Abstract
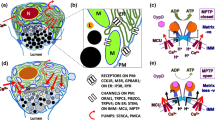
Ascites represents the most common decompensating event in patients with liver cirrhosis. The appearance of ascites is strongly related to portal hypertension, which leads to splanchnic arterial vasodilation, reduction of the effective circulating volume, activation of endogenous vasoconstrictor systems, and avid sodium and water retention in the kidneys. Bacterial translocation further worsens hemodynamic alterations of patients with cirrhosis and ascites. The first-line treatment of uncomplicated ascites is a moderate sodium-restricted diet combined with diuretic treatment. In patients who develop refractory ascites, paracentesis plus albumin represents the most feasible option. Transjugular intrahepatic portosystemic shunt placement is a good alternative for selected patients. Other treatments such as vasoconstrictors and automated low-flow pumps are two potential options still under investigations. Ascites is associated with a high risk of developing further complications of cirrhosis such as dilutional hyponatremia, spontaneous bacterial peritonitis and/or other bacterial infections and acute kidney injury (AKI). Hepatorenal syndrome (HRS) is the most life-threatening type of AKI in patients with cirrhosis. The most appropriate medical treatment in patients with AKI-HRS is the administration of vasoconstrictors plus albumin. Finally, ascites impairs both the quality of life and survival in patients with cirrhosis. Thus, all patients with ascites should be evaluated for the eligibility for liver transplantation. The aim of this article is to review the management of patients with cirrhosis, ascites and HRS.
Similar content being viewed by others
Abbreviations
- AKI:
-
Acute kidney injury
- HRS:
-
Hepatorenal syndrome
- ECV:
-
Effective circulating volume
- SNS:
-
Sympathetic nervous system
- RAAS:
-
Renin angiotensin aldosterone system
- PAMPs:
-
Pathogen-associated molecular patterns
- NO:
-
Nitric oxide
- CO:
-
Carbon monoxide
- SBP:
-
Spontaneous bacterial peritonitis
- SAAG:
-
Serum albumin ascites gradient
- HBV:
-
Hepatitis B virus
- HCV:
-
Hepatitis C virus
- RCT:
-
Randomized controlled trial
- PICD:
-
Paracentesis induced circulatory dysfunction
- TIPS:
-
Transjugular intrahepatic portosystemic shunt
- LT:
-
Liver transplantation
- MELD:
-
Model of end stage liver disease
- GFR:
-
Glomerular filtration rate
- AVP:
-
Arginine vasopressin
- FDA:
-
Food and Drug Administration
- MDR:
-
Multi-drug resistant
- TNF-α:
-
Tumor necrosis factor alpha
- iNOS:
-
Inducible nitric oxide synthase
- ACLF:
-
Acute on chronic liver failure
- sCr:
-
Serum creatinine
- TLR4:
-
Toll like receptor 4
- LPS:
-
Lipopolysaccharide
- DAMPs:
-
Danger associated molecular patterns
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs
- ATN:
-
Acute tubular necrosis
- NGAL:
-
Neutrophil gelatinase-associated lipocalin
- RRT:
-
Renal replacement therapy
- SKL:
-
Simultaneous kidney liver transplantation
- KDIGO:
-
Kidney disease improving global outcomes
References
Ginés P, Quintero E, Arroyo V, et al. Compensated cirrhosis: natural history and prognostic factors. Hepatology 1987;7:122–128
Schrier RW, Arroyo V, Bernardi M, et al. Peripheral arteriolar vasodilation hypothesis: a proposal for the initiation of renal sodium and water retention in cirrhosis. Hepatology 1988;8:151–1157
Arroyo V, Terra C, Ginès P. Advances in the pathogenesis and treatment of type-1 and type-2 hepatorenal syndrome. J Hepatol 2007;46:935–946
Angeli P, Gatta A, Caregaro L et al. Tubular site of renal sodium retention in ascitic liver cirrhosis evaluated by lithium clearance. Eur J Clin Invest 1990;20:111–117
