Abstract
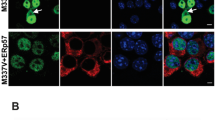
Increased cellular concentration of α-synuclein (α-syn) predisposes it to misfolding and aggregation that in turn impair the degradation pathways. This poses a limitation to the use of overexpression models for studies on α-syn clearance by autophagy, which is widely investigated for its therapeutic potential. This limitation can be overcome with the use of endogenous models. In this study, SK-MEL-28, a melanoma cell model with endogenous α-syn expression, was employed to study α-syn clearance through autophagy. We demonstrated the dual localization of α-syn to nucleus and cytoplasm that varied in response to changes in cellular environment. Autophagy inhibition and exposure to dopamine favored cytoplasmic localization of α-syn, while autophagy induction favored increased localization to the nucleus. The inhibitory effect of dopamine on autophagy was heightened in presence of α-syn. Additionally, because α-syn had a regulatory effect on autophagy, cells showed an increased resistance to autophagy induction in presence of α-syn. This resistance prevented effective induction of autophagy even under conditions of prolonged autophagy inhibition. These results highlight alternate physiological roles of α-syn, particularly in non-neuronal cells. Because autophagy enhancement could reverse neither the increase in α-syn levels nor the autophagy inhibition, there arises a need to evaluate the efficacy of autophagy-based therapeutic strategies.









Similar content being viewed by others
References
Asanuma M, Miyazaki I, Diaz-Corrales FJ et al (2013) Transplantation of melanocytes obtained from the skin ameliorates apomorphine-induced abnormal behavior in rodent hemi-parkinsonian models. PLoS One 8:e65983. doi:10.1371/journal.pone.0065983
Barbour R, Kling K, Anderson JP et al (2008) Red blood cells are the major source of alpha-synuclein in blood. Neurodegener Dis 5:55–59. doi:10.1159/000112832
Bartels T, Choi JG, Selkoe DJ (2011) α-synuclein occurs physiologically as a helically folded tetramer that resists aggregation. Nature 477:107–110. doi:10.1038/nature10324
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Colasanti T, Vomero M, Alessandri C et al (2014) Role of alpha-synuclein in autophagy modulation of primary human T lymphocytes. Cell Death Dis 5:e1265. doi:10.1038/cddis.2014.211
Ebrahimi-Fakhari D, Cantuti-Castelvetri I, Fan Z et al (2011) Distinct roles in vivo for the ubiquitin-proteasome system and the autophagy-lysosomal pathway in the degradation of α-synuclein. J Neurosci 31:14508–14520. doi:10.1523/JNEUROSCI.1560-11.2011
Emmanouilidou E, Stefanis L, Vekrellis K (2010) Cell-produced alpha-synuclein oligomers are targeted to, and impair, the 26S proteasome. Neurobiol Aging 31:953–968. doi:10.1016/j.neurobiolaging.2008.07.008
Glick D, Barth S, Macleod KF (2010) Autophagy: cellular and molecular mechanisms. J Pathol 221:3–12. doi:10.1002/path.2697
Hansen C, Angot E, Bergström A-L et al (2011) α-synuclein propagates from mouse brain to grafted dopaminergic neurons and seeds aggregation in cultured human cells. J Clin Invest 121:715–725. doi:10.1172/JCI43366
Hsu LJ, Sagara Y, Arroyo A et al (2000) alpha-synuclein promotes mitochondrial deficit and oxidative stress. Am J Pathol 157:401–410
Irizarry MC, Kim TW, McNamara M et al (1996) Characterization of the precursor protein of the non-A beta component of senile plaques (NACP) in the human central nervous system. J Neuropathol Exp Neurol 55:889–895
Kim C, Lee S-J (2008) Controlling the mass action of alpha-synuclein in Parkinson’s disease. J Neurochem 107:303–316. doi:10.1111/j.1471-4159.2008.05612.x
