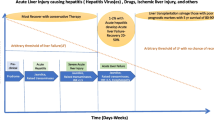
Abstract
Background
Several models have been proposed to predict acute liver failure/death in patients with drug-induced liver injury (DILI), but the predictive performances of them have not been systematically compared. We aim to compare the current models for their predictive potency of mortality at DILI onset.
Methods
DILI patients hospitalized at both Beijing Friendship Hospital and the Fifth Medical Center of PLA General Hospital were categorized into death/liver transplantation (LT) or survival without LT group. Predictive potency of 28-day, 90-day, 6-month and 12-month death/LT outcomes of Hy's Law, nHy's Law, Robles-Diaz Model, drug-induced liver toxicity (DrILTox ALF) Score, Model for End-stage Liver Disease (MELD) Score, and Ghabril Model was compared by Delong method.
Results
A total of 6.3% (83/1314) patients died or received LT within 12 months after DILI onset. The area under receiver operating characteristic of Hy's Law, nHy's Law, and Robles-Diaz Model was all lower than 0.750 for the prediction of within 12 months’ mortality. DrILTox ALF Score, MELD Score and Ghabril Model showed better predictive potency of 28-day [0.896 (0.878–0.912), 0.934 (0.919–0.947), 0.935 (0.921–0.948), respectively], 90-day [0.883 (0.864–0.899), 0.951 (0.938–0.962), 0.952 (0.939–0.963), respectively], 6-month [0.820 (0.799–0.841), 0.905 (0.888–0.921) and 0.908 (0.891–0.923), respectively] and 12-month [0.801 (0.779–0.823), 0.882 (0.863–0.899) and 0.885 (0.866–0.902), respectively] mortality.
Conclusion
Despite the difference of clinical characteristics and implicated-drug categories between China and industrialized countries, we demonstrate that MELD Score and Ghabril Model have the best predictive performance in the prediction of mortality within 12 months after DILI onset.




Similar content being viewed by others
Abbreviations
- ALF:
-
Acute liver failure
- ALP:
-
Alkaline phosphatase
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- AUROC:
-
Area under receiver operating characteristic
- BMI:
-
Body mass index
- CCI:
-
Charlson comorbidity index
- CI:
-
Confidence interval
- Cr:
-
Creatinine
- DILI:
-
Drug-induced liver injury
- DILIN:
-
Drug-induced liver injury network
- DrILTox:
-
Drug-induced liver toxicity
- GGT:
-
Gamma glutamyl transferase
- HDS:
-
Herbal and dietary supplements
- HGB:
-
Hemoglobin
- INR:
-
International normalized ratio
- LT:
-
Liver transplantation
- MELD:
-
Model for end-stage liver disease
- nHy’s Law:
-
New Hy’s Law
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs
- PLT:
-
Platelets
- RUCAM:
-
Roussel Uclaf Causality Assessment Method
- TBA:
-
Total bile acid
- TB:
-
Total bilirubin
- ULN:
-
Upper limit of normal
- WBC:
-
White blood cell
References
Ostapowicz G, Fontana RJ, Schiodt FV, et al. Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Ann Intern Med. 2002;137:947–954
Reuben A, Koch DG, Lee WM, et al. Drug-induced acute liver failure: results of a U.S. multicenter, prospective study. Hepatology. 2010;52:2065–2076
Koch DG, Speiser JL, Durkalski V, et al. The natural history of severe acute liver injury. Am J Gastroenterol. 2017;112:1389–1396
Wei G, Bergquist A, Broome U, et al. Acute liver failure in Sweden: etiology and outcome. J Intern Med. 2007;262:393–401
Chalasani N, Fontana RJ, Bonkovsky HL, et al. Causes, clinical features, and outcomes from a prospective study of drug-induced liver injury in the United States. Gastroenterology. 2008;135:1924-1934e1-4
Chalasani N, Bonkovsky HL, Fontana R, et al. Features and outcomes of 899 patients with drug-induced liver injury: the DILIN Prospective Study. Gastroenterology. 2015;148(1340–52): e7
Andrade RJ, Lucena MI, Fernandez MC, et al. Drug-induced liver injury: an analysis of 461 incidences submitted to the Spanish registry over a 10-year period. Gastroenterology. 2005;129:512–521
Hassan A, Fontana RJ. The diagnosis and management of idiosyncratic drug-induced liver injury. Liver Int. 2019;39:31–41
Tujios SR, Lee WM. Acute liver failure induced by idiosyncratic reaction to drugs: challenges in diagnosis and therapy. Liver Int. 2018;38:6–14
Zimmerman HJ. The spectrum of hepatotoxicity. Perspect Biol Med. 1968;12:135–161
Robles-Diaz M, Lucena MI, Kaplowitz N, et al. Use of Hy’s law and a new composite algorithm to predict acute liver failure in patients with drug-induced liver injury. Gastroenterology. 2014;147:109-118e5
Lo Re III V, Haynes K, Forde KA, et al. Risk of acute liver failure in patients with drug-induced liver injury: evaluation of Hy’s Law and a new prognostic model. Clin Gastroenterol Hepatol. 2015;13:2360–2368
