Abstract
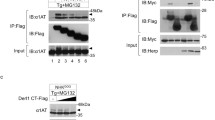
The human Ube2j1 and Ube2j2 are the only ubiquitin-conjugating enzymes (E2s) that are localized to endoplasmic reticulum (ER) through its C-terminal transmembrane domains. Ube2j1 is a known substrate of MAPK signalling pathway and it is phosphorylated at serine-184 during ER stress. Here, we demonstrate that Ube2j1, not Ube2j2 is essential for the recovery of cells from transient ER stress. The ectopic expression of wild-type Ube2j1 and phospho-mimic mutant, Ube2j1S184D but not phospho-mutant Ube2j1S184A can recover cells from ER stress. We also found that ubiquitin-ligase (E3), c-IAP1 preferentially interacts with phosphorylated Ube2j1. Moreover, we noticed that phosphorylated Ube2j1 is rapidly degraded by the proteasome during ER stress cell recovery. Taken together, these data suggest that Ube2j1 and its phosphorylation is important for transient ER stress cell recovery and the phosphorylated Ube2j1 is degraded by the proteasome.




Similar content being viewed by others
Abbreviations
- CHX:
-
Cycloheximide
- ER stress:
-
Endoplasmic reticulum stress
- ERAD:
-
Endoplasmic reticulum-associated degradation
- MAPK:
-
Mitogen-activated protein kinase
References
Biederer T, Volkwein C, Sommer T (1997) Role of Cue1p in ubiquitination and degradation at the ER surface. Science 278:1–5. doi:10.1126/science.278.5344.1806
Bonifacino JS, Weissman AM (1998) Ubiquitin and the control of protein fate in the secretory and endocytic pathways. Annu Rev Cell Dev Biol 14:19–57. doi:10.1146/annurev.cellbio.14.1.19
Burr ML, Cano F, Svobodova S et al (2011) HRD1 and UBE2J1 target misfolded MHC class I heavy chains for endoplasmic reticulum- associated degradation. Proc Natl Acad Sci U S A 108:2034–2039. doi:10.1073/pnas.1016229108
Christianson JC, Ye Y (2014) Cleaning up in the endoplasmic reticulum: ubiquitin in charge. Nat Struct Mol Biol 21:325–335. doi:10.1038/nsmb.2793
Elangovan M, Oh C, Sukumaran L et al (2010) Functional differences between two major ubiquitin receptors in the proteasome; S5a and hRpn13. Biochem Biophys Res Commun 396:425–428. doi:10.1016/j.bbrc.2010.04.108
Glickman MH, Ciechanover A (2002) The ubiquitin-proteasome proteolytic pathway : destruction for the sake of construction. Physiol Rev 82:373–428. doi:10.1152/physrev.00027.2001
Hampton RY, Gardner RG, Rine J (1996) Role of 26S proteasome and HRD genes in the degradation of 3-Hydroxy-3-Methylglutaryl-CoA reductase, an integral endoplasmic reticulum membrane protein. Mol Biol Cell 7:2029–2044. doi:10.1091/mbc.7.12.2029
Lester D, Farquharson C, Russell G, Houston B (2000) Identification of a family of Noncanonical ubiquitin- conjugating enzymes structurally related to yeast UBC6. Biochem Biophys Res Commun 269:474–480. doi:10.1006/bbrc.2000.2302
Menon MB, Tiedje C, Lafera J et al (2013) Endoplasmic reticulum-associated ubiquitin-conjugating enzyme Ube2j1 is a novel substrate of MK2 (MAPKAP kinase-2) involved in MK2-mediated TNFα production. Biochem J 456:163–172. doi:10.1042/BJ20130755
Meusser B, Hirsch C, Jarosch E, Sommer T (2005) ERAD: the long road to destruction. Nat Cell Biol 8:766–772. doi:10.1038/ncb0805-766
Oh RS, Bai X, Rommens JM (2006) Human homologs of Ubc6p ubiquitin-conjugating enzyme and phosphorylation of HsUbc6e in response to endoplasmic reticulum stress. J Biol Chem 281:21480–21490. doi:10.1074/jbc.M601843200
Oh C, Park S, Lee EK, Yoo YJ (2013) Downregulation of ubiquitin level via knockdown of polyubiquitin gene Ubb as potential cancer therapeutic intervention. Sci Rep 3:1–9. doi:10.1038/srep02623
Pickart CM (2004) Back to the future with ubiquitin review. Cell 116:181–190. doi:10.1016/S0092-8674(03)01074-2
Pilon M, Schekman R (1997) Sec61p mediates export of a misfolded secretory protein from the endoplasmic reticulum to the cytosol for degradation. EMBO J 16:4540–4548. doi:10.1093/emboj/16.15.4540
Plemper RK, Böhmler S, Bordallo J, Sommer T, Wolf DH (1997) Mutant analysis links the translocon and BiP to retrograde protein transport for ER degradation. Nature 6645:891–895. doi:10.1038/42276
Wu CJ, Conze DB, Li X, Ying S, John A, Ashwell JD (2005) TNF-alpha induced c-IAP1/TRAF2 complex translocation to a Ubc6-containing compartment. EMBO J 24:1886–1898. doi:10.1038/sj.emboj.7600649
Younger JM, Chen L, Ren H, Rosser MFN, Turnbull EL, Fan C, Patterson C, Cyr DM (2006) Sequential quality-control checkpoints triage misfolded cystic fibrosis transmembrane conductance regulator. Cell 126:571–582. doi:10.1016/j.cell.2006.06.041
Acknowledgement
This work was supported by the GIST Research Institute (GRI) in 2016.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Supplemental Figure 1
a When the relative cell proliferation was assayed by MTT assay after 36 h, it was found that the cell proliferation was inhibited 40% in Ube2j1 siRNA transfected cells (3) whereas the inhibition of cell proliferation by tunicamycin was recovered by the transfection of GFP-Ube2j1/Wt (4) and GFP-Ube2j1/S184D (6), not by GFP (3) and GFP-Ube2j1/S184A (5). b Cells were also lysed and immunoblotted with indicated antibodies. Cleaved PARP (PARPc) was observed in cells expressing GFP (lane 3) and GFP-Ube2j1/S184A (lane 5). Low level of PARPc was observed in cells expressing GFP-Ube2j1 (lane 4) and GFP-Ube2j1/S184D (lane 6). The expression level of actin was used as a loading control. The data are shown as the mean ± SD, **, p < 0.05 (TIFF 469 kb)
Rights and permissions
About this article
Cite this article
Elangovan, M., Chong, H.K., Park, J.H. et al. The role of ubiquitin-conjugating enzyme Ube2j1 phosphorylation and its degradation by proteasome during endoplasmic stress recovery. J. Cell Commun. Signal. 11, 265–273 (2017). https://doi.org/10.1007/s12079-017-0386-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-017-0386-6