Abstract
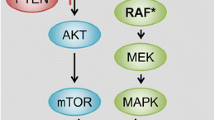
Signalling pathways that emerge from EGFR activation are critical in colon cancer (CC) biology. Its targeting with specific drugs has opened a new window in the treatment of this disease. In this regard, monoclonal antibodies (mAb) have evidenced a high degree of efficiency opposed to the uselessness of tyrosine-kinase inhibitors. Cetuximab is the mAb that has evidenced most activity in CC. After its initial approval as an irinotecan-resistance reversal agent, cetuximab has demonstrated its efficiency from the first line to heavily pretreated patients. In the first line, its addition may increase response rate to chemotherapy, improving liver metastases resection rate. Another promising approach has been suggested from combination schedules with bevacizumab. Panitumumab has been recently approved for CC. Although there is limited clinical experience, the latest data have confirmed its activity in heavily pretreated patients resulting in a clinical benefit vs. best support care. In spite of the clinical benefits, adverse events and the high sanitary cost derived from these drugs force the selection of patients with the highest probability of benefit. At the moment, when EGFR expression evidenced by immunohistochemistry has no value, skin toxicity and, fundamentally, K-Ras mutations may hint at critical information for confirmatory prospective studies.
Similar content being viewed by others
References
de Gramont A, Figer A, Seymour M et al (2000) Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 18:2938–2947
Douillard JY, Cunningham D, Roth AD et al (2000) Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet 355:1041–1047
O’Connell JB, Maggard MA, Ko CY (2004) Colon cancer survival rates with the new American Joint Committee on Cancer sixth edition staging. J Natl Cancer Inst 96:1420–1425
Hurwitz H, Fehrenbacher L, Novotny W et al (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350:2335–2342
Kabbinavar FF, Hambleton J, Mass RD et al (2005) Combined analysis of efficacy: the addition of bevacizumab to fluorouracil/leucovorin improves survival for patients with metastatic colorectal cancer. J Clin Oncol 23:3660–3662
Cunningham D, Humblet Y, Siena S et al (2004) Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med 351:337–345
Spano JP, Fagard R, Soria JC et al (2005) Epidermal growth factor receptor signaling in colorectal cancer: preclinical data and therapeutic perspectives. Ann Oncol 16:189–194
Lockhart AC, Berlin JD (2005) The epidermal growth factor receptor as a target for colorectal cancer therapy. Semin Oncol 32:52–60
Steele RJ, Kelly P, Ellul B et al (1990) Epidermal growth factor receptor expression in colorectal cancer. Br J Surg 77:1352–1354
Goldstein NS, Armin M (2001) Epidermal growth factor receptor immunohistochemical reactivity in patients with American Joint Committee on Cancer Stage IV colon adenocarcinoma: implications for a standardized scoring system. Cancer 92:1331–1346
Radinsky R, Risin S, Fan D et al (1995) Level and function of epidermal growth factor receptor predict the metastatic potential of human colon carcinoma cells. Clin Cancer Res 1:19–31
Giralt J, Eraso A, Armengol M et al (2002) Epidermal growth factor receptor is a predictor of tumor response in locally advanced rectal cancer patients treated with preoperative radiotherapy. Int J Radiat Oncol Biol Phys 54:1460–1465
Spano JP, Lagorce C, Atlan D et al (2005) Impact of EGFR expression on colorectal cancer patient prognosis and survival. Ann Oncol 16:102–108
