Abstract
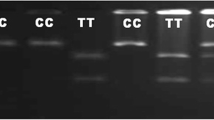
Paratuberculosis is one of the chronic granulomatous enteritis that predominantly affects ruminants worldwide, caused by Mycobacterium avium ssp. paratuberculosis (MAP). In ruminants, microsatellite polymorphisms of the \(3^\prime \) untranslated region (\(3^\prime \)UTR) of the solute carrier family 11 member A1 (SLC11A1) gene were associated with resistance to intracellular pathogen infections. This research was carried out to detect the polymorphisms in A and B regions of the \(3^{\prime }\)UTR of SLC11A1 gene and to evaluate the potential association between these polymorphisms and MAP infection in goats. MAP-specific antibodies were detected by ELISA and MAP infection was confirmed by IS900 PCR in 150 adult goats from different regions of Kerala, India. The polymorphism of microsatellite regions A and B at \(3^{\prime }\)UTR of the SLC11A1 gene was analysed in goats by an automated technique, fragment analysis, using fluorescent-tagged forward primers. Eight alleles with sizes ranging from 221 to 239 bp were found in region A. Region B revealed two alleles, 117 bp (\(\hbox {B}_{7}\)) and 119 bp (\(\hbox {B}_{8}\)). Animals with \(\hbox {B}_{8}\) alleles were found to have higher incidence of paratuberculosis than animals with \(\hbox {B}_{7}\) alleles (\(P< 0.01\)). There was no statistically significant association found between region A genotypes and paratuberculosis incidence. These results suggest that caprine SLC11A1 gene has significant role in paratuberculosis resistance in goats and further studies might help in development of a PCR-based genotyping test for paratuberculosis resistance and selection of superior animals for future goat breeding programmes.
Similar content being viewed by others
References
Awomoyi A. A. 2007 The human solute carrier family 11 member 1 protein (SLC11A1): linking infections, autoimmunity and cancer. FEMS Immunol. Med. Microbiol. 49, 324–329.
Barthel R., Feng J., Piedrathia J. A., Mcmurray D. N., Templeton J. W. and Adams L. G. 2001 Stable transfection of the bovine NRAMP1 gene into murine RAW 264.7 cells: effect on Brucella abortus survival. Infect. Immun. 69, 3110–3119.
Bellamy R., Ruwende C., Corrah T., McAdam K. P. W. J., Whittle H. C. and Hill A. V. S. 1998 Variation in the NRAMP1 gene is associated with susceptibility to tuberculosis in West Africans. N. Engl. J. Med. 338, 640–644.
Borriello G., Capparelli R., Bianco M., Fenizia D., Alfano F., Capuano F. et al. 2006 Genetic resistance to Brucella abortus in the water buffalo (Bubalus bubalis). Infect. Immun. 74, 2115–2120.
Braunstein M., Bardarov S. S. and Jacobs Jr W. R. 2002 Genetic methods for deciphering virulence determinants of Mycobacterium tuberculosis. Methods Enzymol. 358, 67–99.
Capparelli R., Alfano F., Amoroso M. G., Borriello G., Fenizia D., Bianco A. et al. 2007 Protective effect of the Nramp1 BB genotype against Brucelia abortus in water buffalo (Bubalus bubalis). Infect. Immun. 75, 988–996.
Chiodini R. J., Kruininjen H. J. and Merkal R. S. 1984 Ruminant paratuberculosis (Johne’s disease): the current status and future prospects. Cornell Vet. 74, 218–262.
Forbes J. R. and Gros P. 2003 Iron, manganese, and cobalt transport by Nramp1 (Slc11a1) and Nramp2 (Slc11a2) expressed at the plasma membrane. Blood 102, 1884–1892.
Goswami T., Bhattacharjee A., Babal P., Searle S., Moore E., Li M. and Blackwell J. M. 2001 Natural-resistance-associated macrophage protein 1 is an H+/bivalent cation antiporter. Biochem. J. 354, 511–519.
Iacoboni P. A., Hasenauer F. C., Caffaro M. E., Gaido A., Rossetto C., Neumann R. D. et al. 2014 Polymorphisms at the 3\(^{\prime } \) untranslated region of SLC11A1 gene are associated with protection to Brucella infection in goats. Vet. Immunol. Immunopathol. 160, 230–234.