Jeong R, Lee YS, Sohn C, et al. Model for end-stage liver disease score as a predictor of short-term outcome in patients with drug-induced liver injury. Scand J Gastroenterol. 2015;50:439–446
Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–383
Ghabril M, Gu J, Yoder L, et al. Development and validation of a model consisting of comorbidity burden to calculate risk of death within 6 months for patients with suspected drug-induced liver injury. Gastroenterology. 2019;157:1245-1252e3
Yu YC, Mao YM, Chen CW, et al. CSH guidelines for the diagnosis and treatment of drug-induced liver injury. Hepatol Int. 2017;11:221–241
Hayashi PH, Rockey DC, Fontana RJ, et al. Death and liver transplantation within 2 years of onset of drug-induced liver injury. Hepatology. 2017;66:1275–1285
Ishak K, Baptista A, Bianchi L, et al. Histological grading and staging of chronic hepatitis. J Hepatol. 1995;22:696–699
Malinchoc M, Kamath PS, Gordon FD, et al. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology. 2000;31:864–871
Shen T, Liu Y, Shang J, et al. Incidence and etiology of drug-induced liver injury in mainland China. Gastroenterology. 2019;156:2230-2241.e11
Wang L, Wang Y, Wee A, et al. Clinicopathological features of Bu Gu Zhi-induced liver injury, a long-term follow-up cohort study. Liver Int. 2020;40:571–580
Li H, Wang X, Liu Y, et al. Hepatoprotection and hepatotoxicity of Heshouwu, a Chinese medicinal herb: context of the paradoxical effect. Food Chem Toxicol. 2017;108:407–418
Li C, Rao T, Chen X, et al. HLA-B*35:01 allele is a potential biomarker for predicting Polygonum multiflorum-induced liver injury in humans. Hepatology. 2019;70:346–357
Jung KA, Min HJ, Yoo SS, et al. Drug-induced liver injury: twenty five cases of acute hepatitis following ingestion of Polygonum multiflorum Thunb. Gut Liver. 2011;5:493–499
Cardenas A, Restrepo JC, Sierra F, et al. Acute hepatitis due to Shen-Min: a herbal product derived from Polygonum multiflorum. J Clin Gastroenterol. 2006;40:629–632
Wang Y, Wang L, Saxena R, et al. Clinicopathological features of He Shou Wu-induced liver injury: this ancient anti-aging therapy is not liver-friendly. Liver Int. 2019;39:389–400
Zhu L, Zhang CY, Li DP, et al. Tu-San-Qi (Gynura japonica): the culprit behind pyrrolizidine alkaloid-induced liver injury in China. Acta Pharmacol Sin. 2021;42:1212–1222
Devarbhavi H, Singh R, Patil M, et al. Outcome and determinants of mortality in 269 patients with combination anti-tuberculosis drug-induced liver injury. J Gastroenterol Hepatol. 2013;28:161–167
Schmidt LE, Larsen FS. MELD score as a predictor of liver failure and death in patients with acetaminophen-induced liver injury. Hepatology. 2007;45:789–796
Reike-Kunze M, Zenouzi R, Hartel J, et al. Drug-induced liver injury at a tertiary care centre in Germany: model for end-stage liver disease is the best predictor of outcome. Liver Int. 2021;41:2383–2395
Biggins SW, Kim WR, Terrault NA, et al. Evidence-based incorporation of serum sodium concentration into MELD. Gastroenterology. 2006;130:1652–1660
Luca A, Angermayr B, Bertolini G, et al. An integrated MELD model including serum sodium and age improves the prediction of early mortality in patients with cirrhosis. Liver Transpl. 2007;13:1174–1180
Chalasani NP, Maddur H, Russo MW, et al. ACG clinical guideline: diagnosis and management of idiosyncratic drug-induced liver injury. Am J Gastroenterol. 2021;116:878–898
Funding
This study was supported by grants from the National Natural Science Foundation of China (No. 81900526).
Author information
Authors and Affiliations
Contributions
Study concept and design: XZ and ZZ. Acquisition and interpretation: YW, CZ, ZM, TG, YW, LL. Drafting and revision of the manuscript: YW, CZ, XZ, AW, JL, JJ. Statistical analysis: YW, ML. Study supervision: XZ.
Corresponding authors
Ethics declarations
Conflict of interest
Yan Wang, Cailun Zou, Aileen Wee, Jimin Liu, Zikun Ma, Tiantian Guo, Min Li, Yu Wang, Liwei Liu, Jidong Jia, Zhengsheng Zou, and Xinyan Zhao declare that they have no competing interests.
Ethical approval
The protocol was reviewed and approved by the Institutional Review Board with waiver for informed consent (No.2020-P2-300-01).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Zou, C., Wee, A. et al. Comparison of the prognostic models for mortality in idiosyncratic drug-induced liver injury. Hepatol Int 17, 488–498 (2023). https://doi.org/10.1007/s12072-022-10405-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-022-10405-9