Bernardi M, Moreau R, Angeli P et al. Mechanisms of decompensation and organ failure in cirrhosis: from peripheral arterial vasodilation to systemic inflammation hypothesis. J Hepatol 2015;63:1272–1284
Tazi KA, Moreau R, Hervé P, et al. Norfloxacin reduces aortic NO synthases and proinflammatory cytokine up-regulation in cirrhotic rats: role of Akt signaling. Gastroenterology 2005;129:303–314
Angeli P, Fernández-Varo G, Dalla Libera V, et al. The role of nitric oxide in the pathogenesis of systemic and splanchnic vasodilation in cirrhotic rats before and after the onset of ascites. Liver Int 2005;25:429–437
Yang YY, Liu H, Nam SW et al. Mechanisms of TNFalpha-induced cardiac dysfunction in cholestatic bile duct-ligated mice: interaction between TNFalpha and endocannabinoids. J Hepatol 2010;53:298–306
Planas R, Montoliu S, Ballesté B, et al. Natural history of patients hospitalized for management of cirrhotic ascites. Clin Gastroenterol Hepatol 2006;4:1385–1394
Fasolato S, Angeli P, Dallagnese L et al. Renal failure and bacterial infections in patients with cirrhosis: epidemiology and clinical features. Hepatology 2007;45:223–229
Cárdenas A, Ginès P, Uriz J, et al. Renal failure after upper gastrointestinal bleeding in cirrhosis: incidence, clinical course, predictive factors, and short-term prognosis. Hepatology 2001;34:671–676
Runyon BA. Management of adult patients with ascites due to cirrhosis: an update. Hepatology 2009; 50: 2087–2107
Ginès P, Angeli P, Lenz K, et al. EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol 2010;53:397–417
Runyon BA, Montano AA, Akriviadis EA, et al. The serum-ascites albumin gradient is superior to the exudate-transudate concept in the differential diagnosis of ascites. Ann Intern Med 1992;117:215–220
Rimola A, Garcia-Tsao G, Navasa M, et al. Diagnosis, treatment and prophylaxis of spontaneous bacterial peritonitis: a consensus document. International Ascites Club. J Hepatol 2000;32:142–153
Morando F, Rosi S, Gola E, et al. Adherence to a moderate sodium restriction diet in outpatients with cirrhosis and ascites: a real-life cross-sectional study. Liver Int 2015;35:1508–1515
Bernardi M, Servadei D, Trevisani F, et al. Importance of plasma aldosterone concentration on natriuretic effect of spironolactone in patients with liver cirrhosis and ascites. Digestion 1985;31:189–193
Pérez-Ayuso RM, Arroyo V, Planas R, et al. Randomized comparative study of efficacy of furosemide versus spironolactone in nonazotemic cirrhosis with ascites. Gastroenterology 1984;84:961–968
Angeli P, Dalla Pria M, De Bei E, et al. Randomized clinical study of the efficacy of amiloride and potassium canrenoate in nonazotemic cirrhotic patients with ascites. Hepatology 1994;19:72–79
Gatta A, Angeli P, Caregaro L, et al. A pathophysiological interpretation of unresponsiveness to spironolactone in a stepped-care approach to the diuretic treatment of ascites in nonazotemic cirrhotic patients with ascites. Hepatology 1991;14:231–236
Santos J, Planas R, Pardo A, et al. Spironolactone alone or in combination with furosemide in the treatment of moderate ascites in nonazotemic cirrhosis. A randomized comparative study of efficacy and safety. J Hepatol 2003;39:187–192
Angeli P, Fasolato S, Mazza E, et al. Combined versus sequential diuretic treatment of ascites in non-azotemic patients with cirrhosis: results of an open randomised clinical trial. Gut 2010;59:98–104
Gines P, Arroyo V, Quintero E, et al. Comparison of paracentesis and diuretics in the treatment of cirrhotics with tense ascites. Results of a randomized study. Gastroenterology 1987;92:234–241
Simon DM, McCain JR, Bonkovsky HL, et al. Effects of therapeutic paracentesis on systemic and hepatic hemodynamics and on renal and hormonal function. Hepatology 1987;7:423–429