Ranson M, Hammond LA, Ferry D et al (2002) ZD1839, a selective oral epidermal growth factor receptor-tyrosine kinase inhibitor, is well tolerated and active in patients with solid, malignant tumors: results of a phase I trial. J Clin Oncol 20:2240–2250
Herbst RS, Maddox AM, Rothenberg ML et al (2002) Selective oral epidermal growth factor receptor tyrosine kinase inhibitor ZD1839 is generally well-tolerated and has activity in non-small-cell lung cancer and other solid tumors: results of a phase I trial. J Clin Oncol 20:3815–3825
Baselga J, Rischin D, Ranson M et al (2002) Phase I safety, pharmacokinetic, and pharmacodynamic trial of ZD1839, a selective oral epidermal growth factor receptor tyrosine kinase inhibitor, in patients with five selected solid tumor types. J Clin Oncol 20:4292–4302
Rothenberg ML, LaFleur B, Levy DE et al (2005) Randomized phase II trial of the clinical and biological effects of two dose levels of gefitinib in patients with recurrent colorectal adenocarcinoma. J Clin Oncol 23:9265–9274
Chau I, Cunningham D, Hickish T et al (2007) Gefitinib and irinotecan in patients with fluoropyrimidine-refractory, irinotecan-naive advanced colorectal cancer: a phase I–II study. Ann Oncol 18:730–737
Zampino MG, Magni E, Massacesi C et al (2007) First clinical experience of orally active epidermal growth factor receptor inhibitor combined with simplified FOLFOX6 as first-line treatment for metastatic colorectal cancer. Cancer 110:752–758
Donthireddy K, Huffman L, Sandecki A et al (2006) Phase I study of oxaliplatin, capecitabine, and gefitinib in the treatment of metastatic colorectal cancer. J Clin Oncol 24:240 [abstract]
Jimeno A, Sevilla I, Gravalos C et al (2005) Phase I/II trial of capecitabine and gefitinib in patients with advanced colorectal cancer after failure of first-line therapy. J Clin Oncol 23:3176 [abstract]
Kuo T, Cho CD, Halsey J et al (2005) Phase II study of gefitinib, fluorouracil, leucovorin, and oxaliplatin therapy in previously treated patients with metastatic colorectal cancer. J Clin Oncol 23:5613–5619
Hartmann JT, Kroening H, Bokemeyer C et al (2005) Phase I study of gefitinib in combination with oxaliplatin and weekly 5-FU/FA (FUFOX) for second-/third-line treatment in patients with metastatic colorectal cancer. J Clin Oncol 23:3154 [abstract]
Veronese ML, Sun W, Giantonio B et al (2005) A phase II trial of gefitinib with 5-fluorouracil, leucovorin, and irinotecan in patients with colorectal cancer. Br J Cancer 92:1846–1849
Hochhaus A, Hofheinz R, Heike M et al (2005) Phase I study of gefitinib in combination with FOLFIRI as 2nd-/3rd-line treatment in patients with metastatic colorectal cancer. J Clin Oncol 23:3674 [abstract]
Arnold D, Constantin C, Seufferlein T et al (2005) Phase I study of gefitinib in combination with capecitabine and irinotecan for 2nd-and/or 3rd-line treatment in patients with metastatic colorectal cancer. J Clin Oncol 23:3691 [abstract]
Wolpin BM, Clark JW, Meyerhardt JA et al (2006) Phase I study of gefitinib plus FOLFIRI in previously untreated patients with metastatic colorectal cancer. Clin Colorectal Cancer 6:208–213
Meyerhardt JA, Clark JW, Supko JG et al (2007) Phase I study of gefitinib, irinotecan, 5-fluorouracil and leucovorin in patients with metastatic colorectal cancer. Cancer Chemother Pharmacol 60:661–670
Hidalgo M, Siu LL, Nemunaitis J et al (2001) Phase I and pharmacologic study of OSI-774, an epidermal growth factor receptor tyrosine kinase inhibitor, in patients with advanced solid malignancies. J Clin Oncol 19:3267–3279
Keilholz U, Arnold D, Niederle N et al (2005) Erlotinib as 2nd and 3rd line monotherapy in patients with metastatic colorectal cancer: results of a multicenter two-cohort phase II trial. J Clin Oncol 23:3575 [abstract]