Kontopoulos E, Parvin JD, Feany MB (2006) Alpha-synuclein acts in the nucleus to inhibit histone acetylation and promote neurotoxicity. Hum Mol Genet 15:3012–3023. doi:10.1093/hmg/ddl243
Lashuel HA, Overk CR, Oueslati A, Masliah E (2013) The many faces of α-synuclein: from structure and toxicity to therapeutic target. Nat Rev Neurosci 14:38–48. doi:10.1038/nrn3406
Lecker SH, Goldberg AL, Mitch WE (2006) Protein degradation by the ubiquitin-proteasome pathway in normal and disease states. J Am Soc Nephrol JASN 17:1807–1819. doi:10.1681/ASN.2006010083
Lee BR, Matsuo Y, Cashikar AG, Kamitani T (2013a) Role of Ser129 phosphorylation of α-synuclein in melanoma cells. J Cell Sci 126:696–704. doi:10.1242/jcs.122093
Lee H-J, Cho E-D, Lee KW et al (2013b) Autophagic failure promotes the exocytosis and intercellular transfer of α-synuclein. Exp Mol Med 45:e22. doi:10.1038/emm.2013.45
Leng Y, Chase TN, Bennett MC (2001) Muscarinic receptor stimulation induces translocation of an alpha-synuclein oligomer from plasma membrane to a light vesicle fraction in cytoplasm. J Biol Chem 276:28212–28218. doi:10.1074/jbc.M011121200
Li L, Chen Y, Gibson SB (2013) Starvation-induced autophagy is regulated by mitochondrial reactive oxygen species leading to AMPK activation. Cell Signal 25:50–65. doi:10.1016/j.cellsig.2012.09.020
Mahmood T, Yang P-C (2012) Western blot: technique, theory, and trouble shooting. North Am J Med Sci 4:429–434. doi:10.4103/1947-2714.100998
Martinez-Vicente M, Cuervo AM (2007) Autophagy and neurodegeneration: when the cleaning crew goes on strike. Lancet Neurol 6:352–361. doi:10.1016/S1474-4422(07)70076-5
Matsuo Y, Kamitani T (2010) Parkinson’s disease-related protein, alpha-synuclein, in malignant melanoma. PLoS One 5:e10481. doi:10.1371/journal.pone.0010481
McLean JR, Hallett PJ, Cooper O et al (2012) Transcript expression levels of full-length alpha-synuclein and its three alternatively spliced variants in Parkinson’s disease brain regions and in a transgenic mouse model of alpha-synuclein overexpression. Mol Cell Neurosci 49:230–239. doi:10.1016/j.mcn.2011.11.006
Mizushima N, Yoshimori T (2007) How to interpret LC3 immunoblotting. Autophagy 3:542–545
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Murase D, Hachiya A, Takano K et al (2013) Autophagy has a significant role in determining skin color by regulating melanosome degradation in keratinocytes. J Invest Dermatol 133:2416–2424. doi:10.1038/jid.2013.165
Musgrove REJ, King AE, Dickson TC (2012) α-synuclein protects neurons from apoptosis downstream of free-radical production through modulation of the MAPK signalling pathway. Neurotox Res. doi:10.1007/s12640-012-9352-5
Nilsson R, Jain M, Madhusudhan N et al (2014) Metabolic enzyme expression highlights a key role for MTHFD2 and the mitochondrial folate pathway in cancer. Nat Commun 5:3128. doi:10.1038/ncomms4128
Oberman Z, Chayen R, Herzberg M (1970) A rapid fluorimetric method for the determination of dopamine in urine. Clin Chim Acta Int J Clin Chem 29:391–394
Oliveras-Salvá M, Van der Perren A, Casadei N et al (2013) rAAV2/7 vector-mediated overexpression of alpha-synuclein in mouse substantia nigra induces protein aggregation and progressive dose-dependent neurodegeneration. Mol Neurodegener 8:44. doi:10.1186/1750-1326-8-44
Outeiro TF, Klucken J, Bercury K et al (2009) Dopamine-induced conformational changes in alpha-synuclein. PLoS One. doi:10.1371/journal.pone.0006906
Pan T, Li X, Jankovic J (2011) The association between Parkinson’s disease and melanoma. Int J Cancer J Int Cancer 128:2251–2260. doi:10.1002/ijc.25912