Ruiz del Arbol L, Monescillo A, Jimenez W, et al. Paracentesis-induced circulatory dysfunction: mechanism and effect on hepatic hemodynamics in cirrhosis. Gastroenterology 1997;113:579–586
Gines A, Fernandez-Esparrach G, Monescillo A, et al. Randomized controlled trial comparing albumin, dextran-70 and polygelin in cirrhotic patients with ascites treated by paracentesis. Gastroenterology 1996;111:1002–1010
Bernardi M, Caraceni P, Navickis RJ, et al. Albumin infusion in patients undergoing large-volume paracentesis: a meta-analysis of randomized trials. Hepatology 2012; 55:1172–1181
Caraceni P, Riggio O, Angeli P, et al. Long-term albumin administration improves survival in patients with decompensated cirrhosis: final results of the “ANSWER” study. J Hepatol 2017;66:S93 (abstract)
Morando F, Maresio G, Piano S, et al. How to improve care in outpatients with cirrhosis and ascites: a new model of care coordination by consultant hepatologists. J Hepatol 2013;59:257–264
Arroyo V, Gines P, Gerbes AL, et al. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. Hepatology 1996;23:164–176
Moreau R, Delegue P, Pessione F, et al. Clinical characteristics and outcome of patients with cirrhosis and refractory ascites. Liver Int 2004;24:457–464
Lebrec D, Giuily N, Hadengue A, et al. Transjugular intrahepatic portosystemic shunts: comparison with paracentesis in patients with cirrhosis and refractory ascites: a randomized trial. J Hepatol 1996;25:135–44
Rössle M, Ochs A, Gülberg V, et al. A comparison of paracentesis and transjugular intrahepatic portosystemic shunting in patients with ascites. N Engl J Med 2000;342:1701–1707
Gines P, Uriz J, Calahorra B, et al. Transjugular intrahepatic portosystemic shunting versus paracentesis plus albumin for refractory ascites in cirrhosis. Gastroenterology 2002;123:1839–1847
Salerno F, Merli M, Riggio O, et al. Randomized controlled study of TIPS versus paracentesis plus albumin in cirrhosis with severe ascites. Hepatology 2004;40:629–635
Bureau C, Thabut D, Oberti F, et al. Transjugular intrahepatic portosystemic shunts with covered stents increase transplant-free survival of patients with cirrhosis and recurrent ascites. Gastroenterology 2017;152:157–163
Saab S, Nieto JM, Lewis SK, et al. TIPS versus paracentesis for cirrhotic patients with refractory ascites. Cochrane Database Syst Rev 2006; CD004889
Salerno F, Camma C, Enea M, et al. Transjugular intrahepatic portosystemic shunt for refractory ascites: a meta-analysis of individual patient data. Gastroenterology 2007;133:825–834
Cillo U, Burra P, Mazzaferro V, et al. A multistep, consensus-based approach to organ allocation in liver transplantation: toward a “Blended Principle Model”. Am J Transplant 2015;15:2552–2561
Angeli P, Volpin R, Piovan D, et al. Acute effects of the oral administration of midodrine, an alpha-adrenergic agonist, on renal hemodynamics and renal function in cirrhotic patients with ascites. Hepatology 1998;28:937–943
Singh V, Dhungana SP, Singh B, et al. Midodrine in patients with cirrhosis and refractory or recurrent ascites: a randomized pilot study. J Hepatol 2012;56:348–354
Fimiani B, Guardia DD, Puoti C, et al. The use of terlipressin in cirrhotic patients with refractory ascites and normal renal function: a multicentric study. Eur J Intern Med 2011;22:587–590
Gow PJ, Ardalan ZS, Vasudevan A, et al. Outpatient terlipressin infusion for the treatment of refractory ascites. Am J Gastroenterol 2016;111:1041–1042
Bellot P, Welker MW, Soriano G, et al. Automated low flow pump system for the treatment of refractory ascites: a multi-center safety and efficacy study. J Hepatol 2013;58:922–927