Townsley C, Major P, Siu LL et al (2004) Phase II study of OSI-774 in patients with metastatic colorectal cancer. In: Gastrointestinal Cancers Symposium, p 222 [abstract]
Messersmith WA (2005) Phase I trial and biologic study of erlotinib combined with FOLFOX-4 in patients with advanced colorectal cancer: initial results. In: Gastrointestinal Cancers Symposium, p 260 [abstract]
Spigel DR, Hainsworth JD, Burris HA et al (2006) Phase II study of FOLFOX4, bevacizumab, and erlotinib as first-line therapy in patients with advanced colorectal cancer. In: Gastrointestinal Cancers Symposium, p 238 [abstract]
Meyerhardt JA, Zhu AX, Enzinger PC et al (2006) Phase II study of capecitabine, oxaliplatin, and erlotinib in previously treated patients with metastatic colorectal cancer. J Clin Oncol 24:1892–1897
Meyerhardt J, Stuart K, Fuchs C et al (2007) Phase II study of FOLFOX, bevacizumab and erlotinib as first-line therapy for patients with metastatic colorectal cancer. Ann Oncol 18:1185–1189
Messersmith WA, Laheru DA, Senzer NN et al (2004) Phase I trial of irinotecan, infusional 5-fluorouracil, and leucovorin (FOLFIRI) with Erlotinib (OSI-774): early termination due to increased toxicities. Clin Cancer Res 10:6522–6527
Di Bartolomeo M, Bajetta E, Buzzoni R et al (2006) A dose finding study with erlotinib in combination to irinotecan and capecitabine in metastatic colorectal cancer patients. J Clin Oncol 24:13075 [abstract]
Yang XD, Yang XD, Jia XC et al (2001) Development of ABX-EGF, a fully human anti-EGF receptor monoclonal antibody, for cancer therapy. Crit Rev Oncol Hematol 38:17–23
Perrotte P, Matsumoto T, Inoue K et al (1999) Anti-epidermal growth factor receptor antibody C225 inhibits angiogenesis in human transitional cell carcinoma growing orthotopically in nude mice. Clin Cancer Res 5:257–265
Shaheen RM, Ahmad SA, Liu W et al (2001) Inhibited growth of colon cancer carcinomatosis by antibodies to vascular endothelial and epidermal growth factor receptors. Br J Cancer 85:584–589
Huang SM, Li J, Harari PM (2002) Molecular inhibition of angiogenesis and metastatic potential in human squamous cell carcinomas after epidermal growth factor receptor blockade. Mol Cancer Ther 1:507–514
Prewett MC, Hooper AT, Bassi R et al (2002) Enhanced antitumor activity of anti-epidermal growth factor receptor monoclonal antibody IMC-C225 in combination with irinotecan (CPT-11) against human colorectal tumor xenografts. Clin Cancer Res 8:994–1003
Saltz LB, Meropol NJ, Loehrer PJ Sr et al (2004) Phase II trial of cetuximab in patients with refractory colorectal cancer that expresses the epidermal growth factor receptor. J Clin Oncol 22:1201–1208
Lenz HJ, Van Cutsem E, Khambata-Ford S et al (2006) Multicenter phase II and translational study of cetuximab in metastatic colorectal carcinoma refractory to irinotecan, oxaliplatin, and fluoropyrimidines. J Clin Oncol 24:4914–4921
Pessino A, Artale S, Sciallero S et al (2007) First-line single-agent cetuximab in patients with advanced colorectal cancer. Ann Oncol [Epub ahead of print]
Jonker DJ, O’Callaghan CJ, Karapetis CS et al (2007) Cetuximab for the treatment of colorectal cancer. N Engl Med 357:2040–2048
Saltz LB, Lenz HJ, Kindler HL et al (2007) Randomized phase II trial of cetuximab, bevacizumab, and irinotecan compared with cetuximab and bevacizumab alone in irinotecan-refractory colorectal cancer: the BOND-2 study. J Clin Oncol 25:4557–4561
Abubakr Y, Eng C, Pautret V et al (2006) Cetuximab plus irinotecan for metastatic colorectal cancer (mCRC): safety analysis of 800 patients in a randomized phase III trial (EPIC). J Clin Oncol 24:3556 [abstract]
