Abstract
Background
Radiofrequency ablation (RFA) for hepatocellular carcinoma (HCC) is a thermoablative technique to kill tumor tissue by generating areas of coagulative necrosis. Recent reports have raised concern that RFA may lead to a local recurrence of HCC with an aggressive phenotype and unfavorable prognosis, suggesting that RFA may induce further malignant transformation of HCC. However, the biological effects of RFA on HCC cells have not been directly analyzed. The aim of this study was to determine whether heat stress of the type associated with RFA induces malignant transformation of HCC.
Methods
We assessed the sensitivity of three HCC cell lines (HepG2, Alexander, and Huh7) to heat treatment for 10 min. We then determined the temperature at which a heat-resistant subline can be generated. We established and expanded sublines that survived heat treatment. And their proliferation rates, heat sensitivities, and invasive capacities were further examined.
Results
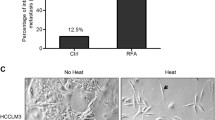
All HepG2 died after 48°C treatment, whereas 49°C treatment was required to kill all Alexander and HuH7. We generated 20 sublines for each parental cell line. A HepG2 subline, HepG2#18, proliferated 100% faster than parental HepG2. Moreover, HepG2#18 survived after 50°C treatment, whereas all parental HepG2 died after heat treatments at 48°C or higher.
Conclusion
Our results showed that even a single heat treatment could induce further transformation of an HCC cell line. Our results suggest that an insufficient treatment of HCC by RFA that enables survival of some cells might induce further malignant transformation in vivo.






Similar content being viewed by others
References
Befeler AS, Di Bisceglie AM. Hepatocellular carcinoma: diagnosis and treatment. Gastroenterology 2002;122:1609–19.
Buscarini L, Buscarini E. Therapy of HCC-radiofrequency ablation. Hepatogastroenterology 2001;48:15–9.
Livraghi T, Lazzaroni S, Meloni F. Radiofrequency thermal ablation of hepatocellular carcinoma. Eur J Ultrasound 2001;13:159–66.
Lopez PM, Villanueva A, Llovet JM. Systematic review: evidence-based management of hepatocellular carcinoma—an updated analysis of randomized controlled trials. Aliment Pharmacol Ther 2006;23:1535–47.
Marrero JA. Hepatocellular carcinoma. Curr Opin Gastroenterol 2006;22:248–53.
Scudamore CH, Lee SI, Patterson EJ, Buczkowski AK, July LV, Chung SW, et al. Radiofrequency ablation followed by resection of malignant liver tumors. Am J Surg 1999;177:411–7.
Goldberg SN, Gazelle GS, Compton CC, Mueller PR, Tanabe KK. Treatment of intrahepatic malignancy with radiofrequency ablation: radiologic-pathologic correlation. Cancer 2000;88:2452–63.
Itoh T, Orba Y, Takei H, Ishida Y, Saitoh M, Nakamura H, et al. Immunohistochemical detection of hepatocellular carcinoma in the setting of ongoing necrosis after radiofrequency ablation. Mod Pathol 2002;15:110–5.
Morimoto M, Sugimori K, Shirato K, Kokawa A, Tomita N, Saito T, et al. Treatment of hepatocellular carcinoma with radiofrequency ablation: radiologic-histologic correlation during follow-up periods. Hepatology 2002;35:1467–75.
Nicoli N, Casaril A, Marchiori L, Mangiante G, Hasheminia AR. Treatment of recurrent hepatocellular carcinoma by radiofrequency thermal ablation. J Hepatobiliary Pancreat Surg 2001;8:417–21.
Seki T, Tamai T, Ikeda K, Imamura M, Nishimura A, Yamashiki N, et al. Rapid progression of hepatocellular carcinoma after transcatheter arterial chemoembolization and percutaneous radiofrequency ablation in the primary tumour region. Eur J Gastroenterol Hepatol 2001;13:291–4.
Koda M, Maeda Y, Matsunaga Y, Mimura K, Murawaki Y, Horie Y. Hepatocellular carcinoma with sarcomatous change arising after radiofrequency ablation for well-differentiated hepatocellular carcinoma. Hepatol Res 2003;27:163–7.