Pan T, Zhu J, Hwu W-J, Jankovic J (2012) The role of alpha-synuclein in melanin synthesis in melanoma and dopaminergic neuronal cells. PLoS One 7:e45183. doi:10.1371/journal.pone.0045183
Roostaee A, Beaudoin S, Staskevicius A, Roucou X (2013) Aggregation and neurotoxicity of recombinant α-synuclein aggregates initiated by dimerization. Mol Neurodegener 8:5. doi:10.1186/1750-1326-8-5
Rubinsztein DC (2006) The roles of intracellular protein-degradation pathways in neurodegeneration. Nature 443:780–786. doi:10.1038/nature05291
Segura-Aguilar J, Paris I, Muñoz P et al (2014) Protective and toxic roles of dopamine in Parkinson’s disease. J Neurochem 129:898–915. doi:10.1111/jnc.12686
Solomon VR, Lee H (2009) Chloroquine and its analogs: a new promise of an old drug for effective and safe cancer therapies. Eur J Pharmacol 625:220–233. doi:10.1016/j.ejphar.2009.06.063
Stefanis L (2012) α-synuclein in Parkinson’s disease. Cold Spring Harb Perspect Med 2:a009399. doi:10.1101/cshperspect.a009399
Suzuki K, Bose P, Leong-Quong RY et al (2010) REAP: a two minute cell fractionation method. BMC Res Notes 3:294. doi:10.1186/1756-0500-3-294
Thoreen CC, Kang SA, Chang JW et al (2009) An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-resistant functions of mTORC1. J Biol Chem 284:8023–8032. doi:10.1074/jbc.M900301200
Uversky VN (2008) Alpha-synuclein misfolding and neurodegenerative diseases. Curr Protein Pept Sci 9:507–540
Vivacqua G, Casini A, Vaccaro R et al (2011) Different sub-cellular localization of alpha-synuclein in the C57BL\6J mouse’s central nervous system by two novel monoclonal antibodies. J Chem Neuroanat 41:97–110. doi:10.1016/j.jchemneu.2010.12.003
Vogiatzi T, Xilouri M, Vekrellis K, Stefanis L (2008) Wild type alpha-synuclein is degraded by chaperone-mediated autophagy and macroautophagy in neuronal cells. J Biol Chem 283:23542–23556. doi:10.1074/jbc.M801992200
Webb JL, Ravikumar B, Atkins J et al (2003) Alpha-synuclein is degraded by both autophagy and the proteasome. J Biol Chem 278:25009–25013. doi:10.1074/jbc.M300227200
Winslow AR, Chen C-W, Corrochano S et al (2010) α-synuclein impairs macroautophagy: implications for Parkinson’s disease. J Cell Biol 190:1023–1037. doi:10.1083/jcb.201003122
Xilouri M, Vogiatzi T, Vekrellis K et al (2009) Abberant alpha-synuclein confers toxicity to neurons in part through inhibition of chaperone-mediated autophagy. PLoS One 4:e5515. doi:10.1371/journal.pone.0005515
Zhou W, Long C, Reaney SH et al (2010) Methionine oxidation stabilizes non-toxic oligomers of alpha-synuclein through strengthening the auto-inhibitory intra-molecular long-range interactions. Biochim Biophys Acta 1802:322–330. doi:10.1016/j.bbadis.2009.12.004
Acknowledgements
Financial Assistance to Swapna Nandakumar was provided by Sree Chitra Tirunal Institute for Medical Science and Technology.
Author information
Authors and Affiliations
Contributions
SN, AK, and AT were involved in the concept and design of experiments, data interpretation, and manuscript preparation. SN and BV carried out the experiments.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest to declare.
Additional information
Asha Kishore and Anoopkumar Thekkuveettil shared equal responsibilities in the study.
Rights and permissions
About this article
Cite this article
Nandakumar, S., Vijayan, B., Kishore, A. et al. Autophagy enhancement is rendered ineffective in presence of α-synuclein in melanoma cells. J. Cell Commun. Signal. 11, 381–394 (2017). https://doi.org/10.1007/s12079-017-0402-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-017-0402-x