Abstract
Objective
To see the efficacy and safety of 250 mg of Saccharomyces boulardii twice daily for 5 d in acute childhood diarrhea.
Methods
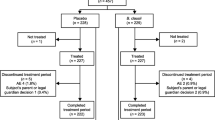
Children aged between 3 mo and 59 mo with acute onset diarrhea (of less than 48 h) admitted in DTTU (diarrhea treatment and training unit) were included and those with clinical evidence of severe malnutrition, systemic infection, encephalopathy and/or convulsion, electrolyte imbalance, invasive diarrhea or previous use of any probiotics were excluded from the study. Those included randomly were given either a placebo or Saccharomyces boullardi (SB) in identical packets mixed with puffed rice powder.
Results
Mean post intervention duration of diarrhea was significantly (95% CI = −28.13 to −5.43) shorter in SB group (52.08 ± 24.57 h) as compared to placebo group (64.04 ± 30.43 h). The time of appearance of first semi formed stool in SB group (39.48 ± 23.09 h) was significantly (95% CI −25.4 to −3.87) shorter than the placebo group (54.13 ± 28.21 h). No statistically significant difference was found in rest of the parameters.
Conclusions
There is initial evidence available that SB may have a therapeutic role in the management of acute childhood diarrhea.
Similar content being viewed by others
References
Kosek M, Bern C, Guerrant RL. The global burden of diarrhoeal disease, as estimated from studies published between 1992 and 2000. Bull World Health Organ. 2003;81:197–204.
O'Ryan M, Prado V, Pickering LK. A millennium update on pediatric diarrheal illness in the developing world. Semin Pediatr Infect Dis. 2005;16:125–36.
Huang JS, Bousvaros A, Lee JW, Diaz A, Davidson EJ. Efficacy of probiotic use in acute diarrhoea in children: a meta-analysis. Dig Dis Sci. 2002;11:2625–3634.
Van Niel CW, Feudtner C, Garrison MM, Christakis DA. Lactobacillus therapy for acute infectious diarrhoea in children:a meta-analysis. Pediatrics. 2002;109:678–84.
McFarland LV, Elmer GW, McFarland M. Meta-analysis of Probiotics for the prevention and treatment of Acute Pediatric Diarrhea. Int J Probiotics Prebiotics. 2006;1:63–76.
Cetina-Sauri G, Sierra Basto G. Therapeutic evaluation of Saccharomyces boulardii in children with acute diarrhea. Annales De Pediatrie. 1994;41:397–400.
Hafeez A, Tariq P, Ali S, Kundi ZU, Khan A, Hassan M. The efficacy of saccharomyces boulardii in the treatment of acute watery diarrhea of children: A multicentre randomized controlled trial. J Coll Physicians Surg Pak. 2002;12:432–4.
Kurugol Z, Koturoglu G. Effect of Saccharomyces boulardii in children with acute diarrhoea. Acta Pediatr. 2005;94:44–7.
Billo AG, Memon MA, Khaskheli SA, et al. Role of probiotic (Saccharomics boulardii) in management and prevention of diarrhea. World J Gastroenterol. 2006;12:4557–60.
Villarruel G, Rubio DM, Lopez F, et al. Saccharomyces boulardii in acute childhood diarrhoea: a randomized, placebo-controlled study. Acta Pediatr. 2007;96:538–41.
Htwe K, Yee KS, Tin M, Vandenplas Y. Effect of Saccharomyces boulardii in the treatment of acute watery diarrhea in Myanmar children: A randomized controlled study. Am J Trop Med Hyg. 2008;78:214–6.
Eren M, Dinleyici EC, Vandenplas Y. Clinical efficacy comparison of Saccharomyces boulardii and yogurt fluid in acute non-bloody diarrhea in children: a randomized, controlled, open label study. Am J Trop Med Hyg. 2010;82:488–91.
Le Luyer B, Makhoul G, Duhamel JF. A multicentric study of a lactose free formula supplemented with Saccharomyces boulardii in children with acute diarrhea. Arch Pediatr. 2010;17:459–65.
Conflict of Interest
None
Role of Funding Source
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Riaz, M., Alam, S., Malik, A. et al. Efficacy and Safety of Saccharomyces boulardii in Acute Childhood Diarrhea: A Double Blind Randomised Controlled Trial. Indian J Pediatr 79, 478–482 (2012). https://doi.org/10.1007/s12098-011-0573-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-011-0573-z