Adebayo D, Bureau C, De Rieu MC, et al. Alphapump system versus large volume paracentesis in the treatment of refractory ascites; results from a multicentre randomised controlled clinical study. J Hepatol 2016;64:(Suppl. 2) S185 (abstract)
Angeli P, Wong F, Watson H, et al. Hyponatremia in cirrhosis: results of a survey. Hepatology 2006;44:1535–1542
Porcel A, Diaz F, Rendom P, et al. Dilutional hyponatremia in patients with cirrhosis and ascites. Arch Intern Med 2002;162:323–328
Ginès P, Guevara M. Hyponatremia in cirrhosis: pathogenesis, clinical significance, and management. Hepatology 2008;48:1002–1010
Restuccia T, Gomez-Anson B, Guevara M, et al. Effects of dilutional hyponatremia on brain organic osmolytes and water content in patients with cirrhosis. Hepatology 2004:39;1613–1622
Guevara M, Baccaro ME, Torre A, et al. Hyponatremia is a risk factor of hepatic encephalopathy in patients with cirrhosis: a prospective study with time dependent analysis. Am J Gastroenterol 2009;1004:1382–1389
Arroyo V, Rodes J, Gutierrez-Lizarraga MA. Prognostic value of spontaneous hyponatremia in cirrhosis with ascites. Dig Dis 1976;21:249–256
Kim WR, Biggins SW, Kremers WK, et al. Hyponatremia and mortality among patients on the liver-transplant waiting list. N Engl J Med 2008;359:1018–1026
Solà E, Watson H, Graupera I, et al. Factors related to quality of life in patients with cirrhosis and ascites: relevance of serum sodium concentration and leg edema. J Hepatol 2012;57:1199–1206
McCormick PA, Mistry P, Kaye G, et al. Intravenous albumin infusion is an effective therapy for hyponatremia in cirrhotic patients with ascites. Gut 1990;31:204–207
Minhtri K, Nguyen MK, Ornekian V, Kao L, Butch AW, Kurtz I. Defining the role of albumin infusion in cirrhosis-associated hyponatremia. Am J Physiol 2014;307:G229–G232
Wong F, Watson H, Gerbes A, et al. Satavaptan for the management of ascites in cirrhosis: efficacy and safety across the spectrum of ascites severity. Gut 2012;61:108–116
Schrier RW, Gross P, Gheorghiade M, et al for the SALT Investigators. Tolvaptan a selective oral vasopressin V2-receptor antagonist, for hyponatremia. New Engl J Med 2006:355–420
Cardenas A, Gines P, Marotta P, et al. Tolvaptan, an oral vasopressin antagonist, in the treatment of hyponatremia in cirrhosis. J Hepatol 2012;56:571–578
Ahluwalia V, Heuman DM, Feldman G, et al. Correction of hyponatremia improves cognition, quality of life, and brain oedema in cirrhosis. J Hepatol 2015;62:75–82
Pose E, Solà E, Piano S, et al. Limited efficacy of tolvaptan in patients with cirrhosis and severe hyponatremia: real-life experience. Am J Med 2017;130:372–375
Wiest R, Lawson M, Geuking M. Pathological bacterial translocation in liver cirrhosis. J Hepatol 2014;60:197–209
Follo A, Llovet JM, Navasa M, et al. Renal impairment after spontaneous bacterial peritonitis in cirrhosis: course, predictive factors and prognosis. Hepatology 1994;20:1495–1501
Karvellas CJ, Abraldes JG, Arabi YM, et al. Appropriate and timely antimicrobial therapy in cirrhotic patients with spontaneous bacterial peritonitis-associated septic shock: a retrospective cohort study. Aliment Pharmacol Ther 2015;41:747–757
Felisart J, Rimola A, Arroyo V, Perez-Ayuso RM, Quintero E, Gines P, et al. Cefotaxime is more effective than is ampicillin-tobramycin in cirrhotics with severe infections. Hepatology 1985;5:457–462
Fernández J, Acevedo J, Castro M, Garcia O, de Lope CR, Roca D, et al. Prevalence and risk factors of infections by multiresistant bacteria in cirrhosis: a prospective study. Hepatology 2012;55:1551–1561
Ricart E, Soriano G, Novella MT, Ortiz J, Sàbat M, Kolle L, et al. Amoxicillin-clavulanic acid versus cefotaxime in the therapy of bacterial infections in cirrhotic patients. J Hepatol 2000;32:596–602
