Abstract
Background
Treatment of high-grade osteosarcoma remains a challenge. The prognostic significance of the pre-treatment serum lactate dehydrogenase (LDH) level is currently controversial.
Patients and methods
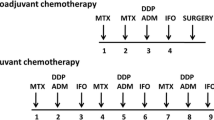
We reviewed records from all patients diagnosed with conventional high-grade osteosarcoma at our institution over a 25-year period and analysed the prognostic significance of LDH in high-grade localised extremity osteosarcomas treated with chemotherapy.
Results
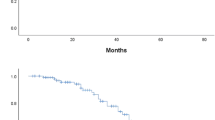
Between June 1977 and March 2003, 66 patients for whom follow-up was available were diagnosed with localised high-grade extremity osteosarcoma and treated with chemotherapy. The median age was 15 years, with only 3% older than 40 years, and the median follow-up was 100 months. The median progression-free survival (PFS) was 67 months and the median overall survival (OS) was 113 months. The absence of a response to chemotherapy was correlated with a trend toward lower PFS and OS. High serum pre-treatment LDH level was associated in multivariate analyses with a poorer prognosis for both PFS (HR=8.623, 95%CI: 1.71–43.37; p=0.009) and for OS (HR=9.38; 95%CI: 1.73–50.74; p=0.009).
Conclusion
In this series, the pre-treatment serum LDH level had an independent prognostic value for both PFS and OS in patients with high-grade localised extremity osteosarcoma. This measurement should be included in a large prospective prognostic series.
Similar content being viewed by others
References
Stillera CA, Bielack SS, Jundt G, Steliarova-Fouchere E (2006) Bone tumours in European children and adolescents, 1978–1997. Report from the Automated Childhood Cancer Information System project. Eur J Cancer 42:2124–2135
Jemal A, Siegel R, Ward E et al (2008) Cancer statistics 2008. CA Cancer J Clin 58:71–96
Dahlin DC (1978) Bone tumors: general aspects and data on 6221 cases, 3rd Edn. Charles C Thomas, Springfield, IL
Lichtenstein L (1977) Bone tumors, 5th Edn. Mosby, St Louis, MO
Jaffe HL (1958) Tumors and tumorous conditions of the bone and joints. Lea & Febiger, Philadelphia
Malawer MM, Link MP, Donaldson SS (2004) Sarcomas of bone. Cancer, principles and practice of oncology, 6th Edn. Lippincott Williams and Wilkins, Philadephia, PA pp 1981–1995
Weingard DN, Rosenberg SA (1978) Early lymphatic spread of osteogenic and soft tissue sarcomas. Surgery 84:231–240
Enneking WF, Spanier SS, Goodman MA (1980) A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop 153:106–115
Marcove RC, Rosen G (1980) En bloc resection for osteogenic sarcoma. Cancer 45:3040–3044
Malawer MM (1983) Distal femoral osteogenic sarcoma, principles of soft tissue resection and reconstruction in conjunction with prosthetic replacement (adjuvant surgery). In: Chao EYS (ed.) Design and application of tumor prosthesis for bone and joint reconstruction. Thieme-Stratton, New York, pp 297–308
Enneking WF, Dunham WK (1978) Resection and reconstruction for primary neoplasms involving the innominate bone. J Bone Joint Surg Am 60: 731–746
Rosen G, Marcove RC, Caparros B et al (1979) Primary osteogenic sarcoma. The rationale for preoperative chemotherapy and delayed survey. Cancer 43:2163–2177
Muggia F, Catani R, Lee YJ et al (1979) Factors responsible for therapeutic success in osteosarcoma. In: Jones S, Salmon S (eds) Adjuvant therapy for cancer, 2nd Edn. Grune & Stratton, New York
Cortes EP, Holland JP (1981) Adjuvant therapy for primary osteogenic sarcoma. Surg Clin North Am 61:1391–1398
Link MP (1986) Adjuvant therapy in the treatment of osteosarcoma. In: DeVita VT, Hellman S, Rosenverg SA (eds) Important advances in oncology. JB Lippincott, Philadelphia, pp 193–194
Link MP, Goorin AM, Miser AW et al (1986) The effect of adjuvant chemotherapy on relapse-free survival in patients with osteosarcoma of the extremity. N Engl J Med 314:1600–1605
