DL, Madafiglio J, Norris MD, Horwitz SB. Altered expression of M beta 2, the class II beta-tubulin isotype, in a murine J774.2 cell line with a high level of taxol resistance. J Biol Chem. 1995;270:31269–75.
Jaffrezou JP, Dumontet C, Derry WB, et al. Novel mechanism of resistance to paclitaxel (Taxol) in human K562 leukemia cells by combined selection with PSC 833. Oncol Res. 1995;7:517–27.
Kavallaris M, Kuo DY, Burkhart CA, et al. Taxol-resistant epithelial ovarian tumors are associated with altered expression of specific beta-tubulin isotypes. J Clin Invest. 1997;100:1282–93.
Goncalves A, Braguer D, Kamath K, et al. Resistance to taxol in lung cancer cells associated with increased microtubule dynamics. Proc Natl Acad Sci USA. 2001;98:11737–42.
Giannakakou P, Poy G, Zhan Z, Knutsen T, Blagosklonny MV, Fojo T. Paclitaxel selects for mutant or pseudo-null p53 in drug resistance associated with tubulin mutations in human cancer. Oncogene. 2000;19:3078–85.
Giannakakou P, Sackett DL, Kang YK, et al. Paclitaxel-resistant human ovarian cancer cells have mutant beta-tubulins that exhibit impaired paclitaxel-driven polymerization. J Biol Chem. 1997;272:17118–25.
Gonzalez-Garay ML, Chang L, Blade K, Menick DR, Cabral F. A beta-tubulin leucine cluster involved in microtubule assembly and paclitaxel resistance. J Biol Chem. 1999;274:23875–82.
Berrieman HK, Lind MJ, Cawkwell L. Do beta-tubulin mutations have a role in resistance to chemotherapy? Lancet Oncol. 2004;5:158–64.
Cullen KJ, Schumaker L, Nikitakis N, Goloubeva O, Tan M, Sarlis NJ, Haddad RI, Posner. Beta-tubulin-II expression strongly predicts outcome in patients receiving induction chemotherapy for locally advanced squamous carcinoma of the head and neck: a companion analysis of the TAX 324 trial. J Clin Oncol. 2009;27:6222–8.
Acknowledgments
This work was supported by a SPORE CDP Grant P50 CA128613-03 to N.F. Saba and S. Muller and Sanofi-Aventis US; now Sanofi Oncology.
Conflict of interest
None of the authors have any conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saba, N.F., Magliocca, K.R., Kim, S. et al. Acetylated Tubulin (AT) as a Prognostic Marker in Squamous Cell Carcinoma of the Head and Neck. Head and Neck Pathol 8, 66–72 (2014). https://doi.org/10.1007/s12105-013-0476-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-013-0476-6