Halldorsdottir S., Englund S., Nilsen S. F. and Olsaker I. 2002 Detection of Mycobacterium avium subsp. paratuberculosis by buoyant density centrifugation, sequence capture PCR and dot blot hybridization. Vet. Microbiol. 87, 327–340.
Kadarmideen H. N., Ali A. A., Thomson P. C., Muller B. and Zinsstag J. 2011 Polymorphisms of the SLC11A1 gene and resistance to bovine tuberculosis in African Zebu cattle. Anim. Genet. 42, 656–658.
Korou L. M., Liandris E., Gazouli M. and Ikonomopoulos J. 2010 Investigation of the association of the SLC11A1 gene with resistance/sensitivity of goats (Capra hircus) to paratuberculosis. Vet. Microbiol. 144, 353–358.
Liandris E., Gazouli M. and Ikonomopoulos J. 2009 Characterization of the caprine (Capra hircus) SLC11A1 gene: innate resistance to paratuberculosis. Online J. Vet. Res. 13, 41–52.
Martínez R., Dunner S., Barrera G. and Cañon J. 2008 Novel variants within the coding regions of the Slc11a1 gene identified in Bos taurus and Bos indcus breeds. J. Anim. Breed. Genet. 125, 57–62.
Pinedo P. J., Buergelt C. D., Donovan G. A., Melendez P., Morel L., Wu R. et al. 2009 Candidate gene polymorphisms (BoIFNG, TLR4, SLC11A1) as risk factors for paratuberculosis infection in cattle. Prev. Vet. Med. 91, 189–196.
Piras G., Pazzola M., Balia F., Pira E., Dettori M. L., Carcangiu V. and Vacca G. M. 2011 Polymorphism of caprine SLC11A1 gene and relationships with hygienic characteristics of milk. Agric. Conspec. Sci. 76, 175–178.
Reddacliff L. A., Beh K., McGregor H. and Whittington R. J. 2005 A preliminary study of possible genetic influences on the susceptibility of sheep to Johne’s disease. Aust. Vet. J. 83, 435–441.
Singh S. V., Singh A. V., Singh P. K. and Sohal J. S. 2008a Prevalence of juvenile capri tuberculosis and genotyping of Mycobacterium avium subsp. paratuberculosis recovered from postnatal kids. Indian J. Anim. Sci. 79, 235–238.
Singh A. V., Singh S. V., Makharia G. K., Singh P. K. and Sohal J. S. 2008b Presence and characterization of Mycobacterium avium subspecies paratuberculosis from clinical and suspected cases of Crohn’s disease and in the healthy human population in India. Int. J. Infect. Dis. 12, 190–197.
Taka S., Liandris E., Gazouli M., Sotirakoglou K., Theodoropoulos G., Bountouri M. et al. 2013 In vitro expression of the SLC11A1 gene in goat monocyte-derived macrophages challenged with Mycobacterium avium subsp paratuberculosis. Infect. Genet. Evol. 17, 8–15.
Taka S., Gazouli M., Sotirakoglou K., Liandis E., Andreadou M., Triantaphyllopoulos K. and Ikonomopoulos J. 2015 Functional analysis of 3’UTR polymorphisms in the caprine SLC11A1 gene and its association with the Mycobacterium avium subsp. paratuberculosis infection. Vet. Immunol. Immunopathol. 167, 75–79.
Thomas N. and Joseph S. 2012 Role of SLC11A1 gene in disease resistance. Biotechnol. Anim. Husbandry 28, 99–106.
Vacca G. M., Pazzola M., Pisano C., Carcangiu V., Diaz M. L., Nieddu M. et al. 2011 Chromosomal localization and genetic variation of the SLC11A1 gene in goats (Capra hircus). Vet. J. 190, 60–65.
Vidal S. M., Malo D., Vogan K., Skamene E. and Gros P. 1993 Natural resistance to infection with intracellular parasites: isolation of a candidate for Bcg. Cell 73, 469–485.
Acknowledgements
The authors acknowledge Kerala Veterinary and Animal Sciences University, Kerala, India, for providing the financial support and laboratory facilities for the successful completion of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: Silvia Garagna
Rights and permissions
About this article
Cite this article
Abraham, A., Naicy, T., Raghavan, K.C. et al. Evaluation of the association of SLC11A1 gene polymorphism with incidence of paratuberculosis in goats. J Genet 96, 641–646 (2017). https://doi.org/10.1007/s12041-017-0820-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12041-017-0820-9