Eilber F, Guiliano A, Eckardt J et al (1987) Adjuvant chemotherapy for osteosarcoma. a randomized prospective trial. J Clin Oncol 5:21–26
Rosen G, Caparrol B, Huvos AC et al (1982) Preoperative chemotherapy for osteogenic sarcoma: selection of postoperative adjuvant chemotherapy based upon the response of the primary tumor to preoperative cancer. Cancer 49:1221–1230
Ferrari S, Smeland S, Mercuri M et al (2005) Neoadjuvant chemotherapy with high-dose Ifosfamide, high-dose methotrexate, cisplatin, and doxorubicin for patients with localized osteosarcoma of the extremity: a joint study by the Italian and Scandinavian Sarcoma Groups. J Clin Oncol 23:8845–8852
Bielack SS, Kempf-Bielack B, Delling G et al (2002) Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol 20:776–790
Ger LP, Wang JW, Lin CC, Wang J (1993) Prognostic factors in 43 cases of osteosarcoma. J Formos Med Ass 92:962–968
Lopez Aguilar E, Rivera Márquez H, Cerecedo Díaz F et al (1996) Láctico dehidrogenasa como factor pronóstico en el desarrollo de enfermedad pulmonar metastásica en pacientes con osteosarcoma. Gac Med Mex 132:363
Pochanugool L, Subhadharophandou T, Danachai M et al (1997) Prognostic factors among 130 patients with osteosarcoma. Clin Orthop Relat Res 345:206–214
Goorin AM, Schwartzentruber DJ, Devidas M et al (2003) Presurgical chemotherapy with immediate surgery and adjuvant chemotherapy for nonmetastatic osteosarcoma: Pediatric Oncology Group study POG-8651. J Clin Oncol 21:1574–1580
Bacci G, Ferrari S, Sangiorgi L et al (1994) Prognostic significance of serum lactate dehydrogenase in patients with osteosarcoma of the extremities. J Chemother 6:204–210
Bacci G, Longhi A, Ferrari S et al (2004) Prognostic significance of serum lactate dehydrogenase in osteosarcoma of the extremity: experience at Rizzoli on 1421 patients treated over the last 30 years. Tumori 90:478–484
Meyers PA, Heller G, Healey J et al (1992) Chemotherapy for nonmetastatic osteogenic sarcoma: the Memorial Sloan-Kettering experience. J Clin Oncol 10:5–15
Rosen G, Murphy ML, Huvos AG et al (1976) Chemotherapy, en bloc resection and prosthetic bone replacement in the treatment of osteogenic sarcoma. Cancer 37:1–11
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Mantel M (1966) Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 50:163–170
Cox DR (1972) Regression models and life-tables. J R Stat Soc 34:187–220
Walenta S, Mueller-Klieser WF (2004) Lactate: mirror and motor of tumor malignancy. Semin Radiat Oncol 14:267–274
Albain KS, Crowley JJ, LeBlanc M, Livingston RB (1990) Determinants of improved outcome in small-cell lung cancer: an analysis of the 2580 patient Southwest Oncology Group database. J Clin Oncol 9:1563–1574
Tas F, Aykan F, Alici S et al (2001) Prognostic factors in pancreatic carcinoma: serum LDH levels predict survival in metastatic disease. Am J Clin Oncol 24:547–550
Kemeny N, Braun DW (1983) Prognostic factors in advanced colorectal carcinoma. Importance of lactic dehydrogenase level, performance status, and white blood cell count. Am J Med 74:786–794
Smaletz O, Scher HI, Small EJ et al (2002) Nomogram for overall survival of patients with progressive metastatic prostate cancer after castration. J Clin Oncol 20:3972–3982
Garcia R, Hernandez JM, Caballero MD et al (1993) Serum lactate dehydrogenase level as a prognostic factor in Hodgkin’s disease. Br J Cancer 68:1227–1231
Bacci G, Ferrari S, Longhi A et al (1999) Prognostic significance of serum LDH in Ewing’s sarcoma of bone. Oncol Rep 6:807–811
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
González-Billalabeitia, E., Hitt, R., Fernández, J. et al. Pre-treatment serum lactate dehydrogenase level is an important prognostic factor in high-grade extremity osteosarcoma. Clin Transl Oncol 11, 479–483 (2009). https://doi.org/10.1007/s12094-009-0388-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-009-0388-9