Abstract
Purpose
Colorectal cancer (CRC) is one most cancer type of high incidence and high mortality rate. Metastasis play an important role in survival rate and life quality of colorectal cancer patients. Nerve growth factor (NGF) has been shown to be involved in the metastasis and deterioration in many cancers, but the detail mechanisms in promoting the metastasis of colorectal cancer remain unknown. In this study, we aimed to explore the mechanism of NGF promoting colorectal cancer metastasis to provide new insights for developing NGF anti-colorectal cancer drugs.
Methods
We examined the expression of NGF in human colorectal cancer by immunohistochemical staining, and Western blot to evaluate the relationship between NGF and colorectal cancer metastasis. Using biochemical experiments including wound healing assay, transwell migration and invasion assay, RT–PCR, Western blot and ELISA to explore the relative mechanism of NGF promoting colorectal cancer cells metastasis in vivo.
Results
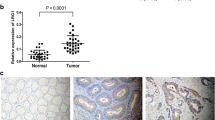
Our results found that the high expression of NGF was related with high incidence of metastasis. The binding of NGF to TrkA phosphorylated TrkA, which activated MAPK/Erk signaling pathway increasing the expression NGAL to enhance the activity of MMP2 and MMP9, promoted colorectal cancer metastasis.
Conclusion
Our finding demonstrated that NGF increased NGAL expression to enhance MMPs activity to promoted colorectal cancer cell metastasis by TrkA-MAPK/Erk axis.






Similar content being viewed by others
Data availability
Data transparency.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin. 2018;68(6):394–424. https://doi.org/10.3322/caac.21492.
Quan J, Ma C, Sun P, Wang S, Zhuang M, Liu Z, et al. Brain metastasis from colorectal cancer: clinical characteristics, timing, survival and prognostic factors[J]. Scand J Gastroenterol. 2019;54(11):1370–5.
Hosseini A, Gharibi T, Marofi F, Javadian M, Babaloo Z, Baradaran B. Janus kinase inhibitors: a therapeutic strategy for cancer and autoimmune diseases[J]. J Cell Physiol. 2020;235(9):5903–24. https://doi.org/10.1080/00365521.2019.1686056.
Denk F, Bennett DL, McMahon SB. Nerve growth factor and pain mechanisms[J]. Ann Rev Neurosci. 2017;40:307–25. https://doi.org/10.1146/annurev-neuro-072116-031121.
Chesa PG, Rettig WJ, Thomson TM, Old LJ, Melamed MR. Immunohistochemical analysis of nerve growth factor receptor expression in normal and malignant human tissues[J]. J Histochem Cytochem Official J Histochem Soc. 1988;36(4):383–9. https://doi.org/10.1177/36.4.2831267.
Chakravarthy R, Mnich K, Gorman AM. Nerve growth factor (NGF)-mediated regulation of p75(NTR) expression contributes to chemotherapeutic resistance in triple negative breast cancer cells[J]. Biochem Biophys Res Commun. 2016;478(4):1541–7. https://doi.org/10.1016/j.bbrc.2016.08.149.
Koizumi H, Morita M, Mikami S, Shibayama E, Uchikoshi T. Immunohistochemical analysis of TrkA neurotrophin receptor expression in human non-neuronal carcinomas[J]. Pathol Internat. 1998;48(2):93–101. https://doi.org/10.1111/j.1440-1827.1998.tb03877.x.
Blondy S, Christou N, David V, Verdier M, Jauberteau MO, Mathonnet M, Perraud A. Neurotrophins and their involvement in digestive cancers[J]. Cell Death Disease. 2019;10(2):123. https://doi.org/10.1038/s41419-019-1385-8.
Wang W, Chen J, Guo X. The role of nerve growth factor and its receptors in tumorigenesis and cancer pain[J]. Biosci Trends. 2014;8(2):68–74. https://doi.org/10.5582/bst.8.68.
Xhima K, Markham-Coultes K, Nedev H, Heinen S, Saragovi HU, Hynynen K, Aubert I. Focused ultrasound delivery of a selective TrkA agonist rescues cholinergic function in a mouse model of Alzheimer’s disease[J]. Sci Adv. 2020;6(4):6646. https://doi.org/10.1126/sciadv.aax6646.
Demir IE, Tieftrunk E, Schorn S, Friess H, Ceyhan GO. Nerve growth factor and TrkA as novel therapeutic targets in cancer[J]. Biochim Biophys Acta. 2016;1866(1):37–50. https://doi.org/10.1016/j.bbcan.2016.05.003.
Lin K, Huang J, Luo H, Luo C, Zhu X, Bu F, et al. Development of a prognostic index and screening of potential biomarkers based on immunogenomic landscape analysis of colorectal cancer[J]. Aging Albany NY. 2020;12(7):5832–57. https://doi.org/10.18632/aging.102979.
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition[J]. Nat Rev Mol Cell Biol. 2014;15(3):178–96. https://doi.org/10.1038/nrm3758.
Mittal V. Epithelial mesenchymal transition in tumor metastasis[J]. Annu Rev Pathol. 2018;24(13):395–412. https://doi.org/10.1146/annurev-pathol-020117-043854.
Ajafi M, Farhood B, Mortezaee K. Extracellular matrix (ECM) stiffness and degradation as cancer drivers[J]. J Cell Biochem. 2019;120(3):2782–90. https://doi.org/10.1002/jcb.27681.
Wang W, Li D, Xiang L, Lv M, Tao L, Ni T, et al. TIMP-2 inhibits metastasis and predicts prognosis of colorectal cancer via regulating MMP-9. Cell Adh Migr. 2019;13(1):273–84. https://doi.org/10.1080/19336918.2019.1639303.