Portolani N, Tiberio GA, Ronconi M, Coniglio A, Ghidoni S, Gaverini G, et al. Aggressive recurrence after radiofrequency ablation of liver neoplasms. Hepatogastroenterology 2003;50:2179–84.
Takada Y, Kurata M, Ohkohchi N. Rapid and aggressive recurrence accompanied by portal tumor thrombus after radiofrequency ablation for hepatocellular carcinoma. Int J Clin Oncol 2003;8:332–5.
Ruzzenente A, Manzoni GD, Molfetta M, Pachera S, Genco B, Donataccio M, et al. Rapid progression of hepatocellular carcinoma after Radiofrequency Ablation. World J Gastroenterol 2004;10:1137–40.
Kojiro M, Sugihara S, Kakizoe S, Nakashima O, Kiyomatsu K. Hepatocellular carcinoma with sarcomatous change: a special reference to the relationship with anticancer therapy. Cancer Chemother Pharmacol 1989;23(Suppl):S4–8.
van Rijn J, van den Berg J, Schamhart DH, van Wijk R. Morphological response and survival of hepatoma cells during fractionated hyperthermia: effect of glycerol. Radiat Res 1984;98:471–8.
Schamhart DH, van Walraven HS, Wiegant FA, Linnemans WA, van Rijn J, van den Berg J, et al. Thermotolerance in cultured hepatoma cells: cell viability, cell morphology, protein synthesis, and heat-shock proteins. Radiat Res 1984;98:82–95.
Wiegant FA, van Bergen en Henegouwen PM, van Dongen G, Linnemans WA. Stress-induced thermotolerance of the cytoskeleton of mouse neuroblastoma N2A cells and rat Reuber H35 hepatoma cells. Cancer Res 1987;47:1674–80.
Coad JE, Kosari K, Humar A, Sielaff TD. Radiofrequency ablation causes ‘thermal fixation’ of hepatocellular carcinoma: a post-liver transplant histopathologic study. Clin Transplant 2003;17:377–84.
Mertyna P, Hines-Peralta A, Liu ZJ, Halpern E, Goldberg W, Goldberg SN. Radiofrequency ablation: variability in heat sensitivity in tumors and tissues. J Vasc Interv Radiol 2007;18:647–54.
Knowles BB, Howe CC, Aden DP. Human hepatocellular carcinoma cell lines secrete the major plasma proteins and hepatitis B surface antigen. Science 1980;209:497–9.
Alexander JJ, Bey EM, Geddes EW, Lecatsas G. Establishment of a continuously growing cell line from primary carcinoma of the liver. S Afr Med J 1976;50:2124–8.
Nakabayashi H, Taketa K, Miyano K, Yamane T, Sato J. Growth of human hepatoma cells lines with differentiated functions in chemically defined medium. Cancer Res 1982;42:3858–63.
Van Rijn J, Van den Berg J, Souren JE, Van Wijk R, Joenje H. Hepatoma cells adapted to proliferate under normally lethal hyperthermic stress conditions show rapid decay of thermoresistance and heat shock protein synthesis when returned to 37 degrees C. Int J Hyperthermia 1995;11:697–708.
Sala M, Llovet JM, Vilana R, Bianchi L, Sole M, Ayuso C, et al. Barcelona Clinical Cancer Group. Initial response to percutaneous ablation predicts survival in patients with hepatocellular carcinoma. Hepatology 2004;40:1352–60.
Morimoto M, Numata K, Sugimori K, Shirato K, Kokawa A, Oka H, et al. Successful initial ablation therapy contributes to survival in patients with hepatocellular carcinoma. World J Gastroenterol 2007;13:1003–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Obara, K., Matsumoto, N., Okamoto, M. et al. Insufficient radiofrequency ablation therapy may induce further malignant transformation of hepatocellular carcinoma. Hepatol Int 2, 116–123 (2008). https://doi.org/10.1007/s12072-007-9040-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-007-9040-3