Abstract
Objectives
To study plasma levels of Thrombin activable fibrinolysis inhibitor (TAFI) in children with β-thalassemia major.
Methods
Fifty β-thalassemia major patients, 1.4 to 17 y of age, with number of transfusions received varying from 21 to 162 were selected at random and complete blood count (CBC), coagulation parameters [Prothrombin time (PT), Activated partial thromboplastin time (aPTT), fibrinogen, D-dimer, protein C, protein S, antithrombin, Tissue plasminogen activator (t-PA), Plasminogen activator inhibitor (PAI-1)] and TAFI were performed.
Results
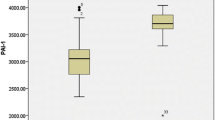
PT and aPTT were prolonged in 18 % and 30 % of cases respectively. Reduced activity of Protein C (PC) was observed in 50 % of cases and Protein S (PS) was reduced in 54 % of cases. t-PA levels were significantly higher in cases. TAFI levels were 17.24 ± 4.05 ng/ml which were significantly higher than the control group (15.01 ± 3.28; p = 0.003) No significant correlation of TAFI was observed with Hb, platelet counts, liver enzymes, serum ferritin, PC, PS, D-dimer, t-PA or PAI-1.
Conclusions
There is an ongoing subclinical activation of coagulation cascade and fibrinolytic system in thalassemia major (TM) patients. Higher levels of TAFI in the present study with no significant correlation with other parameters were noted, thus pointing out to its independent role in contribution to hypercoagulable state in thalassemia. TAFI serves as a link between two limbs of hemostasis, with its higher levels promoting inhibition of fibrinolytic system and thus promoting a hypercoagulable state. Performing TAFI levels in thalassemic patients could help to detect the early coagulopathy in these patients and hence these patients can be closely monitored for any evidence of thrombosis.

Similar content being viewed by others
References
Taher A. Iama’eel H, Cappellini MD. Thalassemia intermedia: revisited. Blood Cells Mol Dis. 2006;37:12–20.
Winichagoon P, Fucharoen S, Wasi P. Increased circulating platelet aggregates in thalassemia. Southeast Asian J Trop Med Public Health. 1981;12:556–60.
Cappellini MD. Coagulation in the pathophysiology of hemolytic anemias. Hematology Am Soc Hematol Educ Program. 2007;1:74–8.
Eldor A, Rachmilewitz EA. The hypercoagulable state in thalassemia. Blood. 2002;99:36–43.
Butthep P, Bunyaratvej A, Funahara Y, et al. Possible evidence of endothelial cell activation and disturbance in thalassemia: an in vitro study. Southeast Asian J Trop Med Public Health. 1997;28:141–8A.
Hovav T, Goldfarb A, Artmann G, Yedgar S, Barshtein G. Enhanced adherence of beta thalassemic erythrocytes to endothelial cells. Br J Haematol. 1999;106:178–81.
Naithani R, Chandra J, Narayan S, Sharma S, Singh V. Thalassemia major- on verge of bleeding or thrombosis? Haematology. 2006;11:57–61.
Mokhtar GM, Matter RM, Shawki H, Abdel Aziz MM. Thrombin activable fibrinolysis inhibitor (TAFI): relationship to haemostatic alteration in patients with beta thalassemia. Pediatr Hematol Oncol. 2010;27:363–73.
Nesheim M, Bajzar L. The discovery of TAFI. J Thromb Haemost. 2005;3:2139–46.
Mosneir LO, Bouma BN. Regulation of fibrinolysis by thrombin activable fibrinolysis inhibitor, an unstable carboxypeptidase B that unites the pathway of coagulation and fibrinolysis. Arterioscler Thromb Vasc Biol. 2006;26:2445–53.
Boffa MB, Koschinsky ML. Curiouser and curiouser: recent advances in measurements of thrombin activable fibrinolysis inhibitor (TAFI) and in understanding its molecular genetics, gene regulation and biological roles. Clin Biochem. 2007;40:431–42.
Bertina RM, Tilburg LH, Haverkate F, et al. Discovery of thrombin activable fibrinolysis inhibitor (TAFI). J Thromb Haemost. 2006;4:256–7.
Mosnier LO, Buijtenhuijs P, Marx PF, Meijers JCM, Bouma BN. Identification of thrombin activable fibrinolysis inhibitor (TAFI) in human platelets. Blood. 2003;10:4844–6.