Lin C, Ren Z, Yang X, Yang R, Chen Y, Liu Z, et al. Nerve growth factor (NGF)-TrkA axis in head and neck squamous cell carcinoma triggers EMT and confers resistance to the EGFR inhibitor erlotinib[J]. Cancer Lett. 2020;472:81–96. https://doi.org/10.1016/j.canlet.2019.12.015.
Romon R, Adriaenssens E, Lagadec C, Germain E, Hondermarck H, Le Bourhis X. Nerve growth factor promotes breast cancer angiogenesis by activating multiple pathways[J]. Mole Cancer. 2010;9:157. https://doi.org/10.1186/1476-4598-9-157.
Xin B, He X, Wang J, Cai J, Wei W, Zhang T, Shen X. Nerve growth factor regulates CD133 function to promote tumor cell migration and invasion via activating ERK1/2 signaling in pancreatic cancer[J]. Pancreatology. 2016;16(6):1005–14. https://doi.org/10.1016/j.pan.2016.09.005.
Wynn TA. Cellular and molecular mechanisms of fibrosis[J]. J Pathol. 2008;214(2):199–210. https://doi.org/10.1002/path.2277.
Scherer RL, McIntyre JO, Matrisian LM. Imaging matrix metalloproteinases in cancer[J]. Cancer Meta Rev. 2008;27(4):679–90. https://doi.org/10.1007/s10555-008-9152-9.
Murphy DA, Courtneidge SA. The “ins” and “outs” of podosomes and invadopodia: characteristics, formation and function[J]. Nature reviews. Mole Cell Biol. 2011;12(7):413–26. https://doi.org/10.1038/nrm3141.
Yan L, Borregaard N, Kjeldsen L, Moses MA. The high molecular weight urinary matrix metalloproteinase (MMP) activity is a complex of gelatinase B/MMP-9 and neutrophil gelatinase-associated lipocalin (NGAL). Modulation of MMP-9 activity by NGAL[J]. J Biol Chem. 2001;276(40):37258–65. https://doi.org/10.1074/jbc.M106089200.
Kubben FJ, Sier CF, Hawinkels LJ, Tschesche H, van Duijn W, Zuidwijk K, et al. Clinical evidence for a protective role of lipocalin-2 against MMP-9 auto-degradation and the impact for gastric cancer[J]. Eur J Cancer (Oxford, England: 1990). 2007;43(12):1869–76. https://doi.org/10.1016/j.ejca.2007.05.013.
Gudasheva TA, Povarnina PY, Antipova TA, Firsova YN, Konstantinopolsky MA, Seredenin SB. Dimeric dipeptide mimetics of the nerve growth factor Loop 4 and Loop 1 activate TRKA with different patterns of intracellular signal transduction[J]. J Biomed Sci. 2015;22(106):8. https://doi.org/10.1186/s12929-015-0198-z.
Garrido MP, Hurtado I, Valenzuela-Valderrama M, Salvatierra R, Hernández A, et al. NGF-enhanced vasculogenic properties of epithelial ovarian cancer cells is reduced by inhibition of the COX-2/PGE2 signaling axis[J]. Cancers. 2019;11(12):1970. https://doi.org/10.3390/cancers11121970.
Alkhadar H, Macluskey M, White S, Ellis I. Nerve growth factor-induced migration in oral and salivary gland tumor cells utilises the PI3K/Akt signaling pathway: is there a link to perineural invasion[J]? J Oral Pathol Med. 2020;49(3):227–34. https://doi.org/10.1111/jop.12979.
Emon B, Bauer J, Jain Y, Jung B, Saif T. Biophysics of tumor microenvironment and cancer metastasis - a mini review[J]. Comput Struct Biotechnol J. 2018;16:279–87. https://doi.org/10.1016/j.csbj.2018.07.003.
Martins-Filho A, Jammal MP, Micheli DC, Tavares-Murta BM, Etchebehere RM, et al. Role of intracystic cytokines and nitric oxide in ovarian neoplasms[J]. Scand J Immunol. 2017;86(6):462–70. https://doi.org/10.1111/sji.12617.
Funding
This research was funded by National Natural Science Foundation of China, grant number “81773143”; Science & Technology Department of Sichuan Province, grant number “19YYJC1252”; Health and Family Planning Commission of Sichuan Province, grant number “16PJ106/18PJ489”; The Scientific Research Fund of Chengdu Medical College, grant number “FYZX17004”.
Author information
Authors and Affiliations
Contributions
TZ and WL conceived the experiments; YL performed the experiments and drafted the manuscript; XH and HH collected sample organization analyzed the data; YH, JL and JY performed the manuscript editing and manuscript reviewing. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
Not applicable.
Ethics approval
The study involving human samples has been approved by the Ethical Committee of Chengdu Medical College (Sichuan, China) in accordance with the Helsinki Declaration. All animal experimental were approved by the Animal Ethical Committee of Chengdu Medical College (Sichuan, China).
Informed consent
Human samples and animal experimental have been approved by the Ethical Committee of Chengdu Medical College (Sichuan, China).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Tao Zhang will handle correspondence at all stages of refereeing and publication, also post-publication.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lei, Y., He, X., Huang, H. et al. Nerve growth factor orchestrates NGAL and matrix metalloproteinases activity to promote colorectal cancer metastasis. Clin Transl Oncol 24, 34–47 (2022). https://doi.org/10.1007/s12094-021-02666-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-021-02666-x