Piano S, Fasolato S, Salinas F, Romano A, Tonon M, Morando F, et al. The empirical antibiotic treatment of nosocomial spontaneous bacterial peritonitis: results of a randomized, controlled clinical trial. Hepatology 2016;63:1299–1309
Jalan R, Fernandez J, Wiest R, Schnabl B, Moreau R, Angeli P, et al. Bacterial infections in cirrhosis: a position statement based on the EASL Special Conference 2013. J Hepatol 2014;60:1310–1324
Sort P, Navasa M, Arroyo V, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med 1999;341:403–409
Fernández J, Monteagudo J, Bargallo X, et al. A randomized unblinded pilot study comparing albumin versus hydroxyethyl starch in spontaneous bacterial peritonitis. Hepatology 2005;42:627–634
Bortoluzzi A, Ceolotto G, Gola E, et al. Positive cardiac inotropic effect of albumin infusion in rodents with cirrhosis and ascites: molecular mechanisms. Hepatology 2013;57:266–276
Arroyo V, García-Martinez R, Salvatella X. Human serum albumin, systemic inflammation, and cirrhosis. J Hepatol 2014;61:396–407
Gines P, Rimola A, Planas R, et al. Norfloxacin prevents spontaneous bacterial peritonitis recurrence in cirrhosis: results of a double-blind, placebo-controlled trial. Hepatology 1990;12:716–724
Soriano G, Guarner C, Tomas A, et al. Norfloxacin prevents bacterial infection in cirrhotics with gastrointestinal hemorrhage. Gastroenterology 1992;103:1267–1272
Fernández J, Ruiz del Arbol L, Gómez C, et al. Norfloxacin vs ceftriaxone in the prophylaxis of infections in patients with advanced cirrhosis and hemorrhage. Gastroenterology 2006;131:1049–1056
Fernández J, Navasa M, Planas R, et al. Primary prophylaxis of spontaneous bacterial peritonitis delays hepatorenal syndrome and improves survival in cirrhosis. Gastroenterology 2007;133:818–824
Salerno F, Gerbes A, Gines P, et al. Diagnosis, prevention and treatment of the hepatorenal syndrome in cirrhosis a consensus workshop of the International Ascites Club. Gut 2007;56:1310–1318
Piano S, Rosi S, Maresio G, et al. Evaluation of the Acute Kidney Injury Network criteria in hospitalized patients with cirrhosis and ascites. J Hepatol 2013;59:482–489
Moreau R, Lebrec D. Acute renal failure in patients with cirrhosis: perspectives in the age of MELD. Hepatology 2003;37:233–243
Moreau R, Jalan R, Gines P, et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology 2013;144:1426–1437
Shah N, Mohamed FE, Jover-Cobos M, et al. Increased renal expression and urinary excretion of TLR4 in acute kidney injury associated with cirrhosis. Liver Int 2013;33:398–409
Shah N, Dhar D, El Zahraa Mohammed F, et al. Prevention of acute kidney injury in a rodent model of cirrhosis following selective gut decontamination is associated with reduced renal TLR4 expression. J Hepatol 2012;56:1047–1053
Trawalé JM, Paradis V, Rautou PE, et al. The spectrum of renal lesions in patients with cirrhosis: a clinicopathological study. Liver Int 2010;30:725–732
Angeli P, Gines P, Wong F, et al. Diagnosis and management of acute kidney injury in patients with cirrhosis: revised consensus recommendations of the International Club of Ascites. J Hepatol 2015;62:968–974
Navasa M, Follo A, Filella X, et al. Tumor necrosis factor and interleukin-6 in spontaneous bacterial peritonitis in cirrhosis: relationship with the development of renal impairment and mortality. Hepatology 1998;27:1227–1232
Clària J, Stauber RE, Coenraad MJ, et al. CANONIC Study Investigators of the EASL-CLIF Consortium and the European Foundation for the Study of Chronic Liver Failure (EF-CLIF). Systemic inflammation in decompensated cirrhosis: characterization and role in acute-on-chronic liver failure. Hepatology 2016;64:1249–1264