Schadinger SL, Linn JHH, Garand M, Boffa MB. Secretion and antifibrinolytic function of thrombin- activable fibrinolysis inhibitor from human platelets. J Thromb Haemost. 2010;8:2523–9.
Zhao L, Morser J, Bajzar L, Nesheim M, Naqashima M. Identification and characterization of two thrombin activable fibrinolysis inhibitor isoforms. Thromb Hemost. 1998;80:949–55.
Bajzar L, Morser J, Neshim M. TAFI, or plasma procarboxypeptidase B, couples the coagulation and fibrinolytic cascades through the thrombin-thrombomodulin complex. J Biol Chem. 1996;271:16603–8.
Marx PF, Hackeng TM, Dawson PE, Griffin JH, Meijers JCM, Bouma BN. Inactivation of thrombin activable fibrinolysis inhibitor takes place by process that involves conformational instability rather than proteolytic cleavage. J Biol Chem. 2000;275:12410–5.
Boffa MB, Wang W, Bajzar L, Nesheim ME. Plasma and recombinant thrombin activable fibrinolysis inhibitor (TAFI) and activated TAFI compared with respect to glycosylation, thrombin/thrombomodulin dependent activation, thermal stability and enzymatic properties. J Biol Chem. 1998;273:2127–35.
Boffa MB, Bell R, Stevens WK, Nesheim ME. Roles of thermal instability and proteolytic cleavage in regulation of activated thrombin activable fibrinolysis inhibitor. J Biol Chem. 2000;275:12868–78.
Mosnier LO, von den Borne PA, Meijers JC, Bouma BN. Plasma TAFI levels influences the clot lysis time in healthy individuals in the presence of an intact intrinsic pathway of coagulation. Thromb Haemost. 1998;80:829–35.
Biswas A, Tiwari AK, Ranjan R, et al. Thrombin activable fibrinolysis inhibitor gene polymorphisms are associated with antigenic levels in the Asian-Indian population but may not be a risk for stroke. Br J Hematol. 2008;143:581–8.
Tilburg N, Rosendaal FR, Bertina RM. Thrombin activatable fibrinolysis inhibitor and risk of deep vein thrombosis. Blood. 2000;95:2855–9.
Eser M, Kement M, Balin S. Is there any role of thrombin activable fibrinolysis inhibitor in the development of a hypercoagulable state in gastric cancer? World J Surg Oncol 2012;10:180.
Caocci L, Alberti M, Burrai P, Corda R. Screening coagulation tests and clotting factors in homozygous β- thalassemia. Acta Haematol. 1978;60:358–64.
Musumeci S, Leonardi S, Dio R, Fischer A, Costa G, Protein C. And antithrombin III in polytransfused thalassemic patients. Acta Haematol. 1987;77:30–3.
Angchaisuksiri P, Atichartakarn V, Aryurachai K, et al. Hemostatic and thrombotic markers in patients with hemoglobin E/ beta- thalassemia disease. Am J Hematol. 2007;82:1001–4.
Tripatara A, Jetsrisuparb A, Teeratakulpisarn J, Kuaha K. Haemostatic alterations in splenectomized and non-splenectomized patients with β-thalassemia/ hemoglobin E disease. Thromb Res. 2007;120:805–10.
Shirahata A, Funahara Y, Opartkiattikul N, Fucharoen S, Laosombat V, Yamada K. Protein C and protein S deficiency in thalassemic patients. Southeast Asian J Trop Med Public Health. 1992;23:65–73.
Teli A, Economou M, Tzovaras F, et al. Subclinical central nervous system involvement and thrombophilic status in young thalassemia intermedia patients of Greek origin. Blood Coag. Fibrinolysis. 2012;23:195–202.
Kemhali S, Gurman C, Egin Y, et al. Hypercoagulability in children with thalassemia major. Clin Appl Thromb Haemostat. 1997;3:129–32.
Contributions
AC: Preparation of manuscript, critical analysis and statistics analysis; SS and AN: Overall supervision of the study; JC: Provided with clinical details. SS will act as guarantor for the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Source of Funding
None.
Rights and permissions
About this article
Cite this article
Chhikara, A., Sharma, S., Chandra, J. et al. Thrombin Activable Fibrinolysis Inhibitor in Beta Thalassemia. Indian J Pediatr 84, 25–30 (2017). https://doi.org/10.1007/s12098-016-2208-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-016-2208-x