Van Custem E, Nowacki M, Lang I et al (2007) Randomized phase III study of irinotecan and 5-FU/FA with or without cetuximab in the first-line treatment of patients with metastatic colorectal cancer: the CRYSTAL trial. J Clin Oncol 25:4000 [abstract]
Tabernero J, Van Cutsem E, Díaz-Rubio E et al (2007) Phase II trial of cetuximab in combination with fluorouracil, leucovorin, and oxaliplatin in the first-line treatment of metastatic colorectal cancer. J Clin Oncol 25:5225–5232
Bokemeyer C, Bondarenko I, Makhson A et al (2007) Cetuximab plus 5-FU/FA/oxaliplatin (FOLFOX-4) versus FOLFOX-4 in the first-line treatment of metastatic colorectal cancer (mCRC): OPUS, a randomized phase II study. J Clin Oncol 25:4035 [abstract]
Adam, R. Aloia T, Lévi F et al (2007) Hepatic resection after rescue cetuximab treatment for colorectal liver metastases previously refractory to conventional systemic therapy. J Clin Oncol 25:4593–4602
Van Cutsem E, Peeters M, Siena S et al (2007) Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J Clin Oncol 25:1658–1664
Hecht J, Chidiac T, Mitchell E et al (2007) An interim analysis of efficacy and safety from a randomized controlled trial of panitumumab with chemotherapy plus bevacizumab in metastatic colorectal cancer. Proceedings of the 9th World Congress on Gastrointestinal Cancer 33 [abstract]
Tejpar S, Peeters M, Humblet Y et al (2007) Phase I/II study of cetuximab dose-escalation in patients with metastatic colorectal cancer (mCRC) with no or slight skin reactions on cetuximab standard dose treatment (EVEREST): pharmacokinetic (PK), pharmacodynamic (PD) and efficacy data. J Clin Oncol 25:4037 [abstract]
Saltz LB, Meropol NJ, Loehrer Sr PJ et al (2004) Phase II trial of cetuximab in patients with refractory colorectal cancer that expresses the epidermal growth factor receptor. J Clin Oncol 22:1201–1208
Vallbohmer D, Zhang W, Gordon M et al (2005) Molecular determinants of cetuximab efficacy. J Clin Oncol 23:3536–3544
Ogino S, Meyerhardt JA, Cantor M et al (2005) Molecular alterations in tumors and response to combination chemotherapy with gefitinib for advanced colorectal cancer. Clin Cancer Res 11:6650–6656
Berlin J, Neubauer M, Swanson P et al (2006) Panitumumab antitumor activity in patients with metastatic colorectal cancer expressing ?10% epidermal growth factor receptor. J Clin Oncol 24:3548 [abstract]
Lenz HJ, Van Cutsem E, Khambata-Ford S et al (2006) Multicenter phase II and translational study of cetuximab in metastatic colorectal carcinoma refractory to irinotecan, oxaliplatin, and fluoropyrimidines. J Clin Oncol 24:4914–4921
Chung KY, Shia J, Kemeny NE et al (2005) Cetuximab shows activity in colorectal cancer patients with tumors that do not express the epidermal growth factor receptor by immunohistochemistry. J Clin Oncol 23:1803–1810
Hecht J, Mitchell E, Baranda J et al (2006) Panitumumab antitumor activity in patients with metastatic colorectal cancer expressing low (1–9%) or negative (<1%) levels of epidermal growth factor receptor. J Clin Oncol 24:3547 [abstract]
Atkins D, Reiffen KA, Tegtmeier CL et al (2004) Immunohistochemical detection of EGFR in paraffin-embedded tumor tissues: variation in staining intensity due to choice of fixative and storage time of tissue sections. J Histochem Cytochem 52:893–901
Moroni M, Veronese S, Benvenuti S et al (2005) Gene copy number for epidermal growth factor receptor (EGFR) and clinical response to antiEGFR treatment in colorectal cancer: a cohort study. Lancet Oncol 6:279–286
Sartore-Bianchi A, Moroni M, Veronese S et al (2007) Epidermal growth factor receptor gene copy number and clinical outcome of metastatic colorectal cancer treated with panitumumab. J Clin Oncol 25:3238–3245