Gomez H, Ince C, De Backer D, et al. A unified theory of sepsis-induced acute kidney injury: inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaptation to injury. Shock 2014;41:3–11
Angeli P, Tonon M, Pilutti C, et al. Sepsis-induced acute kidney injury in patients with cirrhosis. Hepatol Int 2016;10:115–123
Piano S, Schmidt HH, Ariza X, et al. Impact of acute-on-chronic liver failure on response to treatment with terlipressin and albumin in patients with type 1 hepatorenal syndrome. J Hepatol 2017;66:S572 (Abstract)
Fagundes C, Pépin MN, Guevara M, et al. Urinary neutrophil gelatinase-associated lipocalin as biomarker in the differential diagnosis of impairment of kidney function in cirrhosis. J Hepatol 2012;57:267–273
Belcher JM, Sanyal AJ, Peixoto AJ, et al. Kidney biomarkers and differential diagnosis of patients with cirrhosis and acute kidney injury. Hepatology 2014;60:622–632
Boyer TD, Sanyal AJ, Garcia-Tsao G, et al. Impact of liver transplantation on the survival of patients treated for hepatorenal syndrome type 1. Liver Transplant 2011;17(11):1328–1332
Angeli P, Gines P. Hepatorenal syndrome, MELD score and liver transplantation: an evolving issue with relevant implications for clinical practice. J Hepatol 2012;57(5):1135–1140
Angeli P, Volpin R, Gerunda G, et al. Reversal of type 1 hepatorenal syndrome with the administration of midodrine and octreotide. Hepatology 1999;29:1690–1697
Singh V, Ghosh S, Singh B, et al. Noradrenaline vs. terlipressin in the treatment of hepatorenal syndrome: a randomized study. J Hepatol 2012;56:1293–1298
Sharma P, Kumar A, Shrama BC, et al. An open label, pilot, randomized controlled trial of noradrenaline versus terlipressin in the treatment of type 1 hepatorenal syndrome and predictors of response. Am J Gastroenterol 2008;103:1689–1697
Cavallin M, Kamath PS, Merli M, et al. Terlipressin plus albumin versus midodrine and octreotide plus albumin in the treatment of hepatorenal syndrome: a randomized trial. Hepatology 2015;62:567–574
Cavallin M, Piano S, Romano A, et al. Terlipressin given by continuous intravenous infusion versus intravenous boluses in the treatment of hepatorenal syndrome: a randomized controlled study. Hepatology 2016;63:983–992
Piano S, Morando F, Fasolato S, et al. Continuous recurrence of type 1 hepatorenal syndrome and long-term treatment with terlipressin and albumin: a new exception to MELD score in the allocation system to liver transplantation? J Hepatol 2011;55:491–496
Vasudevan A, Ardalan Z, Gow P, et al. Efficacy of outpatient continuous terlipressin infusions for hepatorenal syndrome. Hepatology 2016;64:316–318
Wong F, Pantea L, Sniderman K. Midodrine, octreotide, albumin, and TIPS in selected patients with cirrhosis and type 1 hepatorenal syndrome. Hepatology 2004;40:55–64
Guevara M, Ginès P, Bandi JC, et al. Transjugular intrahepatic portosystemic shunt in hepatorenal syndrome: effects on renal function and vasoactive systems. Hepatology 1998;28:416–422
Nadim MK, Durand F, Kellum JA, et al. Management of the critically ill patient with cirrhosis: a multidisciplinary perspective. J Hepatol 2016;64:717–735
Nadim MK, Sung RS, Davis CL, et al. Simultaneous liver-kidney transplantation summit: current state and future directions. Am J Transplant 2012;12:2901–2908
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S.P. and M.T. have nothing to disclose. P.A.: scientific advisory board of Sequana Medical.
Rights and permissions
About this article
Cite this article
Piano, S., Tonon, M. & Angeli, P. Management of ascites and hepatorenal syndrome. Hepatol Int 12 (Suppl 1), 122–134 (2018). https://doi.org/10.1007/s12072-017-9815-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-017-9815-0