Lievre A, Bachet JB, Le Corre D et al (2006) KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res 66:3992–3995
Barber TD, Vogelstein B, Kinzler KW et al (2004) Somatic mutations of EGFR in colorectal cancers and glioblastomas. N Engl J Med 351:2883
Nagahara H, Mimori K, Ohta M et al (2005) Somatic mutations of epidermal growth factor receptor in colorectal carcinoma. Clin Cancer Res 11:1368–1371
Tsuchihashi Z, Khambata-Ford S, Hanna N et al (2005) Responsiveness to cetuximab without mutations in EGFR. N Engl J Med 353:208–209
Ogino S, Meyerhardt JA, Cantor M et al (2005) Molecular alterations in tumors and response to combination chemotherapy with gefitinib for advanced colorectal cancer. Clin Cancer Res 11:6650–6656
Lievre A, Bachet J, Ychou M et al (2007) KRAS mutations in colorectal cancer is a predictive factor of response and progression free survival in patients treated with cetuximab. In: American Association for Cancer Research Annual Meeting, p 5671 [abstract]
Di Fiore F, Blanchard F, Charbonnier F et al (2007) Clinical relevance of KRAS mutation detection in metastatic colorectal cancer treated by Cetuximab plus chemotherapy. Br J Cancer 96:1166–1169
Amado RG, Wolf M, Freeman D et al (2007) Analysis of KRAS mutations in patients with metastatic colorectal cancer receiving panitumumab monotherapy. ECCO abstract 7LB. 14th European Cancer Conference
De Roock W, Piessevaux H, De Schutter J et al (2007) KRAS wild-type state predicts survival and is associated to early radiological response in metastatic colorectal cancer treated with cetuximab. Ann Oncol [Epub ahead of print]
Baba I, Shirasawa S, Iwamoto R et al (2000) Involvement of deregulated epiregulin expression in tumorigenesis in vivo through activated KRas signaling pathway in human colon cancer cells. Cancer Res 60:6886–6889
De Luca A, Arra C, D’Antonio A et al (2000) Simultaneous blockage of different EGF-like growth factors results in efficient growth inhibition of human colon carcinoma xenografts. Oncogene 19:5863–5871
Khambata-Ford S, Garrett CR, Meropol NJ et al (2007) Expression of epiregulin and amphiregulin and K-ras mutation status predict disease control in metastatic colorectal cancer patients treated with cetuximab. J Clin Oncol 25:3230–3237
Scartozzi M, Bearzi I, Pierantoni C et al (2007) Nuclear factor-kB tumor expression predicts response and survival in irinotecan-refractory metastatic colorectal cancer treated with cetuximab-irinotecan therapy. J Clin Oncol 25:3930–3935
Zhang W, Gordon M, Press OA et al (2006) Cyclin D1 and epidermal growth factor polymorphisms associated with survival in patients with advanced colorectal cancer treated with Cetuximab. Pharmacogenet Genomics 16:475–483
Zhang W, Gordon M, Schultheis AM et al (2007) FCGR2A and FCGR3A polymorphisms associated with clinical outcome of epidermal growth factor receptor expressing metastatic colorectal cancer patients treated with single-agent cetuximab. J Clin Oncol 25:3712–3718
Lenz H, Zhang W, Yang D et al (2007) Pharmacogenomic analysis of a randomized phase II trial (BOND 2) of cetuximab/bevacizumab/irinotecan versus cetuximab/bevacizumab in irinotecan-refractory colorectal cancer. In: Gastrointestinal Cancers Symposium
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by an unrestricted educational grant from GlaxoSmithKline.
Rights and permissions
About this article
Cite this article
de Castro-Carpeño, J., Belda-Iniesta, C., Sáenz, E.C. et al. EGFR and colon cancer: a clinical view. Clin Transl Oncol 10, 6–13 (2008). https://doi.org/10.1007/s12094-008-0147-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-008-0147-3